Transdermal delivery of drugs
Reading File
Finding Sources
Finding Sources
Searching Images
transdermal drug delivery skin patch mechanism
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Transdermal Drug Delivery
Transdermal drug delivery (TDD) involves the application of drugs to the skin surface for absorption into the systemic circulation. It is an important route for both local cutaneous therapy and systemic drug administration.
The Skin Barrier
The primary obstacle to transdermal absorption is the stratum corneum (10–20 microns thick) — a densely packed layer of keratin-filled corneocytes embedded in a lipid matrix. Beneath it lies the viable epidermis (50–100 microns) and dermis (1–2 mm), the latter containing the superficial capillaries that provide systemic uptake.
Conventional TDD is a passive process governed by Fick's Law: flux (J) is proportional to the concentration gradient across the barrier.
J = Kp × Cv
where Kp is the permeability coefficient (dependent on diffusivity D, partition coefficient Km, and path length L), and Cv is the drug concentration in the vehicle.
Pathways of Skin Penetration
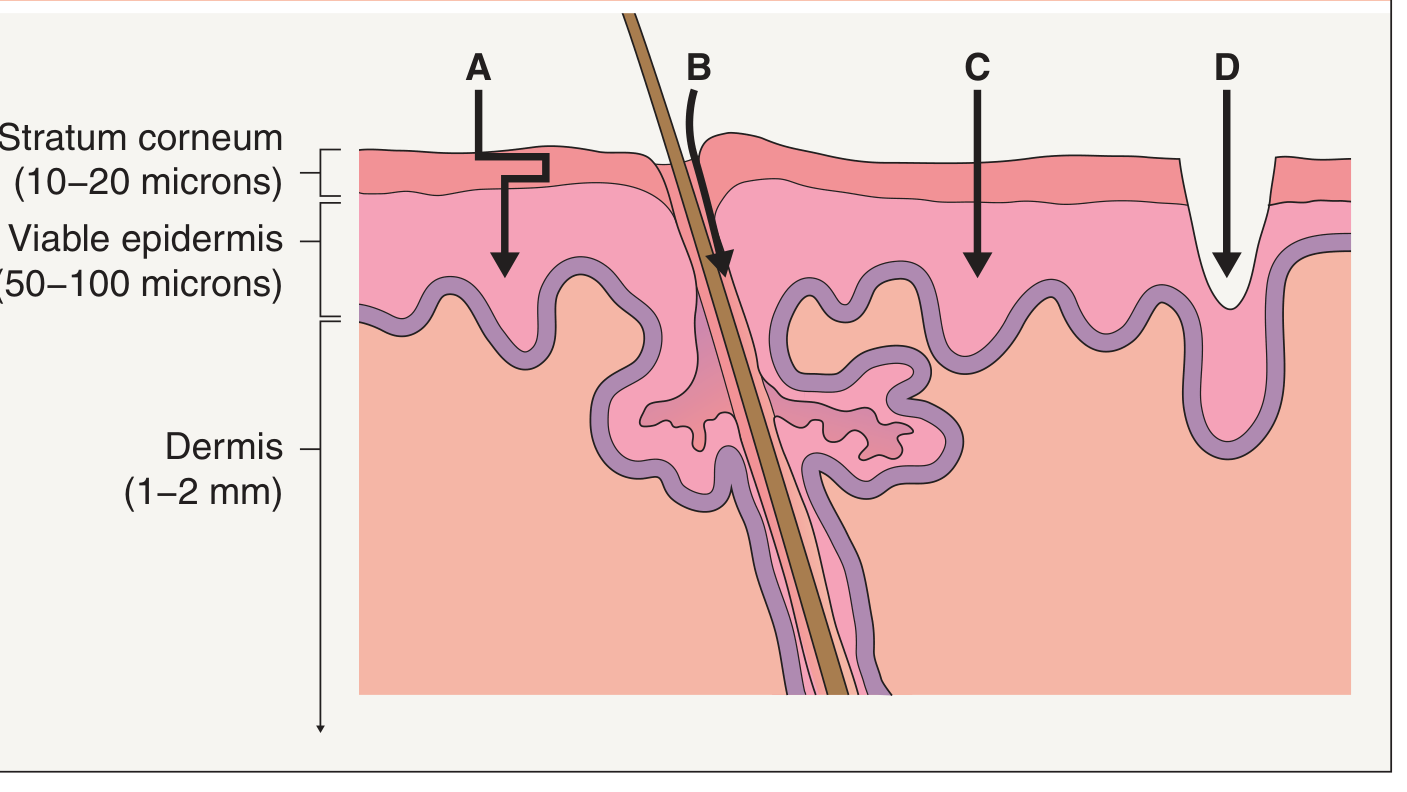
Four major pathways exist:
| Pathway | Description |
|---|---|
| A — Intercellular (tortuous lipid) | Winding path through extracellular lipids of the stratum corneum; used by most passive and chemical-enhancer methods |
| B — Follicular/appendageal | Through hair follicles and sweat ducts; enhanced by iontophoresis and some particle formulations |
| C — Transcellular (electroporation) | Direct crossing of corneocytes; enabled by electroporation |
| D — Microchannel | Physical disruption (microneedles, ablation, abrasion, fractional photothermolysis) creates pores bypassing the stratum corneum |
— Dermatology 2-Volume Set, 5e
Theoretical Advantages
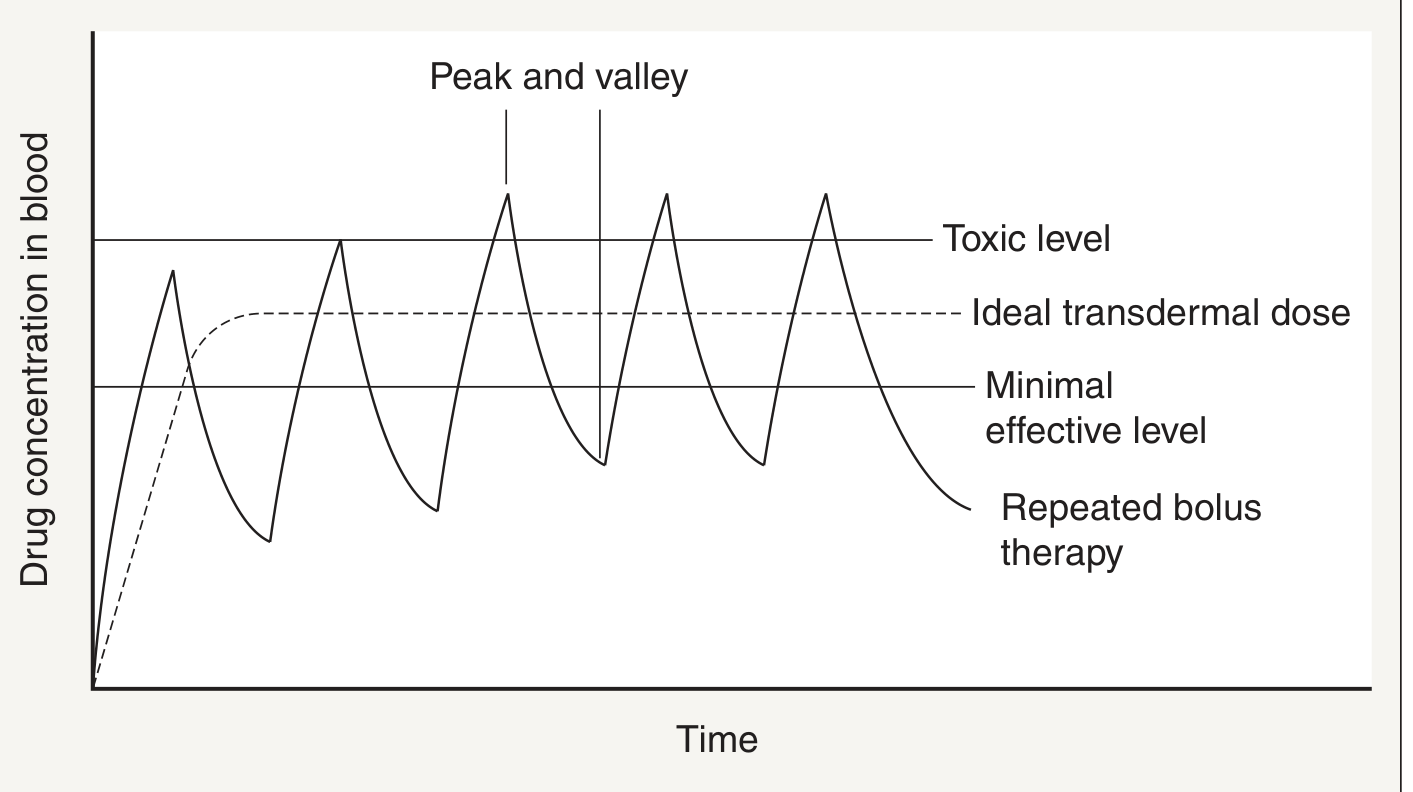
Compared to oral or injectable routes, transdermal delivery offers:
- Avoids first-pass hepatic metabolism — oral drugs are often heavily metabolised before reaching systemic circulation
- Avoids GI degradation and side effects
- Eliminates peak-and-valley fluctuations — maintains near-steady-state plasma levels (as shown above), reducing toxicity and sub-therapeutic troughs
- Reduced dosing frequency — long-acting patches can last days to weeks
- Improved patient adherence
- Avoids painful injections
- Targeted local therapy — for dermatological diseases, drug reaches the site directly
— Dermatology 2-Volume Set, 5e
Physicochemical Requirements for Transdermal Drugs
Not all drugs are suitable. FDA-approved transdermal drugs share these properties:
- Low molecular weight (<400 Da)
- Lipophilic (octanol-water partition coefficient up to 10,000)
- Low dose requirement (typically <10 mg/day)
Drugs Available as Transdermal Patches
Buprenorphine, clonidine, estradiol, fentanyl, granisetron, glyceryl trinitrate (nitroglycerin), levonorgestrel, lidocaine, methylphenidate, methyl salicylate, nicotine, norelgestromin, norethindrone acetate, oxybutynin, rivastigmine, rotigotine, scopolamine, selegiline, sumatriptan, testosterone
— Dermatology 2-Volume Set, 5e
Strategies to Enhance Transdermal Delivery
Fewer than two dozen drugs have been FDA-approved for transdermal administration due to the barrier's efficiency. Two broad categories of enhancement strategies exist:
1. Chemical Enhancement
| Agent | Mechanism |
|---|---|
| Water (occlusion) | Hydration swells corneocytes and distends intercellular spaces, creating "pores"; 24–48 hrs of occlusion needed |
| Solvents (ethanol, acetone, chloroform) | Extract barrier lipids and disrupt lamellar bilayers |
| Surfactants (e.g., sodium lauryl sulfate) | Extract lipids and expand lacunar domains |
| Penetration enhancers (azone, sulfoxides, fatty acids, urea) | Extract lipids AND alter lipid phase organization |
| Liposomes / nanoparticles | Carrier-mediated delivery through follicular route |
| Ionic liquids | Newer approach for enhanced drug solubilisation and delivery |
| Peptides | Cell-penetrating peptides acting as carrier vectors |
2. Physical Enhancement
| Method | Mechanism |
|---|---|
| Iontophoresis | Electrical current drives ionised drug through follicles/sweat ducts |
| Electroporation | High-voltage pulses create transient pores directly across corneocytes |
| Ultrasound (sonophoresis) | Cavitational/thermal effects disrupt lipid bilayers |
| Microneedles | Arrays of micron-scale needles physically breach the stratum corneum without reaching nerve endings |
| Mechanical abrasion / stripping | Remove outer layers of stratum corneum |
| Thermal ablation / fractional photothermolysis | Laser or heat creates microchannels through the stratum corneum |
| STAR particles | Star-shaped particles that physically disrupt the barrier |
— Dermatology 2-Volume Set, 5e
Conventional Patch Design
Transdermal patches consist of several layers:
- Backing layer — impermeable, protects drug from environment
- Drug reservoir or drug-in-matrix layer — controls release rate
- Rate-controlling membrane (in reservoir-type systems) — governs flux
- Adhesive layer — maintains skin contact
- Release liner — peeled off before application
Clinical Pharmacokinetic Considerations
- Onset of action is slower than oral or IV routes due to diffusion lag time through skin
- A depot effect exists in the stratum corneum; drug levels persist after patch removal
- Site selection matters: skin thickness, hair follicle density, hydration, and local blood flow vary by body region
- Example: Transdermal fentanyl avoids hepatic first-pass metabolism but has a slow onset and an inability to rapidly adjust dosing — important in analgesia management — Morgan & Mikhail's Clinical Anesthesiology, 7e
- Testosterone gels and patches provide more stable serum testosterone levels than IM injections of esters — Goodman & Gilman's Pharmacological Basis of Therapeutics
Limitations
- Only suitable for potent, low-dose, small, lipophilic molecules
- Local skin reactions (contact dermatitis, irritation)
- Slow onset — not suited for acute conditions
- Drug absorption varies with skin condition (age, disease, anatomical site)
- Risk of dose dumping if patch is damaged or exposed to heat
This is a shared conversation. Sign in to Orris to start your own chat.