Explain the mechanism of acute respiratory distress syndrome
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I have all the content needed for a comprehensive answer. Here is a structured explanation of the mechanism of ARDS.
Acute Respiratory Distress Syndrome (ARDS) — Mechanism
Definition and Core Concept
ARDS is a syndrome of noncardiogenic pulmonary edema arising from increased alveolar-capillary permeability, resulting in protein-rich exudate flooding the airspaces. Unlike cardiogenic pulmonary edema — driven by elevated left-sided cardiac pressures — ARDS edema is exudative, arising from barrier breakdown rather than hydrostatic forces. The physiological consequences are severe hypoxemia, decreased lung compliance, increased dead space ventilation, and pulmonary hypertension.
— Murray & Nadel's Textbook of Respiratory Medicine, 2-Volume
Triggers: Direct vs. Indirect Injury
ARDS arises from a heterogeneous set of conditions, classified by mechanism of injury:
| Direct (Pulmonary) Injury | Indirect (Nonpulmonary) Injury |
|---|---|
| Pneumonia (bacterial, viral, COVID-19) | Sepsis |
| Aspiration of gastric contents | Major trauma |
| Pulmonary contusion | Multiple blood transfusions |
| Toxic inhalation | Pancreatitis |
| Near-drowning | Cardiopulmonary bypass |
| Reperfusion injury (post-lung transplant) | Drug overdose |
The strongest evidence for causation exists for sepsis, trauma, multiple transfusions, aspiration, pulmonary contusion, pneumonia, and smoke inhalation. A history of chronic alcoholism (RR ~2.0) significantly predisposes individuals to developing ARDS.
The Central Event: Alveolar-Capillary Barrier Failure
Both components of the barrier are involved:
1. Alveolar Epithelium
Damage to the alveolar epithelium is considered the key precipitating event. Multiple mechanisms contribute to epithelial cell death: necrosis, apoptosis, coagulation, and mechanical stretch. Loss of the epithelial layer disrupts barrier integrity (enabling edema formation) and simultaneously prevents alveolar fluid clearance — a double hit.
2. Microvascular Endothelium
Loss of pulmonary vascular endothelial barrier integrity is both necessary and sufficient for ARDS development. Endothelial apoptosis and neutrophil-induced damage are the primary mechanisms of endothelial injury.
Neutrophils: Central Effectors of Injury
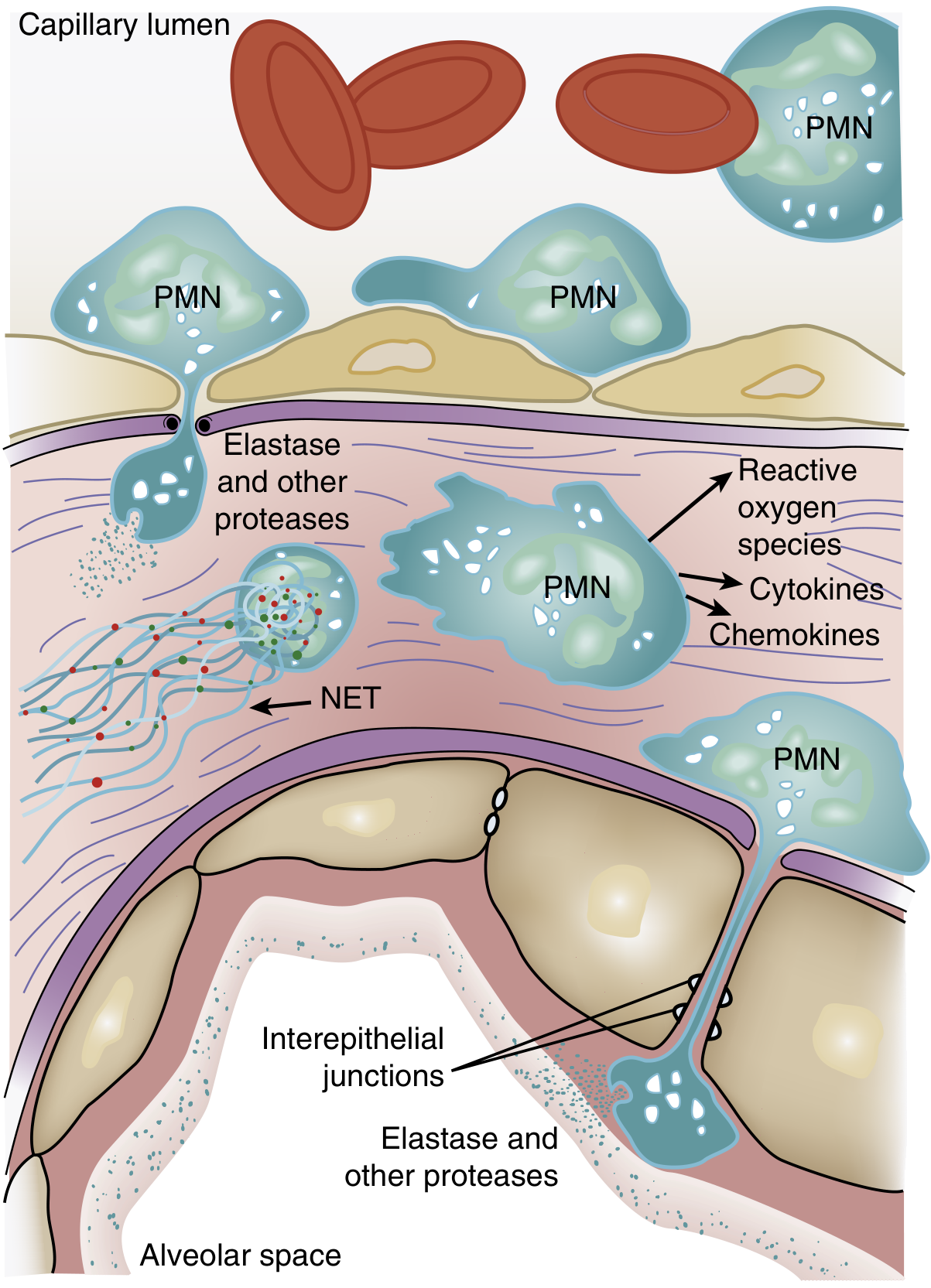
Figure 134.3 — Role of neutrophils in ARDS pathogenesis. Activated PMNs transmigrate across the alveolar-capillary membrane, releasing proteases, reactive oxygen species, cytokines, and neutrophil extracellular traps (NETs). — Murray & Nadel's Textbook of Respiratory Medicine
The hallmark histologic finding of ARDS is accumulation of neutrophils in the pulmonary microvasculature. The sequence is:
- Transient leukopenia — one of the earliest signs, even before hypoxemia, due to sequestration of neutrophils in pulmonary capillaries (neutrophils are larger than capillary diameter; activated neutrophils become "stiff" via actin cytoskeleton changes and cannot deform to pass through).
- Sequestration and activation — once trapped, activated neutrophils induce endothelial barrier breakdown, facilitating their own migration into the interstitium and alveolar space.
- Release of cytotoxic mediators: reactive oxygen species (ROS), proteolytic enzymes (leukocyte elastase), cationic peptides (defensins), eicosanoids, TNF-α, IL-1β, and neutrophil extracellular traps (NETs).
- Elastase degrades surfactant protein A in vitro, compounding the surfactant dysfunction described below.
Neutrophil–platelet interactions have also been identified as a mechanism of mutual activation contributing to lung injury.
Surfactant Dysfunction
ARDS is characterized by:
- Depletion of critical surfactant phospholipids (dipalmitoylphosphatidylcholine and phosphatidylglycerol)
- A shift from large (active) to small (inactive) surfactant aggregates, due to decreased production and increased conversion
- Degradation of surfactant proteins by leaked plasma proteins and neutrophil elastase
The result is alveolar collapse (atelectasis), further reducing functional residual capacity (FRC) and worsening V/Q mismatch.
Gas Exchange Failure
ARDS produces hypoxemia through two mechanisms:
- Right-to-left shunt — flooded and collapsed alveoli are perfused but not ventilated
- Low V/Q regions — partially flooded alveoli with reduced ventilation relative to perfusion
Together, these increase dead space/tidal volume ratios, impair CO₂ elimination (requiring increased minute ventilation), and produce the refractory hypoxemia characteristic of ARDS.
Impaired Alveolar Fluid Clearance
Under normal conditions, apical Na⁺ channels (ENaC) on alveolar epithelial cells mediate Na⁺ uptake, drawing water out of the alveolar space via osmotic gradient; basolateral Na⁺/K⁺-ATPases maintain the intracellular Na⁺ gradient. In ARDS:
- Hypoxia downregulates ENaC expression and suppresses Na⁺/K⁺-ATPase activity
- Loss of epithelium eliminates this clearance mechanism entirely
- The result is persistent alveolar flooding
Angiopoietin Signaling
Angiopoietin-2 (Ang2), released by endothelial cells, promotes vascular leak by acting as a competitive antagonist to the barrier-stabilizing Angiopoietin-1 (Ang1). Elevated Ang2 levels are found in patients with sepsis-associated ARDS. Exogenous Ang2 in animal models causes pulmonary vascular leak reversible by Ang1, supporting a role for this signaling axis in mediating endothelial barrier loss.
Cytokine Network and NF-κB Signaling
Downstream of the initial insult:
- p38 MAPK is activated by LPS and other PAMPs, stimulating TNF-α production and macrophage inflammatory protein-2 (a macrophage chemoattractant)
- TNF-α and IL-1β amplify the inflammatory cascade, recruiting more neutrophils and perpetuating alveolar damage
- NF-κB is a central transcription factor coordinating the pro-inflammatory response; heat shock protein 70 (HSP70) exerts a protective effect partly by suppressing NF-κB activity
Pathologic Phases
The pathology is termed Diffuse Alveolar Damage (DAD) and evolves through three overlapping stages:
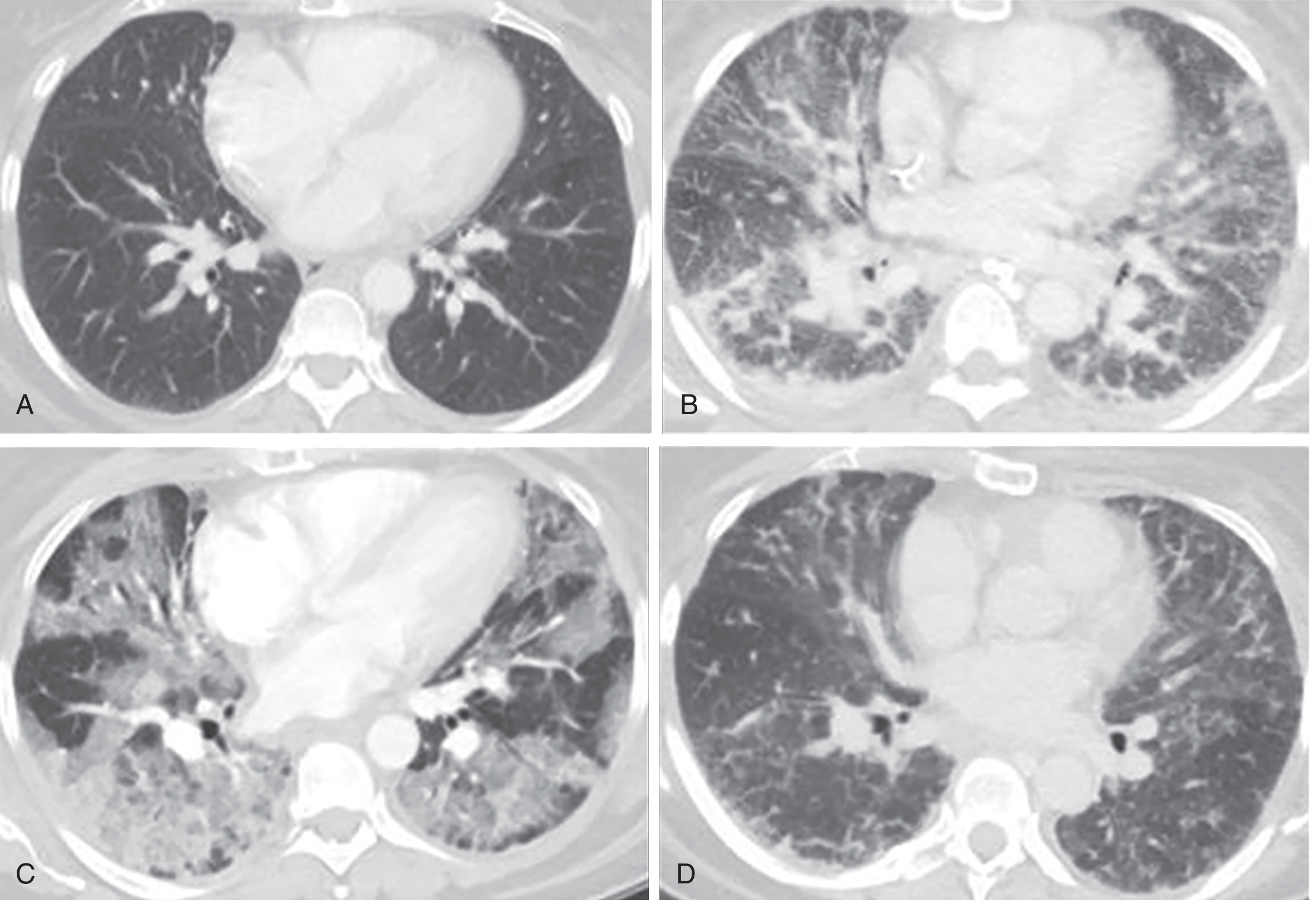
Figure 134.2 — CT demonstration of ARDS phases. B: Exudative phase; C: Proliferative phase; D: Fibrotic phase. — Murray & Nadel's Textbook of Respiratory Medicine
| Phase | Timing | Pathology |
|---|---|---|
| Exudative | Days 1–7 | Hyaline membranes (cellular debris + proteins + surfactant), protein-rich alveolar fluid, neutrophilic infiltrate, widespread epithelial disruption |
| Proliferative | Days 7–21 | Reorganization of hyaline membranes, early fibrosis, obliteration of pulmonary capillaries, decreased neutrophils |
| Fibrotic | >2 weeks (subset) | Pulmonary fibrosis; fibroproliferation may begin as early as 24 hours after onset (elevated N-terminal procollagen III in BAL fluid) |
Importantly, only ~50% of ARDS patients show DAD on biopsy or autopsy. Those with confirmed DAD are younger, more severely ill, have worse compliance and oxygenation, and are five times more likely to die of hypoxemic respiratory failure.
Pulmonary Hypertension in ARDS
Multiple mechanisms contribute:
- Hypoxic pulmonary vasoconstriction
- Intravascular fibrin deposition
- Compression of blood vessels by positive-pressure ventilation
Summary Pathway
Predisposing insult (sepsis, pneumonia, aspiration, trauma...)
↓
Systemic/local inflammatory activation
↓
Neutrophil sequestration in pulmonary capillaries
↓
Transmigration → ROS, elastase, cytokines, NETs
↓
Alveolar-capillary barrier breakdown
(endothelial + epithelial injury)
↓
┌─────────────────┬──────────────────────┐
│ Protein-rich │ Surfactant dysfunction│
│ alveolar edema │ + impaired fluid │
│ (exudate) │ clearance (↓ENaC) │
└────────┬────────┴──────────────────────┘
↓
Alveolar collapse + flooding
↓
Shunt + low V/Q → Refractory hypoxemia
↑ Dead space → ↑ Minute ventilation requirement
↓ Compliance → ↑ Work of breathing
↓
DAD (Diffuse Alveolar Damage)
→ Exudative → Proliferative → Fibrotic phases
Sources: Murray & Nadel's Textbook of Respiratory Medicine, 2-Volume; Goldman-Cecil Medicine International Edition, 2-Volume Set
This is a shared conversation. Sign in to Orris to start your own chat.