Cochlear implants in detail
Reading File
Finding Sources
Searching Images
cochlear implant anatomy diagram
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Now I have comprehensive material to provide a detailed response. Let me synthesize everything.
Cochlear Implants — Comprehensive Overview
1. Definition and Principle
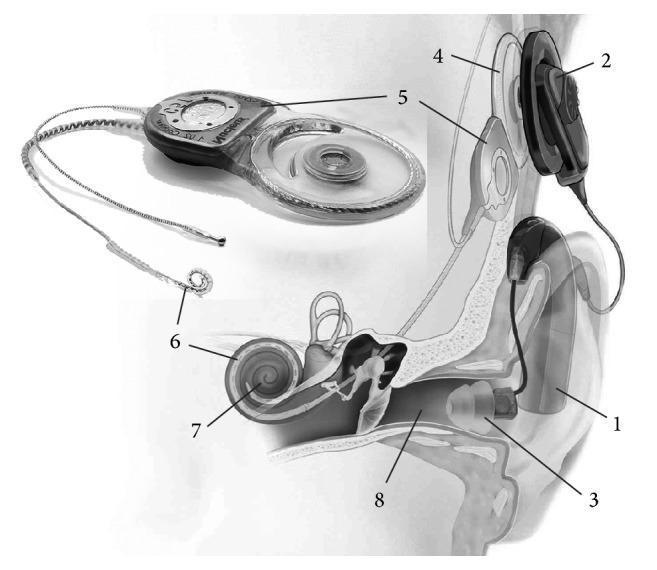
A cochlear implant (CI) is an electronic device that bypasses damaged hair cells in the cochlea and directly stimulates the spiral ganglion cells (and auditory nerve) with electrical impulses. Unlike a hearing aid, which amplifies sound, a CI converts sound into electrical signals that are delivered directly to the auditory nerve, bypassing the non-functional sensory epithelium.
2. Components
External Components
| Component | Function |
|---|---|
| Microphone | Placed near the ear hook; captures acoustic sound and converts it to an analogue electrical signal |
| Sound processor | Amplifies, compresses, and filters the signal; digitises it via Fourier analysis to encode timing, frequency, and intensity |
| RF transmitter antenna | Sends the processed signal transcutaneously to the internal receiver |
| External magnet | Aligns with the internal magnet for transcutaneous RF signal transmission |
| Battery | Powers the system; rechargeable or disposable 675 hearing-aid batteries depending on the manufacturer |
Internal Components
| Component | Function |
|---|---|
| Internal RF antenna | Receives the transcutaneous signal |
| Internal magnet | Maintains alignment with the external coil |
| Receiver/Stimulator | Translates the electrical signal into rapid electrical impulses distributed to individual wires within the electrode array |
| Electrode array | Inserted into the scala tympani; stimulates spiral ganglion cells at multiple frequency-coded locations |
— K J Lee's Essential Otolaryngology, p. 406
3. How It Works (Signal Processing)
- Sound is picked up by the microphone.
- The sound processor performs frequency analysis (Fourier), compression, and filtering.
- The digitised signal is transmitted via radiofrequency (RF) across the skin to the internal receiver.
- The receiver/stimulator decodes the signal and delivers electrical pulses to specific electrodes along the array.
- Electrodes are positioned tonotopically in the scala tympani — basal electrodes stimulate high-frequency fibres; apical electrodes stimulate low-frequency fibres.
- The auditory nerve is activated, and signals travel to the brainstem and auditory cortex.
4. Electrode Array Design
Three major manufacturers provide different arrays:
Cochlear (Nucleus)
- Precurved perimodiolar arrays (Contour Advance) and straight lateral-wall arrays
- Nucleus Hybrid: shortened 19.5 mm array for combined electric-acoustic stimulation (EAS)
Advanced Bionics (HiFocus)
- 1j electrode: banana-shaped with flat contacts oriented toward the modiolus; raised partitions between contacts reduce electrode interactions
- Helix electrode: same flat-contact configuration, precurved for perimodiolar positioning
- Mid-Scale electrode: shorter length but achieves 420° insertion depth due to precurved configuration
MED-EL (SYNCHRONY)
- Titanium-housed receiver/stimulator, MR Conditional at 1.5 T
- Standard 31.5 mm array with contacts every 2.4 mm
- FLEX electrode family (FLEX23 to FLEXSOFT) — 20–31.5 mm options
- Custom common-cavity electrode for severe malformations
- Compressed array (15 mm) for ossified cochleas
Electrode design philosophy:
- Perimodiolar arrays: closer to the modiolus → lower stimulation thresholds, reduced current spread, better frequency specificity
- Straight/lateral-wall arrays: less traumatic insertion, better hearing conservation for EAS candidates
— Cummings Otolaryngology, p. 3077–3078
5. Candidacy
Adults
- Bilateral moderate-to-profound SNHL
- Aided sentence scores ≤50% in the ear to be implanted and ≤60% in the best-aided condition
- CMS (Medicare/Medicaid) uses stricter criterion: ≤40%
- FDA guidelines per device — generally considered conservative relative to current evidence
Children
- 12–24 months: profound SNHL (>90 dB HL) with limited hearing-aid benefit (MAIS score criteria)
- >24 months: severe-to-profound SNHL (>70 dB HL); aided open-set word recognition ≤30% (MLNT or LNT)
- Minimum 3-month hearing-aid trial (waived for meningitis-related ossification)
- Earlier implantation (<12 months) is increasingly common off-label — studies show language outcomes comparable to normal-hearing children
Cochlear Nucleus Hybrid System (EAS Candidacy)
- Low-frequency thresholds ≤60 dB HL at 500 Hz
- PTA (2, 3, 4 kHz) ≥75 dB HL in ear to be implanted
- Aided CNC word score 10–60% in the implant ear; contralateral ear ≤80%
— K J Lee's Essential Otolaryngology, p. 406–410
Special Populations
| Condition | Consideration |
|---|---|
| Auditory Neuropathy Spectrum Disorder (ANSD) | Normal OAEs but absent ABR; reliable open-set speech recognition can be achieved with CI |
| Single-sided deafness (SSD) | Reduces tinnitus and improves sound localisation (currently off-label in the US) |
| NF2 / bilateral vestibular schwannomas | CI attempted when cochlear nerve intact; outcomes generally superior to auditory brainstem implant (ABI) |
| Meningitis with ossification | Expedited implantation; hearing-aid trial waived |
Contraindications
- Complete labyrinthine aplasia
- Cochlear aplasia
- Complete cochlear ossification
- Absent cochlear nerve
- Retrocochlear pathology along the auditory pathway (relative contraindication)
6. Preoperative Evaluation
Imaging
- MRI (preferred): confirms cochlear nerve presence, identifies labyrinthine malformations
- HRCT of temporal bone: evaluates bony anatomy, cochlear malformations, narrow IAC (<3 mm); assesses surgical anatomy for:
- Poorly pneumatised mastoid / facial recess
- Anterior sigmoid sinus
- High-riding jugular bulb
- Anomalous facial nerve
- Risk of CSF gusher (thin cribriform area, widened IAC, enlarged vestibular aqueduct)
Audiometric assessment
- Children: OAE, ABR, behavioural audiometry, parental questionnaires (age-dependent)
- Adults: AzBio sentences, CNC monosyllabic words, BKB-SIN test
Jervell and Lange-Nielsen Syndrome (JLNS): must be excluded — prolonged QT + deafness → risk of perioperative sudden death. ECG mandatory in appropriate candidates.
— Cummings Otolaryngology, p. 3086
7. Surgical Procedure
The standard surgical approach:
- Postauricular incision and creation of a tight periosteal pocket for the receiver/stimulator
- Mastoidectomy — drilling of the mastoid cortex to expose the antrum and posterior tympanum
- Posterior tympanotomy / facial recess approach — opening between the facial nerve and chorda tympani to access the middle ear
- Cochleostomy or round window membrane access — entry into the scala tympani
- Electrode array insertion — gentle, atraumatic ("soft surgery") insertion to preserve residual hearing
Hearing conservation ("soft surgery") principles:
- Avoid direct suctioning of perilymph from the scala tympani
- Use non-ototoxic topical antibiotics, corticosteroids, and hyaluronate lubricant
- Immediately seal cochlea with fascia after insertion
- Use round window approach where possible
Special paediatric surgical considerations:
| Issue | Modification |
|---|---|
| Small head / parietal cortex <1.5 mm | Tight periosteal pocket; no seat/tie-down holes |
| Mastoid bone marrow | Diamond burr to reduce haemorrhage |
| Absent mastoid tip | Inferior flap incision |
| Small facial recess | Extend facial recess as needed |
| Anomalous facial nerve | Monitor all cases |
| Dysplasia | Prepare for CSF gusher, fistula, anomalous facial nerve |
— Cummings Otolaryngology, p. 3091–3092
8. Bilateral Cochlear Implantation
Why bilateral?
- More spiral ganglion cell populations stimulated — ensures the better ear is implanted
- Reduces risk of auditory deprivation from single-device failure
- Confers binaural hearing advantages:
- Head shadow effect: preferential use of the ear with better signal-to-noise ratio
- Binaural summation: central processing draws more information from two ears than one
- Squelch effect ("cocktail party effect"): focus on one talker among competing noise
- Improved sound localisation: brain uses interaural time and level differences
Timing:
- Simultaneous bilateral implantation is preferred over sequential implantation with >12-month inter-implant delay
- Sequential implantation with <12-month delay achieves comparable outcomes
- Critical "window of opportunity": birth to 2 years, when neural plasticity and language receptivity are at their peak
- If this window is missed, cross-modal plasticity occurs — auditory cortex is reorganised for vision or somatosensation
— K J Lee's Essential Otolaryngology, p. 412–413
9. Electric-Acoustic Stimulation (EAS / Hybrid)
For candidates with ski-slope hearing loss (severe-to-profound high-frequency SNHL but preserved low-frequency hearing):
- Short electrode array (10–20 mm) inserted atraumatically to stimulate the basal (high-frequency) cochlea
- Acoustic component via hearing aid earmould provides low-frequency amplification
- Outcomes in EAS condition exceed either electric or acoustic stimulation alone
- Shorter arrays → better hearing conservation; longer arrays → more effective electrical stimulation — a trade-off managed per patient
10. The CI Team
The multidisciplinary team typically includes:
- Core: CI surgeon, audiologist, speech-language pathologist
- Extended: psychologist, educational specialist, social worker
- Community: early interventionists, deaf/hard-of-hearing itinerant teachers, educational audiologist
- In complex cases: physical/occupational therapists, developmental paediatricians, behavioural interventionists
— Cummings Otolaryngology, p. 3721
11. Outcomes and Rehabilitation
- Adults (including those aged 65–80) achieve significant improvements in speech recognition pre- to post-implantation across varied stimulus presentation levels
- Auditory training (rehabilitation) is an important component of post-CI care — especially in children where habilitation directly drives language development
- Spiral ganglion cell (SGC) survival directly correlates with word recognition scores — supporting the case for earliest possible implantation
12. Complications
Surgical complications (~8.6% overall in prospective multi-centre US data):
- Wound dehiscence / infection (most common)
- Otitis media
- Incomplete electrode insertion
- Facial nerve injury (rare but monitored intraoperatively)
- CSF gusher (labyrinthine dysplasia)
- Subdural haematoma (rare)
Device failure
- Hard failure: device no longer meets manufacturer specifications; sudden significant change in auditory perception → requires revision
- Soft failure: performance decline and adverse symptoms with normal device imaging → heavily dependent on clinical judgement
Revision rates:
| Study | Paediatric | Adult |
|---|---|---|
| Brown et al. | 7.3% | 3.8% |
| Cullen et al. | 11.2% | — |
| Gosepath et al. | 15.4% | 9.3% |
Higher paediatric rates attributable to falls (learning to walk) and wire fatigue from movement.
Meningitis:
- Rare but serious — predominantly Streptococcus pneumoniae
- Mandates universal antipneumococcal vaccination
— Cummings Otolaryngology, p. 3283
13. Vaccination (Meningitis Prophylaxis)
Children
- PCV13 at least 2 weeks before implantation
- PPSV23 at least 8 weeks after completing PCV13 series (after age 2)
- Children 6–18 years: single dose PCV13 regardless of prior history
Adults (>19 years)
- Single dose PCV13 followed by PPSV23 at least 8 weeks later
- Second PPSV23 dose recommended if >65 years
- If previously received PPSV23: give PCV13 at least 1 year later; then PPSV23 no sooner than 8 weeks after PCV13
AOM management in CI patients
- Higher meningitis risk after AOM
- No observation period allowed — treat promptly
- AOM <2 months post-surgery: parenteral antibiotics
- AOM >2 months post-surgery: high-dose amoxicillin-clavulanate
- Lumbar puncture with CSF cultures if meningitis suspected
— K J Lee's Essential Otolaryngology, p. 444
14. MRI Compatibility
All current FDA-approved devices have MR Conditional ratings. Most are rated at 1.5 T with the internal magnet in place (e.g., MED-EL SYNCHRONY). Older devices required magnet removal before scanning. This is an important consideration for patients who may require future MRI.
Key sources: K J Lee's Essential Otolaryngology (9781260122237); Cummings Otolaryngology Head and Neck Surgery (9780323612173)
This is a shared conversation. Sign in to Orris to start your own chat.