lupus nephritis
Reading File
Finding Sources
Finding Sources
Searching Images
lupus nephritis histology WHO class
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive information to give a thorough answer. Here is a structured overview of lupus nephritis:
Lupus Nephritis
Overview
Lupus nephritis (LN) is kidney inflammation caused by systemic lupus erythematosus (SLE), representing the most serious organ manifestation of the disease. Clinically significant kidney disease occurs in approximately 30% of SLE patients. It is most common in Black females aged 15–45, though it disproportionately affects all non-White populations and is the leading cause of secondary glomerular disease in East Asian countries (>50% of secondary glomerulonephropathies in China, Korea, Japan).
— Henry's Clinical Diagnosis and Management by Laboratory Methods, p. 1261
Pathogenesis
The pathogenesis is multi-factorial:
- Genetic and epigenetic predisposition — >50 polymorphisms associated with LN susceptibility, including variants in CCR6 (Th17 pathway)
- Autoantibodies — ANA are nearly universal; anti-dsDNA (most specific), anti-Sm, anti-histone, anti-C1q, and antiphospholipid antibodies are common
- Immune complex deposition — Glomerular deposits activate complement (classical pathway via C1q), causing inflammation
- B- and T-lymphocyte dysregulation, macrophages, dendritic cells, and neutrophil extracellular traps (NETs) all contribute
- "Full house" immunofluorescence — simultaneous staining of IgG, IgM, IgA, C3, and C1q is pathognomonic
— Henry's Clinical Diagnosis and Management by Laboratory Methods, p. 1265
Clinical Presentation
| Feature | Details |
|---|---|
| Hematuria | Usually microscopic; red cell casts indicate active glomerulonephritis |
| Proteinuria | Ranges from mild to nephrotic-range (>3.5 g/day) |
| Nephrotic syndrome | Particularly in Class V |
| Renal insufficiency / AKI | In active proliferative disease |
| Hypertension | Common |
| Tubular defects | Tubular acidosis, Fanconi-type in severe cases |
Classification (ISN/RPS 2003, revised 2018)
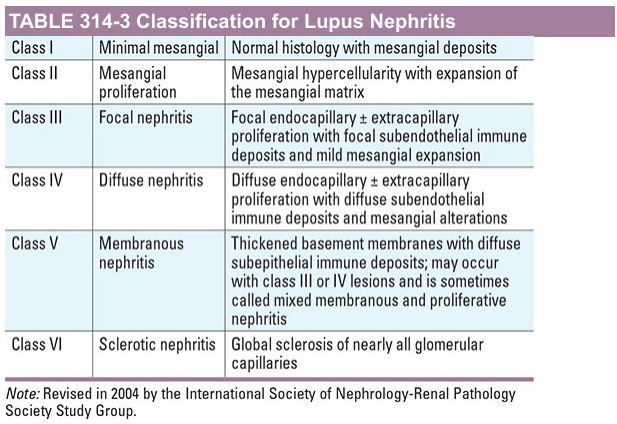
— Harrison's Principles of Internal Medicine, 21st Edition
Histologic detail by class:
- Class I (Minimal mesangial): Normal light microscopy; mesangial deposits on IF and EM only. No clinical renal disease.
- Class II (Mesangial proliferative): Mesangial hypercellularity and matrix expansion; mesangial deposits on IF/EM. Mild proteinuria/hematuria.
- Class III (Focal): Endocapillary hypercellularity in <50% of glomeruli, often segmental, ± necrosis and crescents. Focal subendothelial deposits.
- Class IV (Diffuse): Endocapillary hypercellularity in ≥50% of glomeruli. Key features:
- Wire loops — eosinophilic thickening of peripheral capillary loops (massive subendothelial deposits)
- "Hyaline thrombi" — intraluminal immune deposits (misnomer)
- Karyorrhectic nuclear debris
- Massive mesangial + subendothelial deposits on EM
- The most severe and clinically aggressive class
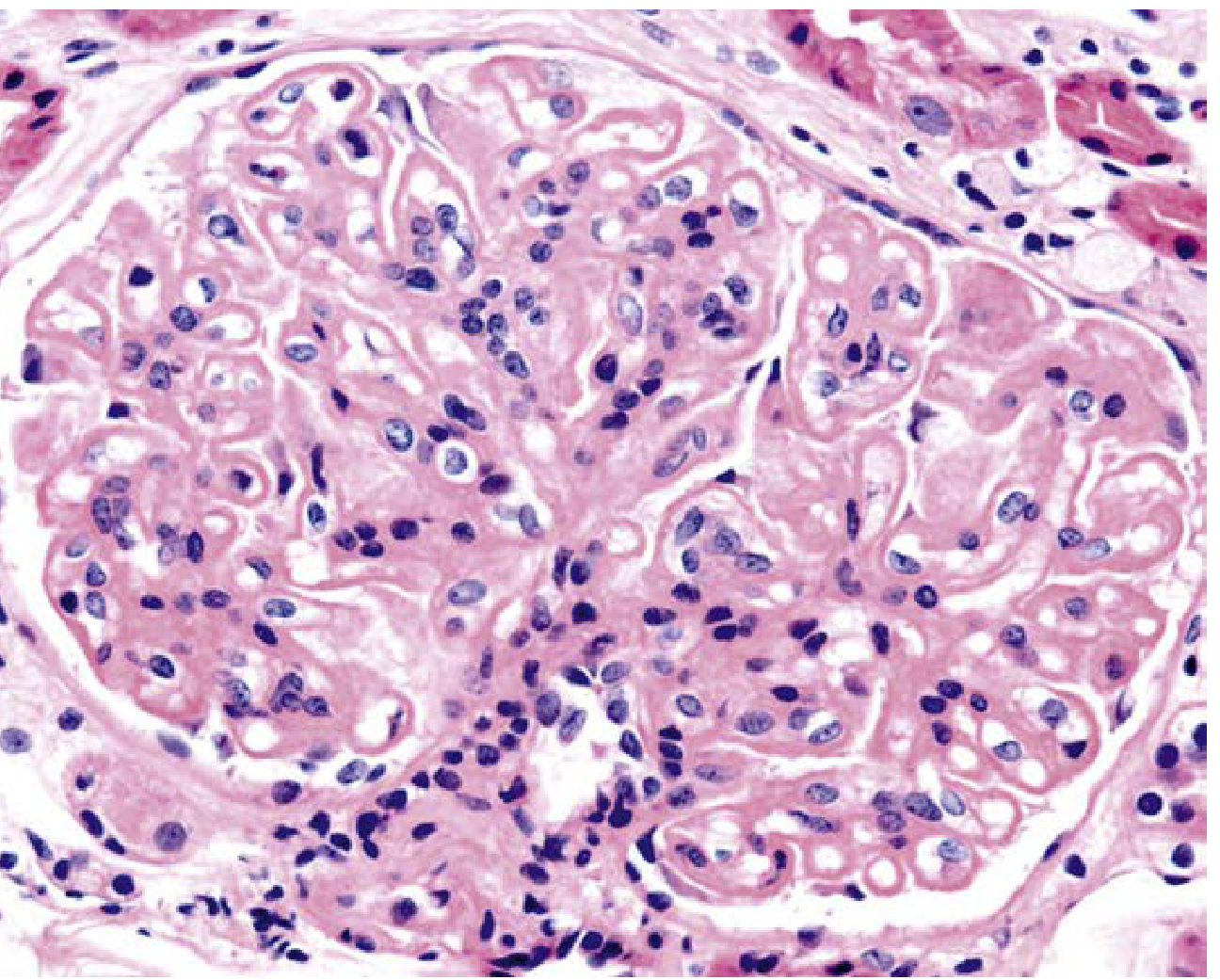
- Class V (Membranous): Thickened GBM with subepithelial deposits; GBM "spikes" on silver stain; may coexist with Class III or IV ("mixed membranous-proliferative"). Presents as nephrotic syndrome.
- Class VI (Advanced sclerosing): >90% global glomerulosclerosis, no residual activity. Represents burnt-out disease.
Class III/IV subclassification:
- (A) — Active lesions (proliferative)
- (A/C) — Active + chronic
- (C) — Chronic (sclerosing)
— Henry's Clinical Diagnosis and Management by Laboratory Methods, pp. 1261–1265
Class V histology (H&E, ×400) — spherical capillary wall thickening with mild mesangial hypercellularity:
Diagnosis
- Renal biopsy is the gold standard; required for accurate classification and treatment guidance
- Serologic markers: anti-dsDNA titres, complement (low C3/C4), ANA
- Urinalysis: active sediment (RBC casts, dysmorphic red cells), proteinuria
- SLICC criteria allow stand-alone renal criterion: biopsy-proven LN + ANA or anti-dsDNA
- Extraglomerular lesions (tubulointerstitial, vascular) add prognostic value beyond glomerular classification
Management
Induction Therapy (active proliferative LN — Class III/IV ± V)
| Agent | Regimen |
|---|---|
| High-dose corticosteroids | Oral prednisolone ± IV methylprednisolone pulses (500–1000 mg × 3) |
| Mycophenolate mofetil (MMF) | 2–3 g/day; preferred in many guidelines for non-Asian populations |
| IV Cyclophosphamide (IVC) | Low-dose Euro-Lupus regimen (500 mg × 6 fortnightly) or high-dose NIH |
| Voclosporin + MMF + belimumab | Newer triple therapy with improved remission rates |
Shift from cyclophosphamide-dominant to MMF-dominant regimens has improved outcomes and reduced adverse effects.
Maintenance Therapy
- Azathioprine (1.5–2.5 mg/kg/day) or MMF (1–2 g/day)
- Hydroxychloroquine — given to all LN patients; reduces flare risk and improves long-term outcomes
- Continued for minimum 3–5 years after remission; many require indefinite treatment
Class V (Membranous LN)
- Low-level proteinuria: RAS blockade + BP control + hydroxychloroquine ± immunosuppression guided by extrarenal SLE
- Nephrotic-range proteinuria: Glucocorticoids + MMF or cyclophosphamide or calcineurin inhibitor or rituximab + hydroxychloroquine
Additional measures
- RAS blockade (ACEi/ARB) — antiproteinuric and renoprotective in all classes
- Thromboprophylaxis — antiphospholipid syndrome is common; LMWH/warfarin if APS present
- Statins — dyslipidemia management
- Rituximab — for refractory disease; not superior to standard therapy in initial trials but used in treatment-resistant cases
Plasma Exchange
Not recommended as routine adjunct — the Lupus Nephritis Collaborative Study showed no improvement in outcomes despite faster antibody clearance. May be considered in select cases with severe thrombotic microangiopathy.
— Brenner and Rector's The Kidney, 2-Volume Set; Comprehensive Clinical Nephrology, 7th Ed
Monitoring and Response
- Complete renal response: proteinuria <0.5 g/24h, stable/improved creatinine, inactive urine sediment
- Repeat biopsy may be warranted at transformation (change in clinical picture) or suspected flare
- Anti-dsDNA titres and complement levels correlate with disease activity
Pregnancy
- MMF is teratogenic — must be switched to azathioprine ≥6 weeks before conception
- Hydroxychloroquine should be maintained throughout pregnancy
- Cyclophosphamide and rituximab — contraindicated
- Tacrolimus — increasingly used as alternative
- Predictors of poor outcome: creatinine >0.9 mg/dL, proteinuria >0.5 g/24h, antiphospholipid syndrome, hypertension, active disease at conception
- Low-dose aspirin for preeclampsia prophylaxis; preeclampsia and LN flare can be clinically indistinguishable
— National Kidney Foundation Primer on Kidney Diseases, 8e; Comprehensive Clinical Nephrology, 7th Ed
Prognosis
- Class IV carries the worst renal prognosis without treatment
- High baseline creatinine, significant chronic damage on biopsy, crescents, and vascular lesions portend inferior outcomes
- Anti-Sm positivity and older age at presentation (>50 years) associated with worse prognosis in Korean cohorts
- With modern immunosuppression, 10-year kidney survival has improved substantially, though ESKD remains a significant risk in severe/refractory disease
2026 TB guidelines
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I have enough to give a complete, honest, and well-sourced answer.
An Important Clarification First
There are no published "2026 TB guidelines" as of my knowledge cutoff. The most recent TB guideline updates are from 2022–2024, primarily from the WHO. No major guideline body (WHO, ATS/CDC/IDSA) has published a 2026 iteration. The most up-to-date guidance available — including what Harrison's 22E (2025) covers — reflects these recent updates.
Here is a comprehensive summary of the current TB guidelines as of 2024–2025:
Current TB Treatment Guidelines (2024–2025)
Primary sources: Harrison's Principles of Internal Medicine, 22nd Edition (2025); Murray & Nadel's Textbook of Respiratory Medicine
Drug-Susceptible TB (DS-TB)
Standard 6-Month Regimen (Adults & Children ≥12 yrs)
The regimen of choice for virtually all forms of drug-susceptible TB:
| Phase | Duration | Drugs |
|---|---|---|
| Intensive (initial) | 2 months | HRZE (isoniazid + rifampin + pyrazinamide + ethambutol) |
| Continuation | 4 months | HR (isoniazid + rifampin) |
- Cures >90% of patients
- Daily administration throughout is strongly preferred; intermittent regimens are associated with higher treatment failure, relapse, and resistance
- ATS/CDC/IDSA allow thrice-weekly supervised (DOT) regimens only in HIV-negative, non-cavitary, low-relapse-risk patients
4-Month Regimen (TBESC2/Study 31 — newer option for adults ≥12 yrs)
| Phase | Duration | Drugs |
|---|---|---|
| Intensive | 2 months | HPMz (isoniazid + rifapentine + moxifloxacin + pyrazinamide) |
| Continuation | 2 months | HPM (isoniazid + rifapentine + moxifloxacin) |
WHO now recommends this 4-month regimen for new drug-susceptible pulmonary TB in patients ≥12 years. This represents a significant evolution in shortening treatment duration.
Children (3 months – 16 years), Non-Severe TB
| Phase | Duration | Drugs |
|---|---|---|
| Intensive | 2 months | HRZ(E) |
| Continuation | 2 months | HR |
Ethambutol is generally omitted in children without suspected isoniazid resistance or HIV.
Extrapulmonary TB
- Most forms: treat with the standard 6-month pulmonary regimen
- TB meningitis: extend continuation phase to 7–10 months (ATS/CDC/IDSA) or 10-month consolidation (WHO/AAP)
- Bone/joint TB, miliary TB in children: up to 12 months total
Latent TB Infection (LTBI) — Preferred Regimens
Shorter rifamycin-based regimens are now preferred over 9-month isoniazid monotherapy:
| Regimen | Duration | Frequency | Notes |
|---|---|---|---|
| INH + Rifapentine (3HP) | 3 months | Weekly | Preferred; DOT or self-administered |
| INH + Rifapentine (1HP) | 1 month | Daily | HIV-infected ≥13 yrs; limited data |
| Rifampin (4R) | 4 months | Daily | Good alternative |
| Isoniazid (9H) | 9 months | Daily | Still acceptable; preferred in pregnancy (high-risk) |
| Isoniazid (6H) | 6 months | Daily | Not for HIV+ or fibrotic lesions |
— Murray & Nadel's Textbook of Respiratory Medicine, p. 938
Drug-Resistant TB — The Major 2022–2024 Updates
This is where the most significant recent guideline changes have occurred, driven by the Nix-TB, Ze-Nix, TB-PRACTECAL, BEAT-TB, and endTB trials.
MDR-TB / RR-TB: Four WHO-Recommended Approaches (2024 Update)
1. BPaLM — 6-Month, Fully Oral (Preferred for most MDR/RR-TB)
Bedaquiline + Pretomanid + Linezolid 600 mg + Moxifloxacin
- Treatment success ~90%
- For: MDR/RR-TB or pre-XDR-TB, age ≥14, regardless of HIV status
- Excludes: disseminated TB, CNS TB, osteoarticular TB
- Excludes: prior exposure ≥1 month to bedaquiline, pretomanid, or linezolid
- Fluoroquinolone DST guides whether moxifloxacin is retained or dropped (→ BPaL if FQ-resistant)
- Contraindicated in pregnancy (pretomanid safety data insufficient)
2. BDLLfxCfz — 6-Month, All-Oral
Bedaquiline + Delamanid + Linezolid 600 mg + Levofloxacin + Clofazimine
- Option for those without prior exposure to bedaquiline, delamanid, and linezolid
- Either levofloxacin or clofazimine may be omitted based on DST
3. Shorter 9-Month Oral Bedaquiline-Containing Regimens
Options include:
- BLMZ (bedaquiline + linezolid + moxifloxacin + pyrazinamide)
- BLLfCfZ (bedaquiline + linezolid + levofloxacin + clofazimine + pyrazinamide)
- BDLLfxZ
- For patients with no prior exposure to B, D, L, and in whom fluoroquinolone resistance is excluded
4. Individualized Longer Regimen (≥18 months)
- Optimal combination of oral drugs using WHO priority grouping
- For complex drug resistance patterns, prior treatment exposure, or contraindications to shorter regimens
Pre-XDR-TB and XDR-TB
- BPaL (without moxifloxacin) is the backbone
- Individualized regimens based on DST and exposure history
Isoniazid-Resistant TB (Hr-TB)
BPaLM or RZELfx (rifampin + pyrazinamide + ethambutol + levofloxacin) for 6 months
— Harrison's Principles of Internal Medicine, 22nd Edition (2025), pp. 1441–1443
Special Populations
| Situation | Recommendation |
|---|---|
| Pregnancy | 2HRZE / 7HR (9 months total); pyrazinamide not routine in US; streptomycin contraindicated; avoid bedaquiline, delamanid, thioamides in MDR-TB |
| Chronic renal failure | Avoid aminoglycosides; use ethambutol only with serum level monitoring; standard HRZ doses for mild-moderate CKD; reduce pyrazinamide dose in severe CKD |
| Hepatic disease | Avoid pyrazinamide in liver failure; use ethambutol + streptomycin ± fluoroquinolone if severe; monitor closely if using HR |
| HIV co-infection | Treat TB first (or simultaneously if very immunosuppressed); manage drug interactions especially between rifamycins and ART |
| Silicotuberculosis | Extend therapy by ≥2 months |
| Cavitary TB + positive culture at 2 months | Retest for drug resistance; consider regimen change; complete full 6 months without interruptions >4 weeks |
Prevention
- BCG vaccine: Given at birth in high-burden countries; 80% efficacy against severe childhood TB (meningitis, miliary); variable efficacy against adult pulmonary TB
- Infection control: Rapid diagnosis, isolation, and effective treatment render patients non-infectious within 2–4 weeks
- LTBI treatment in high-risk contacts (recent exposure, immunosuppressed, young children, HIV+)
Key Drug Abbreviations
| Symbol | Drug |
|---|---|
| H | Isoniazid |
| R | Rifampin |
| Z | Pyrazinamide |
| E | Ethambutol |
| P | Rifapentine |
| M | Moxifloxacin |
| B | Bedaquiline |
| Pa | Pretomanid |
| L | Linezolid |
| Lf/Lfx | Levofloxacin |
| D | Delamanid |
| Cf/Cfz | Clofazimine |
Bottom line: No "2026 guidelines" exist yet. The most current guidance reflects the WHO 2022 BPaLM recommendation for MDR-TB and the 2024 WHO update formalising four MDR/RR-TB treatment approaches, along with the 4-month HPMz/HPM regimen for drug-susceptible TB — all incorporated into Harrison's 22E (2025). If you are looking for a specific guideline body (WHO, ATS/CDC/IDSA, NICE, ECDC), I can narrow the answer further.
This is a shared conversation. Sign in to Orris to start your own chat.