Traumatic facial nerve palsy essay under 800 words with diagrams
Reading File
Finding Sources
Finding Sources
Searching Images
facial nerve anatomy diagram
Reading File
Reading File
Searching Images
traumatic facial nerve palsy temporal bone fracture
Finding Sources
Finding Sources
Reading File
I now have rich, comprehensive textbook content. Let me compile the essay.
Traumatic Facial Nerve Palsy
Introduction
Traumatic facial nerve palsy is a peripheral lower motor neuron palsy of cranial nerve VII (CN VII) caused by mechanical injury. It is a clinically significant condition because CN VII controls all muscles of facial expression; its disruption leads to ipsilateral facial weakness or complete paralysis, with functional, cosmetic, and psychological consequences. Traumatic causes are distinct from the idiopathic Bell's palsy and demand a different diagnostic and management approach.
Anatomy and Vulnerability
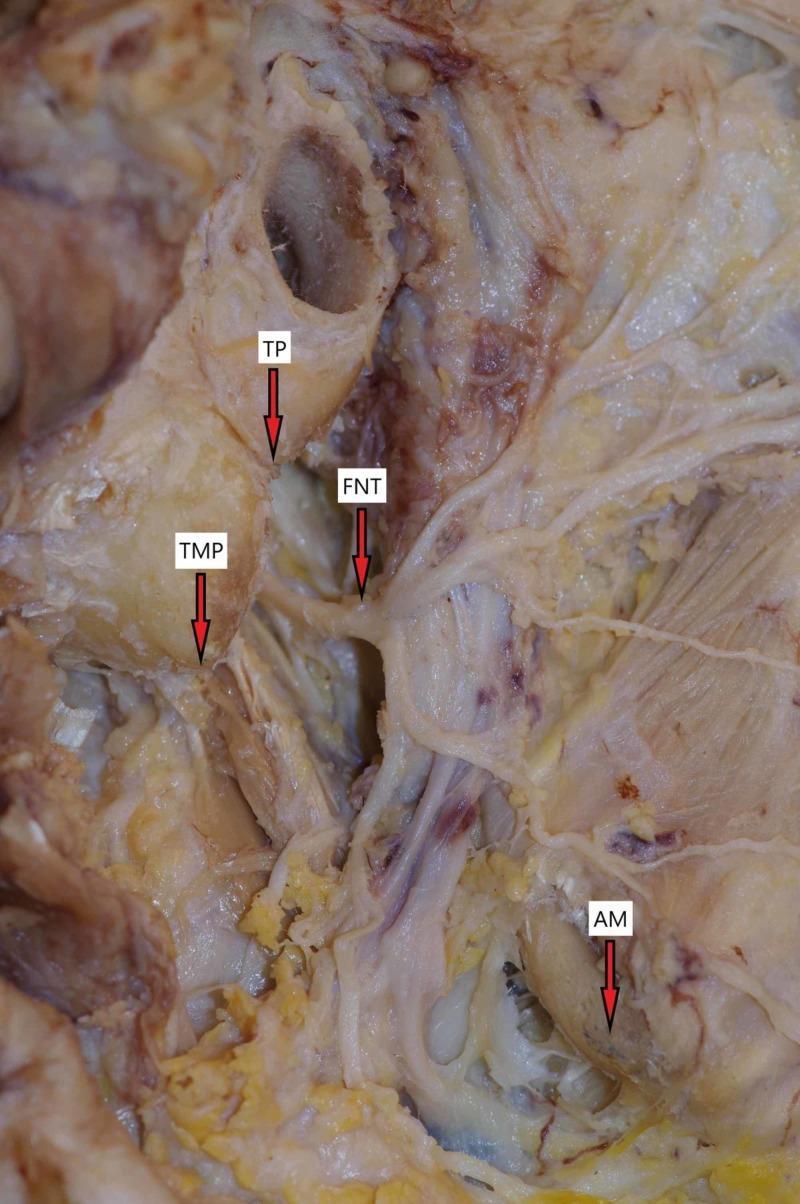
The facial nerve exits the brainstem at the pontomedullary junction and traverses the posterior cranial fossa before entering the internal auditory canal (IAC). From there it travels through the fallopian canal in four segments: the labyrinthine, tympanic (horizontal), mastoid (vertical), and extratemporal segments. After exiting the stylomastoid foramen, it enters the parotid gland and divides at the pes anserinus into temporal, zygomatic, buccal, marginal mandibular, and cervical branches.
The labyrinthine segment is the narrowest and most vulnerable — it is only ~0.7 mm wide and is the most common site of injury in temporal bone fractures.
Aetiology and Classification
Traumatic facial nerve palsy arises from two broad categories:
1. Temporal bone fractures (most common cause)
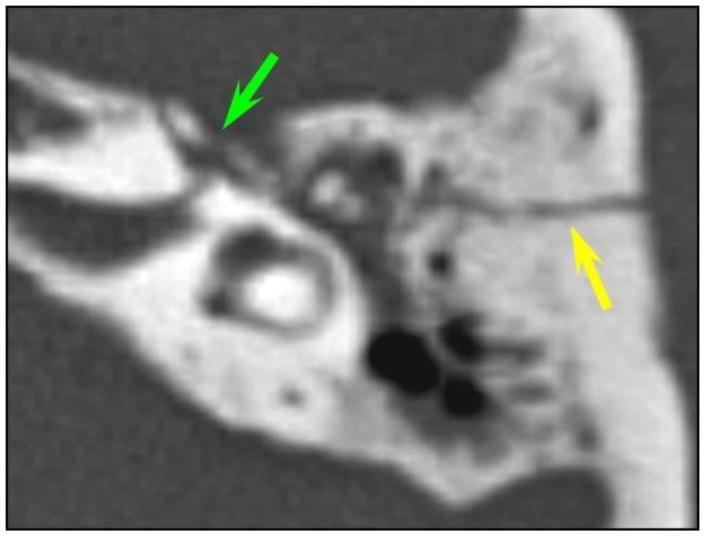
- Longitudinal fractures (70–80% of temporal bone fractures): run parallel to the petrous pyramid, typically caused by blows to the temporoparietal region. Facial nerve injury occurs in 10–25% of cases, usually at the geniculate ganglion or the tympanic segment.
- Transverse fractures (20–30%): perpendicular to the petrous pyramid, result from occipital impacts, and carry a higher risk of facial nerve involvement (50%). The labyrinthine segment is the most commonly injured site. Sensorineural hearing loss and vestibular dysfunction are also prominent.
- Mixed/oblique fractures are increasingly recognised as the dominant pattern on modern CT.
2. Extratemporal injuries
- Lacerations or blunt trauma to the face (parotid region, cheek, periorbital area)
- Birth trauma (forceps delivery) — predominantly affects the extratemporal trunk
- Iatrogenic: parotidectomy, mastoidectomy, and other skull base surgeries

Pathophysiology
The mechanisms of nerve injury are classified by Sunderland's grading:
| Grade | Injury | Prognosis |
|---|---|---|
| I (Neuropraxia) | Conduction block only; axons intact | Full recovery |
| II (Axonotmesis) | Axon disrupted; endoneurium intact | Good recovery |
| III | Axon + endoneurium disrupted | Variable |
| IV | Only epineurium intact | Poor without surgery |
| V (Neurotmesis) | Complete nerve transection | Requires surgical repair |
In temporal bone trauma, the mechanism is typically compression or oedema within the bony fallopian canal, causing a neuropraxia or axonotmesis — these cases often recover spontaneously. True neurotmesis (laceration or avulsion by bone fragments) requires surgical intervention.
Clinical Features
Onset timing is diagnostically critical:
- Immediate palsy at the time of injury strongly implies structural nerve disruption (compression, laceration) and carries a worse prognosis.
- Delayed palsy (hours to days post-injury) implies neuropraxia from oedema and has a significantly better prognosis — comparable to Bell's palsy.
Clinical findings include:
- Ipsilateral facial weakness (forehead, eye, mouth)
- Lagophthalmos (incomplete eye closure) — risk of corneal exposure
- Loss of nasolabial fold, drooping of the oral commissure
- Hyperacusis (if stapedius nerve involved)
- Loss of taste over anterior 2/3 tongue (chorda tympani involvement)
- Associated findings: haemotympanum, Battle's sign, CSF otorrhoea
Severity is graded using the House-Brackmann (HB) Scale (I = normal, VI = complete paralysis).
Investigations
Imaging:
- High-resolution CT of temporal bones is the investigation of choice. It characterises fracture type, identifies bone fragments in the fallopian canal, and guides surgical planning. MRI with gadolinium can assess nerve oedema and continuity but is less practical acutely.
Electrodiagnostic testing (most useful days 3–14 post-injury in complete paralysis):
- Electroneuronography (ENoG / EEMG): The most accurate electrodiagnostic test. It measures the evoked compound muscle action potential (CAP). Degeneration exceeding 90% within 6 days of traumatic onset is a threshold for considering surgical decompression — Cummings Otolaryngology Head and Neck Surgery.
- Nerve Excitability Test (NET): Compares threshold stimulus between affected and unaffected sides; >3.5 mA difference suggests degeneration.
- EMG: Presence of voluntary activity in the acute phase predicts good recovery. Fibrillation potentials (denervation) appear only 2–3 weeks post-injury.
Management
Conservative Management
- Indicated for delayed-onset palsy, incomplete palsy, or HB grades I–III.
- Corneal protection: lubricating eye drops, moisture chamber, taping the eyelid at night.
- Short-course corticosteroids are sometimes used but evidence for benefit in traumatic (vs. idiopathic) palsy is limited.
- Serial clinical assessment and electrodiagnostic monitoring.
Surgical Management
Indications for surgical decompression:
- Immediate complete palsy with radiological evidence of nerve compression by bone
- ENoG degeneration >90% within 6 days of injury
- Failure to recover or progressive deterioration
Surgical approaches:
- Middle cranial fossa (MCF) approach: Access the labyrinthine and geniculate ganglion segments. Preferred when hearing is serviceable.
- Transmastoid approach: Access the mastoid (vertical) and tympanic segments.
- Translabyrinthine approach: Used when hearing is already lost; provides broadest exposure.
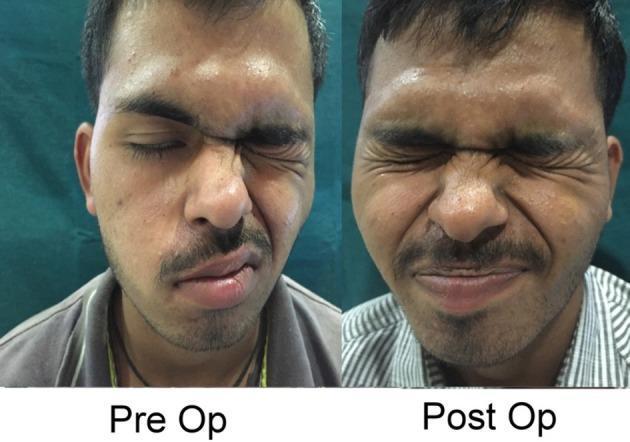
If the nerve is transected, primary nerve grafting (using the great auricular or sural nerve) is performed. The results of nerve repair are better than no repair but rarely achieve HB grade I.
Outcome data from non-operative series show spontaneous good recovery (HB I–II) in ~52–63% of complete traumatic palsies; surgical decompression series report good outcomes in ~70–85% — Cummings Otolaryngology.
Prognosis
Key prognostic factors:
- Timing of onset: Delayed palsy → better prognosis
- Degree of degeneration on ENoG: <90% → likely good recovery
- Fracture type: Longitudinal fractures carry better prognosis than transverse
- Site of injury: Labyrinthine segment injuries (narrowest canal) tend to be most severe
- Age: Children generally recover better than adults
Conclusion
Traumatic facial nerve palsy ranges from a transient neuropraxia that resolves fully with conservative care to a complete neurotmesis requiring surgical repair. Systematic evaluation — establishing onset timing, grade of palsy, radiological fracture pattern, and electrophysiological testing — drives management decisions. The primary immediate priority is corneal protection; the secondary priority is accurate prognostication to identify the minority of patients who require surgical decompression before irreversible neural degeneration occurs.
Sources: Cummings Otolaryngology Head and Neck Surgery; Schwartz's Principles of Surgery, 11th Ed.; Gray's Anatomy for Students
Pain management in advanced Head and neck cancer patients - essay under 800 words
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Loading Image
Reading File
I now have everything needed — rich textbook content from Cummings, the WHO ladder image, and comprehensive pharmacological detail. Let me compose the essay.
Pain Management in Advanced Head and Neck Cancer
Introduction
Pain is one of the most distressing and prevalent symptoms in advanced head and neck cancer (HNC). It arises from the tumour itself, from treatment (surgery, radiotherapy, chemotherapy), or from both simultaneously. Because HNC involves complex anatomy encompassing cranial nerves, muscles of mastication, swallowing, and the airway, the pain syndromes encountered are multidimensional and often refractory. Effective management requires systematic assessment, a stepwise pharmacological framework, and integration of non-pharmacological and interventional strategies within a multidisciplinary palliative team.
Nature and Mechanisms of Pain
Pain in advanced HNC is rarely of a single type. Three principal mechanisms operate concurrently:
- Nociceptive somatic pain: from tumour invasion of bone, muscle, or skin (e.g., mandibular erosion, skin infiltration). Characteristically well-localised, aching.
- Nociceptive visceral pain: from mucosal ulceration, airway involvement, oropharyngeal destruction. Often poorly localised; referred pain is common (e.g., otalgia referred via CN IX/X).
- Neuropathic pain: from perineural invasion or direct cranial nerve involvement; described as burning, shooting, or electric-shock in quality. Particularly common in nasopharyngeal and parotid tumours.
Myofascial pain is an often-overlooked contributor: biomechanical changes after extensive surgery or radiation therapy produce muscular trigger points that can generate significant chronic pain in the neck, shoulder, and jaw — Cummings Otolaryngology Head and Neck Surgery.
Breakthrough pain occurs in 40% of patients with cancer pain. It may be spontaneous, incident (triggered by a specific activity such as swallowing or dressing changes), or end-of-dose failure. Identifying the pattern directly guides management.
Pain Assessment
Accurate and repeated assessment is essential. The Numeric Rating Scale (NRS, 0–10) and Visual Analogue Scale (VAS) are validated for pain intensity. For patients with communication difficulties — common in advanced HNC due to tracheostomy, glossectomy, or cognitive impairment — the Faces Pain Scale or behavioural observation tools are substituted.
Assessment must also characterise pain type (nociceptive vs. neuropathic), temporal pattern, functional impact (swallowing, sleep, mobility), and psychological burden. Underassessment remains the single most common barrier to adequate pain control.
Pharmacological Management: The WHO Analgesic Ladder
In the 1980s, the World Health Organization developed a three-step analgesic ladder that remains the cornerstone of cancer pain management worldwide.
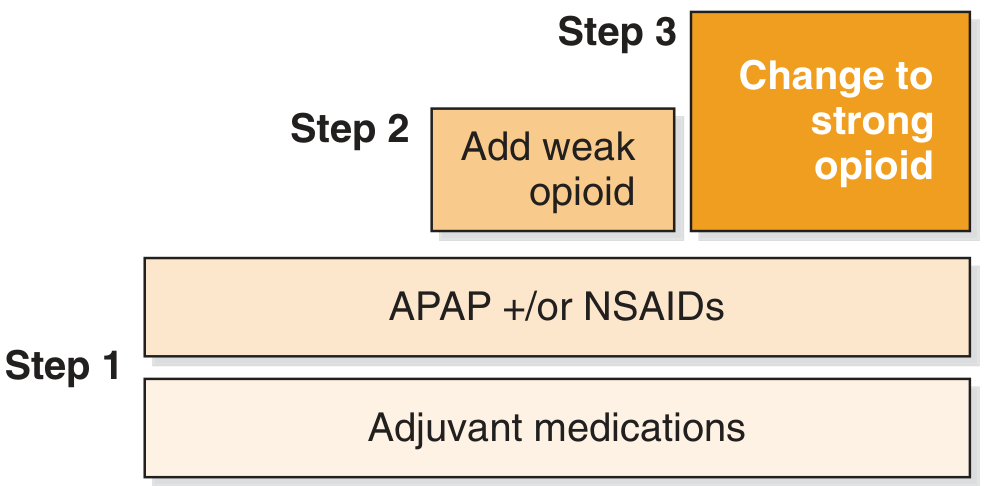
The five guiding principles accompanying the ladder are: by mouth (oral route preferred), by the clock (regular dosing, not PRN), by the ladder (titrate upward as needed), for the individual (dose to effect), and with attention to detail (anticipate side effects).
Step 1 — Mild pain: Paracetamol (up to 4 g/day) and/or NSAIDs. In HNC patients with dysphagia, intravenous paracetamol achieves faster onset and better first-hour analgesia than enteral routes. NSAIDs carry risk of mucosal bleeding and renal impairment — use with caution, particularly after mucositis or systemic chemotherapy.
Step 2 — Moderate pain: A weak opioid is added. Tramadol and codeine are the most used; tramadol also has serotonergic and noradrenergic activity that may assist neuropathic components. Codeine is a prodrug requiring CYP2D6 conversion — ultra-rapid metabolisers risk toxicity; poor metabolisers gain no benefit.
Step 3 — Severe pain: Strong opioids — morphine, oxycodone, fentanyl, hydromorphone, or methadone. Morphine remains the reference standard. In patients unable to swallow (tracheostomy, post-laryngectomy, severe mucositis), transdermal fentanyl patches or subcutaneous infusions via syringe driver are indispensable. Methadone has particular utility in neuropathic pain due to its NMDA-receptor antagonism.
Opioid side-effects (constipation, nausea, sedation, opioid-induced hyperalgesia) must be proactively managed. Laxatives should be prescribed prophylactically with every opioid prescription — Cummings Otolaryngology.
Adjuvant (Co-analgesic) Medications
Adjuvants potentiate analgesia and address specific pain mechanisms. They are used at every step of the ladder:
| Drug Class | Examples | Indication |
|---|---|---|
| Anticonvulsants | Gabapentin, pregabalin | Neuropathic pain, burning, shooting pain |
| Tricyclic antidepressants | Amitriptyline | Neuropathic pain; also aids sleep |
| Corticosteroids | Dexamethasone | Nerve compression, oedema, bone pain |
| Bisphosphonates | Zoledronic acid | Bone metastases pain |
| Ketamine (low-dose) | — | Opioid-refractory or neuropathic pain |
| Muscle relaxants | Baclofen, diazepam | Myofascial pain, trismus |
Gabapentin warrants specific mention: clinical studies in HNC patients demonstrate significant reduction in pain intensity and opioid requirements when used perioperatively and in the chronic cancer setting — Cummings Otolaryngology.
Non-Pharmacological and Interventional Approaches
When systemic analgesia is inadequate or poorly tolerated, interventional techniques offer targeted relief:
- Nerve blocks: Glossopharyngeal nerve block for refractory otalgia and oropharyngeal pain; trigeminal branch blocks for facial pain.
- Cervical plexus block: For neck pain following surgery or nodal invasion.
- Intrathecal/epidural analgesia: Rarely used in HNC but considered for refractory lower head/neck pain.
- Palliative radiotherapy: A single fraction of 8 Gy provides effective pain relief for bone metastases and ulcerating nodal disease.
- Physiotherapy and TENS: For myofascial pain syndromes post-treatment.
- Psychological support: Cognitive behavioural therapy, mindfulness, and chaplaincy. Pain catastrophising significantly amplifies perceived intensity and must be addressed.
Practical Challenges Unique to HNC
Several factors make pain management in HNC particularly challenging:
- Dysphagia and mucositis limit oral drug administration — alternative routes (transdermal, subcutaneous, IV, sublingual) must be anticipated.
- Trismus from radiation fibrosis restricts mouth opening and makes examination and drug administration difficult.
- Airway compromise raises safety concerns with sedating analgesics.
- Psychological distress — disfigurement, communication loss, and fear of suffocation amplify pain perception.
- Opioid misuse risk — careful screening and monitoring are necessary without allowing this concern to lead to undertreatment.
Conclusion
Pain in advanced head and neck cancer is complex, multimechanistic, and profoundly affects quality of life. Management centres on the WHO analgesic ladder, individualised to each patient's functional status, swallowing ability, and pain type. Neuropathic and breakthrough components demand specific adjuvant strategies. Non-pharmacological modalities and interventional techniques fill gaps where systemic therapy is insufficient. Early, proactive, multidisciplinary involvement — combining oncology, palliative care, pain medicine, and allied health professionals — is the standard of care.
Sources: Cummings Otolaryngology Head and Neck Surgery; Harrison's Principles of Internal Medicine, 22nd Ed.; Katzung's Basic and Clinical Pharmacology, 16th Ed.
This is a shared conversation. Sign in to Orris to start your own chat.