Bone anchored hearing device in detail according to standard ENT textbook
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Reading File
Loading Image
I now have all the content needed for a thorough, well-structured response. Here is a comprehensive account of bone-anchored hearing devices based on Cummings Otolaryngology Head and Neck Surgery.
Bone Anchored Hearing Device (BAHD / BAHA)
Source: Cummings Otolaryngology Head and Neck Surgery
Overview and Interface with the Auditory System
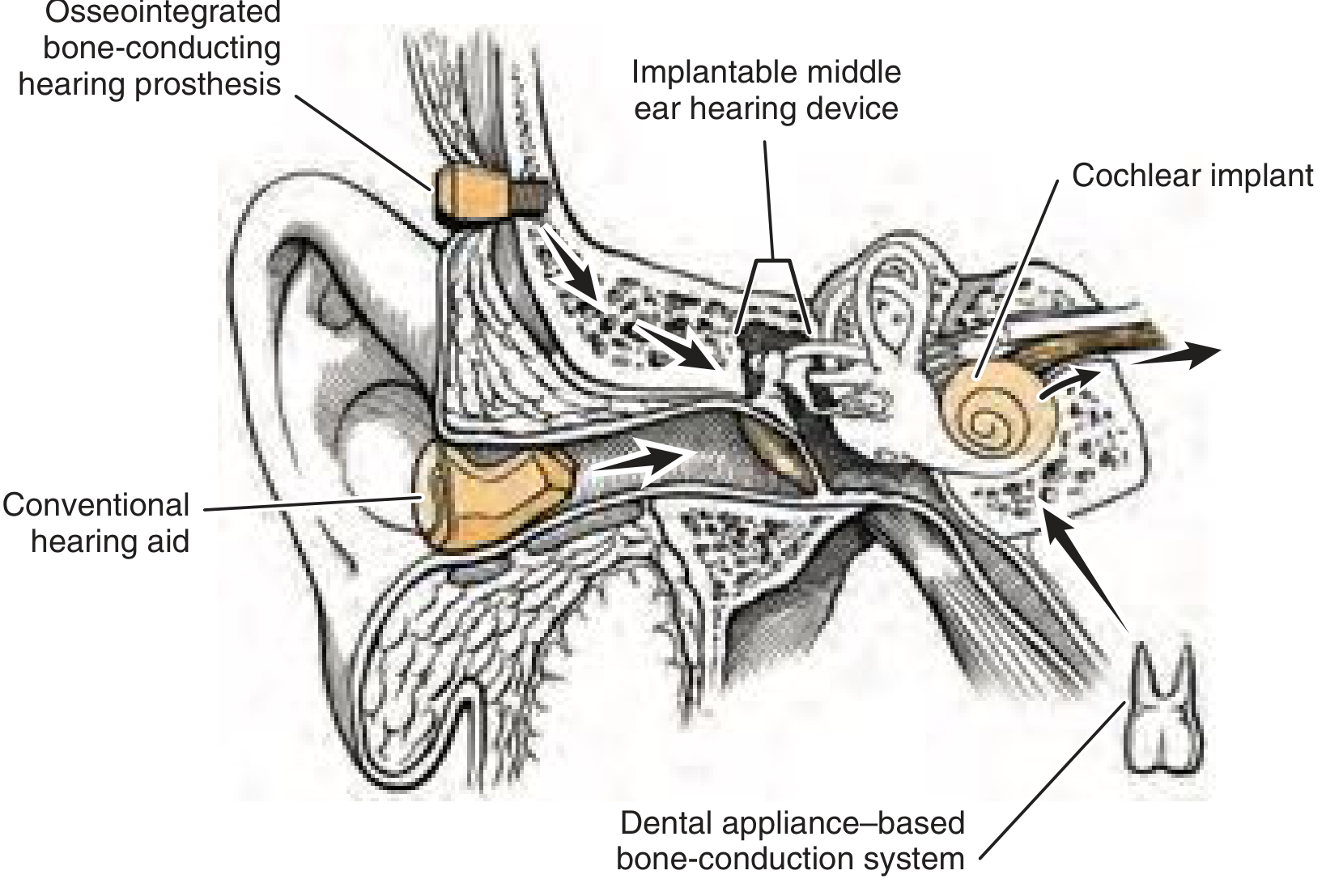
Fig. 159.1 — Osseointegrated bone-conducting hearing prostheses are applied to the retroauricular skull to provide hearing via bone conduction, bypassing the external and middle ear entirely.
Bone-anchored hearing devices belong to the category of osseointegrated bone-conducting hearing prostheses (OBHPs). Unlike conventional hearing aids that amplify sound within the canal, or middle ear implants that couple to the ossicles, OBHPs transmit sound energy directly to the skull bone, which in turn stimulates the cochlea via bone conduction — bypassing a diseased or absent outer/middle ear.
Rationale and Patient Population
Conventional hearing aids and surgery fail a specific subset of patients:
- Chronically draining ears (chronic suppurative otitis media)
- Mastoid bowl, meatoplasty, or surgically closed external auditory canals
- Aural atresia, canal stenosis, or microtia
- Otosclerosis or tympanosclerosis with contraindication to surgery
- Intolerance of ear mold occlusion
- Unilateral sensorineural hearing loss / single-sided deafness (SSD)
Non-implanted bone-conduction devices worn on headbands are limited by pain at the contact site, headache, skin irritation, soft-tissue attenuation, and variable placement. Coupling the bone vibrator to an osseointegrated implant circumvents all of these.
Historical Background
- Per-Ingvar Brånemark (Gothenburg, Sweden, 1960s) discovered that titanium could become firmly anchored in bone without interposed soft tissue — he coined the term "osseointegration."
- Osseointegration was initially applied to dental implants. By 1975, Brånemark's group began studying percutaneous skin-penetrating implants.
- Tjellström and colleagues first coupled a bone vibrator to an osseointegrated implant in 1977 at the Institute of Applied Biotechnology in Sweden.
- By the 1980s, a clinical program was established providing titanium percutaneous implants for facial prosthetics and bone-anchored hearing devices.
Principle of Osseointegration
Implant Material
Titanium is the standard. Two forms are used:
- Pure titanium: 99.75% pure
- Titanium alloy: 90% Ti, 6% Al, 4% V
Titanium's key property is its ability to form a tightly bonded, corrosion-resistant oxide layer on its surface. It is this oxide coating that interfaces with bone, creating a stable, reaction-free bond. This property differentiates titanium from all other metals.
Key Biophysical Requirements for Successful Osseointegration
- Implant surface chemistry — titanium oxide layer forms stable direct contact with bone
- Implant surface geometry — threaded design maximises surface area
- Surgical technique — atraumatic, low-speed drilling with copious irrigation to prevent thermal damage
- Osteocytes are lost if tissue is heated to ≥47°C for 1 minute
- High-speed drilling can generate temperatures as high as 89°C — irrigation is essential
- Biomechanical loading — optimal force transfer without micromotion at the interface
- Host bone quality — adequate cortical bone thickness (minimum ~3 mm in adults; skull may be thinner in children <5 years)
Implant Design
The standard BAHA/Ponto implant is a self-tapping titanium screw 3–4 mm in length. A titanium abutment connects the implant (percutaneously) to the external sound processor that snaps onto it. A loading period of 3 months in adults (and traditionally longer in children) is observed before loading the implant, allowing osseointegration to mature.
The BAHA System — Components

Fig. 159.10 — (A) Baha 4 Connect System with abutment and titanium screw implant. (B) Cordelle II device for patients with greater sensorineural component, offering increased amplification.
The system has three components:
| Component | Description |
|---|---|
| Titanium fixture (implant) | Screwed into the mastoid cortex; osseointegrates over ~3 months |
| Abutment | Percutaneous titanium post connecting implant to the external processor |
| Sound processor | External device worn behind the ear, snaps onto the abutment |
The Ponto (Oticon Medical, introduced 2009) is the main competitor to BAHA (Cochlear Americas). Both are percutaneous devices and are considered functionally similar.
Indications
Conductive and Mixed Hearing Loss
- Pure-tone average (PTA) bone conduction threshold <45 dB HL = best results (89% improvement rate)
- PTA 45–60 dB HL: still beneficial (61% improvement)
- PTA >60 dB HL: limited benefit (22% improvement) — poor cochlear reserve limits what bone conduction can achieve
- Minimum speech discrimination score on the better ear should be adequate for meaningful benefit
Single-Sided Deafness (SSD) / Unilateral SNHL
- The BAHA acts as a contralateral routing of signal via bone conduction (CROS via bone)
- Sound picked up on the deaf side is transmitted across the skull to stimulate the better cochlea
- Studies show significantly better speech recognition in noise with BAHA vs. CROS hearing aids
- Sound localization does NOT improve (only one cochlea is stimulated)
- Importantly, BAHA reduces informational masking — a study by May et al. showed that adding OBCH to SSD patients reduced error rate in competing sentence conditions from 40% to 24%
- Lin et al. found consistent satisfaction with Baha amplification and poor acceptance of CROS amplification in unilateral SNHL
Surgical Technique
Single-Stage Procedure
Modern BAHA implantation is performed in a single stage (Figs. 159.10 and 159.11 in Cummings). It can be performed under general or local anesthesia, including in outpatient settings.
Traditional Technique
- Site selection: Posterior to the auricle, typically 5–5.5 cm from the external auditory canal
- Skin preparation: Hair-bearing skin is thinned and hair follicles removed. The final skin flap should be <1 mm thick at the implant site. A Cochlear-specific dermatome minimizes skin necrosis
- Bony preparation: Cortical bone drilled with a slow high-torque drill (1500–3000 rpm) using sharp, disposable bits with copious irrigation
- Implant insertion: Self-tapping titanium screw inserted; abutment attached
- Skin closure: Edges sutured to periosteum to minimize tension
Minimally Invasive Techniques (Modern)
- Linear skin incision without skin thinning, or a single skin punch
- Abutment must protrude ≥3 mm above the skin surface
- Multiple studies show no increase in skin complications vs. traditional techniques; some show lower complication rates
- Advantages: significantly decreased surgical time, decreased healing time, improved cost-effectiveness
Pediatric Considerations
- Children younger than 5 years of age may have inadequate skull thickness for standard implant
- A soft-band headband is used in young children as a non-surgical interim solution, allowing early auditory stimulation while awaiting adequate skull development
- Once older, children proceed to surgical implantation
Outcomes and Results
Large Series (Håkansson et al., 10-year experience, n=147):
| PTA Bone Threshold | Subjective Hearing Improvement |
|---|---|
| <45 dB HL | 89% improved |
| 46–60 dB HL | 61% improved |
| >60 dB HL | 22% improved |
- Speech discrimination scores improved from 14% unaided → 81% with BAHA (89% if PTA <45 dB)
- Recommended PTA threshold for "high success rate": <45 dB HL (improvement still expected up to 60 dB)
U.S. Experience (initial review):
- Common indications: chronic otitis media/draining ears, EAC stenosis/aural atresia, post-skull base surgery canal closure
- Average functional gain: ~25 dB across speech frequencies
Children (Birmingham experience):
- 4 of 7 children showed ≥5 dB PTA improvement over their prior aid
- Average speech discrimination ~90% (vs. 0% unaided)
Complications
Skin/Soft Tissue Complications (Holgers Classification)
| Grade | Finding |
|---|---|
| 0 | No reaction |
| 1 | Slight redness |
| 2 | Moist/red skin, no granulation |
| 3 | Granulation tissue, treatment required |
| 4 | Skin overgrowth, device removal may be needed |
Meta-analysis (Kiringoda & Lustig, 20 studies, n=2310):
- Skin reactions (Holgers Grade 2–4): 2.4–38.1%
- Failure of osseointegration: 0–18%
- Revision surgery (adults): 1.7–34.5%
- Revision surgery (pediatric): 0–44.4%
Note: Wide ranges reflect heterogeneity and variable study quality.
Other complications:
- Wound infection, hematoma
- Implant extrusion
- Numbness/dysesthesia around implant site
Alternatives: Transcutaneous Bone-Anchored Hearing Devices
To address limitations of percutaneous abutment (skin breakdown, daily maintenance, poor cosmesis), several transcutaneous (no skin-penetrating post) systems have been developed:
1. Alpha 2 (Sophono Inc.)
- First used in Germany 2006; FDA clearance 2011
- External component: sound processor + bone-conducting oscillator
- Implanted component: titanium-encased samarium-cobalt twin magnetic implant, secured with 5 titanium screws into a bony recess (≥2.6 mm deep)
- Magnetic coupling transmits vibrations transcutaneously — no percutaneous abutment
- Skin flap thinned to 4–6 mm in adults
- FDA conditional MRI approval up to 3T; head shadow ~5 cm
- Indications: age >5 years; CHL/MHL with conductive component ≤45 dB; SSD with BC ≤20 dB
2. BAHA Attract (Cochlear)
- FDA clearance 2013
- Magnet-based transcutaneous system
- External magnet-coupled processor; implanted magnet
3. Bonebridge (MED-EL)
- CE mark (Europe); FDA approval July 2018
- Unique active transcutaneous design: the transducer itself is implanted subcutaneously
- Relatively newer data in the literature
Trade-off of transcutaneous systems: Eliminate skin complications and maintenance burden, but soft tissue interposition between the magnet and the implant slightly attenuates vibrational energy transmission compared to percutaneous systems — reducing acoustic output, particularly at high frequencies.
Summary Comparison
| Feature | Percutaneous BAHA/Ponto | Transcutaneous (Alpha 2 / Attract / Bonebridge) |
|---|---|---|
| Skin penetration | Yes — abutment exits skin | No |
| Energy transmission efficiency | Higher (direct bone coupling) | Slightly lower (skin/tissue attenuation) |
| Skin complications | Possible (Holgers reactions) | Eliminated |
| Daily maintenance | Required (abutment cleaning) | Not required |
| Cosmesis | Visible abutment | Better (hidden) |
| MRI compatibility | Processor removed; titanium screw safe | Depends on device generation |
| Indications (CHL) | PTA BC <45 dB (optimal) | Similar; slightly narrower output range |
— Cummings Otolaryngology Head and Neck Surgery, Chapter 159 (Implantable Hearing Devices)
This is a shared conversation. Sign in to Orris to start your own chat.