give an outline on mendelian disorders, autosomal dominant/recessive, x-linked dominant/recessive w.r.t. Robbins and Cotran pathology
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material from Robbins & Cotran. Let me compile the full detailed outline.
Mendelian Disorders — Outline per Robbins, Cotran & Kumar: Pathologic Basis of Disease
I. Overview: Categories of Human Genetic Disease
Genetic disorders are broadly grouped into three categories:
- Monogenic (Mendelian) Disorders — single-gene mutations with large effects; highly penetrant; follow classic Mendelian inheritance (autosomal dominant, autosomal recessive, X-linked).
- Chromosomal Disorders — structural or numerical alterations affecting autosomes or sex chromosomes; often arise de novo in the germline.
- Complex Multigenic (Multifactorial) Disorders — multiple genetic variants + environmental factors (e.g., atherosclerosis, diabetes, hypertension).
A fourth category of somatically acquired mutations (driving clonal hematopoiesis, cancers, overgrowth syndromes) is also increasingly recognized.
II. Mutations Underlying Mendelian Disorders
Mutations are permanent changes in DNA sequence. Key types:
| Type | Mechanism | Example |
|---|---|---|
| Point mutation | Single nucleotide substitution | Missense → altered amino acid; Nonsense → premature stop codon |
| Frameshift | Insertion or deletion of non-multiples of 3 bases | 4-bp insertion in HEXA → Tay-Sachs disease |
| Trinucleotide repeat expansions | Expansion of repetitive sequences | Fragile X, Huntington |
| Deletions/duplications | Larger DNA copy number changes | Affects gene dosage |
Functional Consequences of Mutations
- Loss-of-function: enzyme/transport protein deficiencies (usually AR)
- Gain-of-function: constitutively active or toxic protein products (usually AD)
Key Concepts
- Pleiotropism: one mutation → many end-organ effects (e.g., sickle cell anemia: hemolysis + vascular occlusion + splenic fibrosis + bone changes)
- Genetic heterogeneity: different gene loci → same phenotype (e.g., congenital deafness)
III. Transmission Patterns of Single-Gene Disorders
A. Autosomal Dominant (AD) Disorders
Fundamental Rules:
- Manifested in the heterozygous state
- Both males and females are equally affected and can transmit
- Affected parent → 50% chance each child is affected
- Vertical transmission through generations
Exceptions and Modifiers:
| Concept | Definition |
|---|---|
| De novo mutation | No affected parent; propensity for new mutations in germ cells of older fathers (e.g., FGFR2/FGFR3 mutations → Apert syndrome, achondroplasia) |
| Incomplete penetrance | Mutant gene carried but disease not expressed (e.g., 50% penetrance = 50% carrying the gene are affected) |
| Variable expressivity | Trait present in all carriers but at differing severity |
| Anticipation | Earlier onset and increasing severity in successive generations (e.g., trinucleotide repeat disorders) |
Biochemical Mechanisms in AD Disorders:
- Loss of regulatory proteins (e.g., tumor suppressors) — one mutant allele reduces the gene product sufficiently to alter function
- Gain-of-function — mutant protein acquires new toxic activity
- Dominant-negative effect — mutant product interferes with normal allele product (common with structural proteins like collagen and fibrillin)
Examples of AD Disorders (Table 5.1):
| System | Disorder |
|---|---|
| Neurologic | Huntington disease, neurofibromatosis, myotonic dystrophy |
| Urologic | Adult polycystic kidney disease |
| Cardiovascular | Familial hypercholesterolemia, hereditary hemorrhagic telangiectasia |
| Hematopoietic | Hereditary spherocytosis, von Willebrand disease |
| Skeletal | Marfan syndrome, Ehlers-Danlos syndrome (some variants), achondroplasia |
| Metabolic | Familial hypercholesterolemia |
Featured AD Disorder — Marfan Syndrome:
- Gene: FBN1 (fibrillin-1), chromosome 15q21
- Fibrillin-1 is an extracellular glycoprotein → component of microfibrils in connective tissue
- Mutations disrupt elastin meshworks and cause excess release of TGF-β from ECM (unopposed TGF-β signaling → tissue remodeling)
- Morphology:
- Skeletal: tall stature, dolichocephaly, arachnodactyly, hypermobile joints, pectus excavatum/carinatum, kyphoscoliosis
- Ocular: bilateral ectopia lentis (upward/outward lens subluxation) — a near-pathognomonic sign
- Cardiovascular: mitral valve prolapse (40–50%), medial degeneration of aorta → aortic dilation, aortic regurgitation, aortic dissection (most common cause of death)
B. Autosomal Recessive (AR) Disorders
Fundamental Rules:
- Disease manifests only in the homozygous state (both alleles mutated)
- Heterozygous carriers are phenotypically normal
- Both parents are typically carriers (heterozygotes)
- 25% chance of affected offspring per pregnancy; 50% carriers; 25% normal
- Often presents in siblings, not parents ("horizontal" pedigree pattern)
- More common when parents are consanguineous
Key Features Distinguishing AR from AD:
- Expression is more uniform (less variable expressivity)
- Complete penetrance is more common
- Onset often in early childhood (enzyme deficiencies manifest when both alleles are absent)
- Enzyme deficiency is the predominant mechanism — 50% residual enzyme from one functional allele is usually sufficient (hence carriers are unaffected)
Examples of AR Disorders (Table 5.2):
| System | Disorder |
|---|---|
| Metabolic | Cystic fibrosis, phenylketonuria, galactosemia, homocystinuria, lysosomal storage diseases, α₁-antitrypsin deficiency, Wilson disease, hemochromatosis, glycogen storage diseases |
| Hematopoietic | Sickle cell anemia, thalassemias |
| Endocrine | Congenital adrenal hyperplasia |
| Skeletal | Ehlers-Danlos syndrome (some variants), alkaptonuria |
| Nervous | Spinal muscular atrophy, Friedreich ataxia, neurogenic muscular atrophies |
Featured AR Disorder — Lysosomal Storage Diseases (prototype of AR enzyme deficiency):
- Inherited deficiency of lysosomal enzymes → incomplete catabolism → accumulation of insoluble intermediates within lysosomes
- Pathogenic consequences:
- Primary accumulation — engorged lysosomes interfere with cell function
- Defective autophagy — impaired mitophagy → dysfunctional mitochondria persist → free radical generation → intrinsic apoptosis
- Secondary accumulation — aggregation-prone proteins (α-synuclein, Huntingtin) accumulate
- ~70 lysosomal storage diseases identified; frequency ~1 in 5000 live births
- Treatment approaches: (1) Enzyme replacement / gene therapy; (2) Substrate reduction therapy; (3) Molecular chaperone therapy
C. X-Linked Disorders
General Principles:
- Caused by mutations in genes on the X chromosome; males are hemizygous (no corresponding Y-linked locus to compensate)
- Y chromosome male-specific region encodes few genes (mostly spermatogenesis); Y-linked mutations → male infertility → cannot be transmitted
C1. X-Linked Recessive (XLR) Disorders
Fundamental Rules:
- Males are predominantly affected (hemizygous — one mutant X is sufficient for disease)
- Females are typically carriers (heterozygous); generally unaffected because of the normal allele
- An affected male does not transmit disorder to sons (sons inherit Y chromosome from father); all daughters of an affected male are obligate carriers
- Sons of heterozygous women: 50% affected; daughters: 50% carriers
Lyon Hypothesis (X-Inactivation):
- In females, one X chromosome is randomly inactivated in each somatic cell → females are mosaics
- In most carriers, random inactivation results in ~50% cells with the normal X active → no disease
- If X-inactivation is skewed to favor inactivation of the wild-type allele → manifesting carrier female
Examples of XLR Disorders (Table 5.3):
| System | Disorder |
|---|---|
| Musculoskeletal | Duchenne muscular dystrophy |
| Hematopoietic | Hemophilia A and B, chronic granulomatous disease, G6PD deficiency |
| Immune | Agammaglobulinemia (Bruton), Wiskott-Aldrich syndrome |
| Metabolic | Diabetes insipidus (nephrogenic), Lesch-Nyhan syndrome |
| Nervous | Fragile X syndrome |
Featured XLR Disorder — G6PD Deficiency:
- X-linked; enzyme deficiency → episodic hemolytic anemia triggered by infection or oxidant drugs
- Expressed principally in males; rare manifesting carrier females result from skewed X-inactivation
C2. X-Linked Dominant (XLD) Disorders
Fundamental Rules:
- Rare; caused by dominant mutations on X chromosome
- Heterozygous females are affected (unlike XLR where females are carriers)
- Affected males tend to be more severely affected than females, or the condition may be lethal in males (e.g., incontinentia pigmenti)
- Transmission: affected mother → 50% daughters affected, 50% sons affected; affected father → all daughters affected, no sons affected
Examples:
- Fragile X syndrome (technically complex — trinucleotide expansion, discussed separately under non-classic inheritance)
- Incontinentia pigmenti (IKBKG/NEMO mutations) — lethal in males, affects females with skin, eye, neurologic, and dental abnormalities
- Rett syndrome (MECP2 mutations) — predominantly affects females
- X-linked hypophosphatemia (PHEX mutations) — rickets, affects both sexes but females less severely
IV. Comparing the Four Inheritance Patterns at a Glance
| Feature | AD | AR | XLR | XLD |
|---|---|---|---|---|
| State in which disease appears | Heterozygous | Homozygous | Hemizygous (males) | Heterozygous (females) + hemizygous (males) |
| Sex affected | Both equally | Both equally | Males >> Females | Both; females may be less severely affected |
| Carrier state | Not applicable | Heterozygotes | Heterozygous females | Not applicable (heterozygotes are affected) |
| Father → son transmission | Yes | Yes | No | No (sons get Y) |
| Risk to offspring of carrier | 50% affected | 25% affected, 50% carriers | Sons 50% affected; daughters 50% carriers | 50% children affected |
| Variable expressivity | Common | Less common | Common | Variable |
| New mutations | Common | Rare | Occasional | Occasional |
| Key molecular mechanism | Gain-of-function / dominant-negative / haploinsufficiency | Enzyme deficiency | Enzyme/protein deficiency (hemizygous) | Dominant gain- or loss-of-function on X |
V. Selected Specific Diseases Covered in Detail by Robbins & Cotran
| Disorder | Inheritance | Gene/Defect |
|---|---|---|
| Marfan syndrome | AD | FBN1 (fibrillin-1) — TGF-β excess |
| Familial hypercholesterolemia | AD | LDLR (LDL receptor) loss-of-function |
| Huntington disease | AD | HTT CAG repeat expansion |
| Neurofibromatosis type 1 | AD | NF1 (neurofibromin) |
| Cystic fibrosis | AR | CFTR — defective Cl⁻ channel |
| Phenylketonuria | AR | PAH (phenylalanine hydroxylase) deficiency |
| Sickle cell anemia | AR | HBB — Glu→Val substitution at position 6 |
| Tay-Sachs disease | AR | HEXA — 4-bp insertion → frameshift → absent hexosaminidase A |
| Gaucher disease | AR | GBA (glucocerebrosidase) deficiency |
| α₁-Antitrypsin deficiency | AR | SERPINA1 — misfolded protein accumulation in liver + uninhibited elastase in lungs |
| Duchenne muscular dystrophy | XLR | DMD (dystrophin) — frameshift deletions → absent dystrophin |
| Hemophilia A | XLR | F8 (Factor VIII) deficiency |
| G6PD deficiency | XLR | G6PD — oxidant-induced hemolysis |
| Fragile X syndrome | XLD/complex | FMR1 — CGG trinucleotide repeat expansion |
VI. Exceptions and Departures from Classic Mendelian Inheritance
Robbins & Cotran also highlights non-classic patterns that are covered in the same chapter:
- Trinucleotide repeat disorders (anticipation) — Huntington, myotonic dystrophy, Fragile X
- Genomic imprinting — Prader-Willi (paternal 15q11-q13 deletion) vs. Angelman syndrome (maternal deletion same region)
- Mitochondrial inheritance — maternally transmitted; affects organs with high energy demand
- Mosaicism — somatic or germline; explains variable expressivity
- Uniparental disomy — both copies of a chromosome derived from one parent
Source: Robbins, Cotran & Kumar — Pathologic Basis of Disease (ISBN 9780443264528), Chapter 5: Genetic Disorders, pp. 139–165
tutorial on microcytic and macrocytic anemia
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Reading File
Loading Image
I have all the content needed for a comprehensive tutorial. Let me now compose the full tutorial.
Tutorial: Microcytic & Macrocytic Anemias
Based on Robbins & Kumar Basic Pathology and Robbins, Cotran & Kumar Pathologic Basis of Disease
Conceptual Framework: Classifying Anemia by MCV
| MCV | Category | Key Causes |
|---|---|---|
| < 80 fL | Microcytic | Iron deficiency, thalassemia, anemia of chronic disease, sideroblastic anemia |
| 80–100 fL | Normocytic | Hemolytic anemias, aplastic anemia, acute blood loss |
| > 100 fL | Macrocytic | Megaloblastic (B12/folate deficiency), non-megaloblastic (liver disease, hypothyroidism, drugs) |
PART I — MICROCYTIC ANEMIAS (MCV < 80 fL)
The unifying pathophysiology is impaired hemoglobin synthesis, affecting heme (iron deficiency, sideroblastic), globin (thalassemia), or both.
1. Iron Deficiency Anemia (IDA)
Epidemiology: The most common nutritional deficiency worldwide. ~10% of people in high-resource countries and 25–50% in low-resource countries are anemic, with IDA the leading cause.
Iron Metabolism (Essential Background)
- Total body iron: ~3.5 g (men) / ~2.5 g (women)
- Functional pool (80%): Hemoglobin, myoglobin, iron-containing enzymes (catalase, cytochromes)
- Storage pool (15–20%): Ferritin and hemosiderin in liver macrophages, spleen, and bone marrow
| Parameter | Normal value |
|---|---|
| Serum iron | ~120 µg/dL (men), ~100 µg/dL (women) |
| Transferrin saturation | ~33% |
| TIBC | 300–350 µg/dL |
| Dietary iron (Western diet) | 10–20 mg/day |
| Daily iron loss | 1–2 mg/day (mucosal/skin cell shedding) |
Iron absorption pathway (duodenum):
- Fe³⁺ → Fe²⁺ via duodenal cytochrome B (ferric reductase)
- Fe²⁺ enters enterocyte via DMT-1 (divalent metal transporter-1)
- Fe²⁺ exits basolateral membrane via ferroportin
- Reoxidised to Fe³⁺ by hephaestin/ceruloplasmin → binds transferrin
Hepcidin: A liver-secreted peptide that negatively regulates ferroportin. Iron levels are sensed by HFE protein on hepatocytes → rising iron → rising hepcidin → less ferroportin → less absorption. Hepcidin is also upregulated by IL-6 (inflammation) and downregulated by erythroferrone (from erythroblasts during active erythropoiesis).
Causes of IDA
| Setting | Cause |
|---|---|
| High-resource countries | Chronic blood loss — GI tract (peptic ulcer, colon cancer, hemorrhoids); female genital tract (menorrhagia) |
| Low-resource countries | Inadequate intake — vegetarian diets, low bioavailability |
| Universal | Increased demand — pregnancy, infancy |
| Malabsorption | Celiac disease, gastritis, post-gastrectomy |
Stages of Iron Deficiency (in sequence)
- Depletion of iron stores → ↓ serum ferritin, absent bone marrow iron staining; no anemia yet
- Iron-limited erythropoiesis → ↓ serum iron, ↑ TIBC, ↓ transferrin saturation
- Frank IDA → microcytic hypochromic anemia, ↑ erythropoietin (marrow response blunted by iron lack)
Morphology
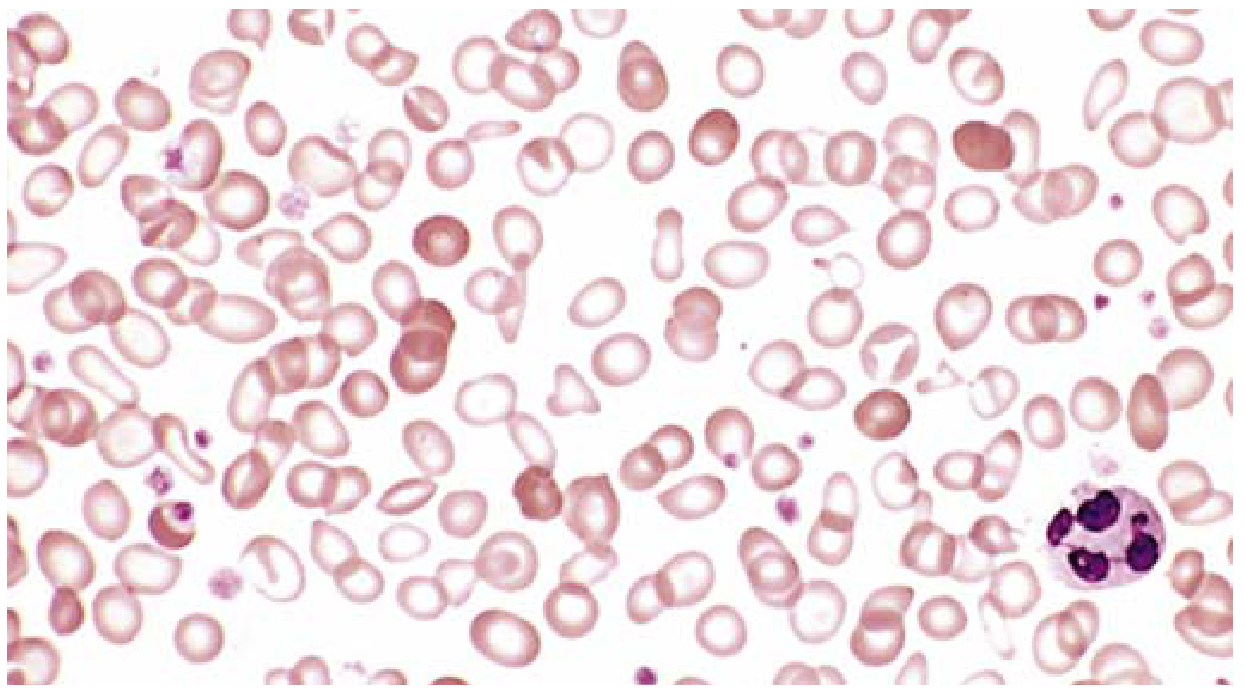
Fig. 10.10 — Iron deficiency anemia peripheral smear: microcytic, hypochromic red cells with increased central pallor. Scattered fully haemoglobinised cells from a recent transfusion appear darker. (Robbins Basic Pathology)
- Microcytic, hypochromic RBCs (MCV ↓, MCH ↓, MCHC ↓)
- Increased central pallor (>1/3 of cell diameter)
- Anisocytosis + poikilocytosis
- Platelets often elevated (reactive thrombocytosis)
- Reticulocyte count: normal or slightly low (response blunted)
Lab Findings Summary
| Test | IDA | ACD | Thalassemia trait |
|---|---|---|---|
| Serum iron | ↓ | ↓ | Normal |
| Ferritin | ↓ | ↑ | Normal/↑ |
| TIBC | ↑ | ↓ | Normal |
| Transferrin saturation | ↓ | ↓ | Normal |
| RBC count | ↓ | ↓ | ↑ or normal |
| RDW | ↑ | Normal | Normal/↑ |
Clinical Features
- Often mild and asymptomatic
- Weakness, listlessness, pallor in severe cases
- Long-standing: koilonychia (spoon nails), thin/flat nails
- Pica — compulsion to eat non-food items (dirt, clay, ice/pagophagia) — a neurobehavioral complication
- Angular cheilitis, glossitis (sore tongue)
- Impaired cognitive performance and reduced immunocompetence
"Persons often die with iron deficiency anemia but virtually never of it. Microcytic hypochromic anemia is not a disease but a symptom — always investigate the underlying cause." — Robbins Basic Pathology
2. Anemia of Chronic Disease (ACD) / Anemia of Inflammation
A functional iron deficiency — iron is abundant but sequestered and unavailable for erythropoiesis.
Common underlying conditions:
- Chronic infections: osteomyelitis, bacterial endocarditis, lung abscess
- Chronic immune disorders: rheumatoid arthritis, Crohn disease
- Cancers: Hodgkin lymphoma, lung/breast carcinoma
Pathogenesis: Pro-inflammatory cytokines (especially IL-6) → ↑ hepatic hepcidin → hepcidin downregulates ferroportin on marrow macrophages → iron is trapped in macrophages and cannot be delivered to erythroblasts. Additionally, chronic inflammation blunts renal erythropoietin synthesis.
Key distinguishing lab feature:
- Serum iron: ↓ (same as IDA)
- Ferritin: ↑ (iron sequestered, not depleted) — key differentiator
- TIBC: ↓ (unlike IDA where TIBC is ↑)
- Red cells: mildly hypochromic and microcytic or normocytic
Treatment: Treat the underlying condition; erythropoietin + iron can temporarily improve anemia.
3. Thalassemias
Definition: Inherited disorders of globin chain synthesis causing reduced (or absent) production of α- or β-globin chains.
Genetics:
- β-globin gene: single gene on chromosome 11 (mutations = mainly point mutations affecting transcription, splicing, or translation of β-globin mRNA)
- α-globin genes: two tandem genes on chromosome 16 per haploid genome (4 total; mutations = mainly gene deletions)
- Autosomal codominant inheritance
Pathogenesis: Reduced globin synthesis → (1) hemoglobin deficiency → microcytic hypochromic anemia; (2) excess unpaired globin chains precipitate → intracellular inclusions → RBC membrane damage → hemolysis and ineffective erythropoiesis.
β-Thalassemia
| Syndrome | Genotype | Clinical Features |
|---|---|---|
| β-Thalassemia major (Cooley anemia) | β⁰/β⁰ (no β-chain) | Severe transfusion-dependent anemia; splenomegaly; growth retardation; extramedullary hematopoiesis; facial bone changes ("chipmunk face"); iron overload |
| β-Thalassemia intermedia | β⁺/β⁰ or β⁺/β⁺ | Moderately severe; transfusions not required |
| β-Thalassemia minor (trait) | β⁺/β (one normal allele) | Asymptomatic; mild/absent anemia; ↑ HbA2; often mistaken for IDA — MCV low but RBC count high |
α-Thalassemia
| Syndrome | Gene deletions | Clinical Features |
|---|---|---|
| Silent carrier | 1 deleted (−/α, α/α) | No abnormality; asymptomatic |
| α-Thalassemia trait | 2 deleted | Asymptomatic; resembles β-thal minor |
| HbH disease | 3 deleted (−/−, −/α) | Moderate anemia (resembles β-thal intermedia); HbH (β₄ tetramers) |
| Hydrops fetalis | 4 deleted (−/−, −/−) | Lethal in utero; Hb Bart's (γ₄ tetramers); incompatible with extrauterine life |
Key point: Thalassemia trait is commonly misdiagnosed as IDA — distinguish by: normal/↑ ferritin, normal/↑ TIBC, ↑ RBC count, ↑ HbA2 on HPLC (in β-thal minor).
4. Sideroblastic Anemia
Defining lesion: Ringed sideroblasts — abnormal erythroid precursors in which iron-laden mitochondria form a perinuclear ring (seen on Prussian blue stain of bone marrow).
Mechanism: Disruption of heme synthesis → iron cannot be incorporated into protoporphyrin → accumulates in mitochondria around the nucleus.
Types:
| Form | Cause |
|---|---|
| Inherited (X-linked) | Mutations in ALAS2 gene (ALA synthase 2 — first step of heme synthesis) |
| Inherited (AR) | Mutations in SLC25A38 (glycine importer) |
| Acquired — MDS | Myelodysplastic syndrome (most common acquired form) |
| Acquired — drugs/toxins | Ethanol, isoniazid, pyrazinamide, linezolid |
| Acquired — nutritional | Copper deficiency (also zinc excess) |
Clinical: Microcytic anemia in inherited forms; dimorphic RBC population (microcytic + normocytic/macrocytic mix) in acquired forms. Copper deficiency also causes myelopathy.
Treatment: Pyridoxine (vitamin B6) for ALAS2 mutations (some respond); discontinue offending drug for acquired forms; treat underlying MDS.
PART II — MACROCYTIC ANEMIAS (MCV > 100 fL)
Divided into megaloblastic and non-megaloblastic types.
Megaloblastic Anemias
Common theme: Impaired DNA synthesis → nuclear-cytoplasmic asynchrony → ineffective hematopoiesis.
Pathogenesis: Vitamin B12 and folate are required for synthesis of thymidine (one of the four DNA bases). Deficiency → defective DNA replication → rapidly dividing cells most affected (marrow, GI epithelium). Two consequences:
- Many progenitors trigger DNA damage response → apoptosis (ineffective erythropoiesis)
- Surviving progenitors produce fewer, larger red cells (fewer cell divisions → larger cells)
Universal Morphologic Features of Megaloblastic Anemia
- Macro-ovalocytes (large, oval RBCs without central pallor — hyperchromic appearance, but MCHC is not truly elevated)
- Marked anisocytosis and poikilocytosis
- Hypersegmented neutrophils (5+ lobes in a single neutrophil, or ≥1 neutrophil with 6+ lobes) — pathognomonic
- Low reticulocyte count
- Hypercellular bone marrow with megaloblastic changes: giant bands, giant metamyelocytes, large erythroid precursors with immature-appearing ("open") nuclei relative to mature cytoplasm
5. Folate Deficiency Anemia
Sources of folate: Green leafy vegetables, liver, dairy. Heat-labile (destroyed by cooking).
Body stores: Only 5–20 mg total; sufficient for only 3–4 months — deficiency develops quickly.
Causes of folate deficiency:
| Category | Examples |
|---|---|
| Decreased intake | Poor diet, alcoholism (most common in high-resource countries), infancy |
| Impaired absorption | Malabsorption, intrinsic intestinal disease, anticonvulsants, oral contraceptives |
| Increased loss | Hemodialysis |
| Increased requirement | Pregnancy, infancy, disseminated cancer, markedly increased hematopoiesis |
| Impaired utilization | Folate antagonists (methotrexate, trimethoprim) |
Clinical features:
- Megaloblastic anemia (identical hematology to B12 deficiency)
- GI mucosal changes: sore tongue, glossitis
- NO neurologic manifestations (key distinguishing feature from B12 deficiency)
Diagnosis: ↓ serum folate, ↓ RBC folate, ↑ serum homocysteine, normal methylmalonate (distinguishes from B12 deficiency).
Critical: Folate supplementation corrects the anemia of B12 deficiency but does NOT prevent — and may worsen — the neurologic damage. Always exclude B12 deficiency before starting folate therapy.
6. Vitamin B12 (Cobalamin) Deficiency Anemia
Sources: Animal products (meat, fish, dairy, eggs). Heat-stable. Also synthesised by gut flora.
Body stores: Liver stores 2–5 mg — sufficient for 5–20 years. Clinical presentation therefore follows years of unrecognised malabsorption.
Absorption pathway:
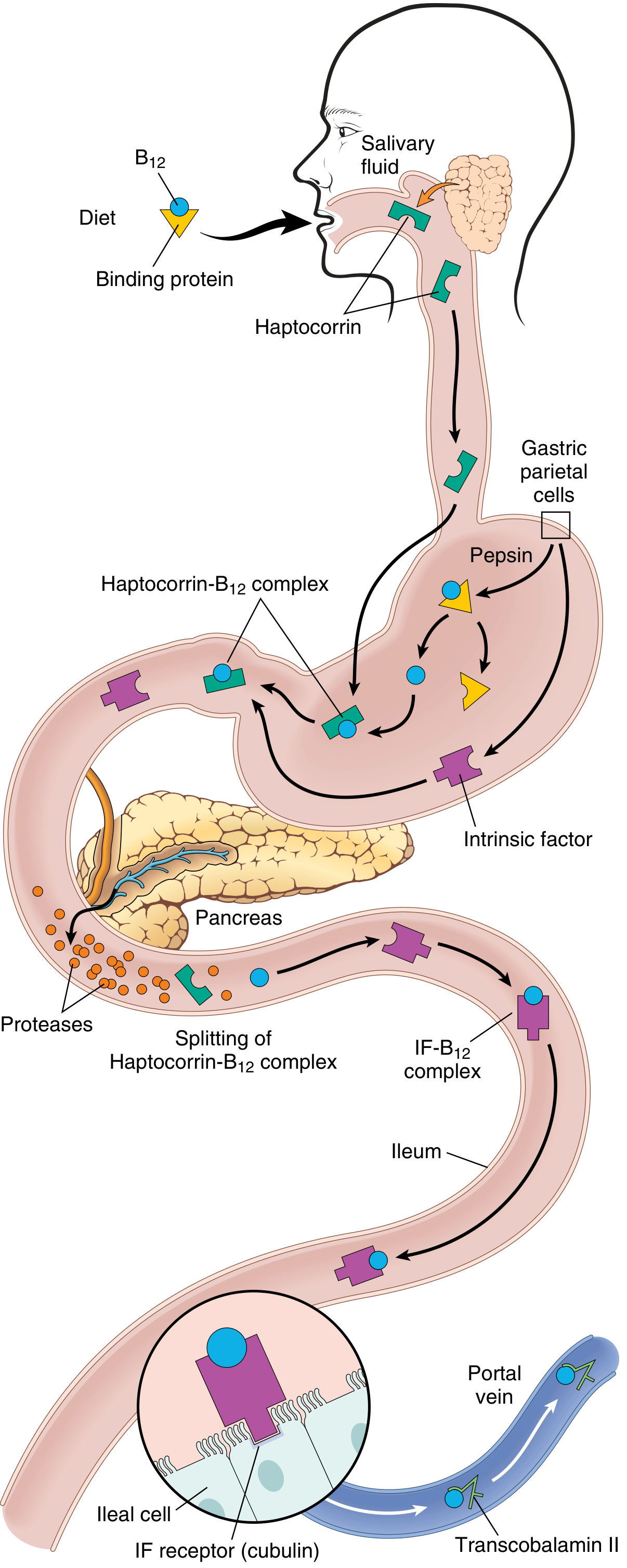
Fig. 10.12 — Vitamin B12 absorption: dietary B12 → stomach (freed by pepsin, binds haptocorrin) → duodenum (pancreatic proteases release B12, binds intrinsic factor) → terminal ileum (IF-B12 complex binds cubilin receptor on ileal enterocytes) → absorbed, bound to transcobalamin II → delivered to liver and bone marrow. (Robbins Basic Pathology)
Causes of B12 Deficiency
| Cause | Mechanism |
|---|---|
| Pernicious anemia (most common) | Autoimmune atrophic gastritis → loss of parietal cells → absent intrinsic factor. Serum autoantibodies to IF (diagnostic but not primary pathogen). |
| Gastrectomy | Loss of IF-producing cells |
| Ileal resection / Crohn disease / Whipple disease | Loss of IF-B12 absorbing cells |
| Blind loop / diverticulosis | Bacterial overgrowth → competitive uptake |
| Fish tapeworm (Diphyllobothrium) | Competitive parasitic uptake |
| Gastric atrophy / achlorhydria | Cannot release B12 from food-bound form (especially elderly) |
| Strict veganism | Only cause of dietary B12 deficiency |
Why B12 Deficiency Causes Neurologic Damage (and Folate Deficiency Does Not)
Vitamin B12 has two unique metabolic roles:
- Methylation of homocysteine → methionine (requires methylcobalamin; regenerates tetrahydrofolate → thymidine synthesis)
- Isomerisation of methylmalonyl-CoA → succinyl-CoA (requires adenosylcobalamin)
Folate deficiency only affects role #1 (thymidine synthesis). B12 deficiency affects both. Defective methylmalonyl-CoA conversion accumulates methylmalonic acid — which disrupts myelin synthesis in neuronal cells.
Neurologic lesion — Subacute Combined Degeneration (SCD):
- Demyelination of posterior columns (dorsal) — loss of vibration sense, proprioception
- Demyelination of lateral columns (corticospinal tracts) — spastic weakness, hyperreflexia
- Peripheral neuropathy — symmetric tingling, numbness, burning in feet/hands
- Neurologic damage may be irreversible even after B12 treatment
Clinical Features of Pernicious Anemia
| Feature | Detail |
|---|---|
| Anemia | Pallor, fatigue, dyspnea, palpitations |
| Mild jaundice | Ineffective erythropoiesis → intramedullar haemolysis |
| Glossitis | "Beefy red tongue" — megaloblastic changes in oral mucosa |
| Neurologic | SCD: symmetric paraesthesias → unsteady gait → loss of position sense |
| Gastric | Autoimmune atrophic gastritis; increased risk of gastric carcinoma |
Diagnosis of B12 Deficiency
| Finding | Result |
|---|---|
| Serum B12 | ↓ |
| Serum folate | Normal or ↑ |
| Serum homocysteine | ↑ |
| Serum methylmalonate | ↑ (unique to B12 deficiency) |
| Blood smear | Macro-ovalocytes, hypersegmented neutrophils |
| Bone marrow | Hypercellular, megaloblastic changes |
| Anti-intrinsic factor antibodies | Present in pernicious anemia (specific) |
Response to treatment: Reticulocytosis in 2–3 days after parenteral B12; anemia resolves over weeks. Neurologic deficits often persist or are only partially reversible.
Differentiating B12 from Folate Deficiency
| Feature | B12 Deficiency | Folate Deficiency |
|---|---|---|
| Serum B12 | ↓ | Normal |
| Serum folate | Normal or ↑ | ↓ |
| RBC folate | Normal | ↓ |
| Homocysteine | ↑ | ↑ |
| Methylmalonate | ↑ | Normal |
| Neurologic deficits | YES (SCD) | NO |
| Onset | Slow (years) | Faster (months) |
Non-Megaloblastic Macrocytosis
Macrocytic anemia without hypersegmented neutrophils or megaloblastic marrow changes. Mechanisms differ:
| Cause | Mechanism |
|---|---|
| Liver disease / alcoholism | Lipid membrane abnormalities → ↑ RBC membrane surface area → macrocytes (also folate deficiency component) |
| Hypothyroidism | Slowed RBC maturation |
| Drugs (hydroxyurea, AZT, azathioprine) | Inhibit DNA synthesis but without classic megaloblastic morphology |
| Reticulocytosis | Reticulocytes are larger → MCV rises after hemolysis/hemorrhage treatment |
| MDS | Dyserythropoiesis |
Master Summary Table
| Feature | IDA | ACD | Thalassemia Minor | Sideroblastic | Folate Def | B12 Def |
|---|---|---|---|---|---|---|
| MCV | ↓ | Normal/↓ | ↓ | ↓ (inherited); variable (acquired) | ↑ | ↑ |
| Serum iron | ↓ | ↓ | Normal | ↑ | Normal | Normal |
| TIBC | ↑ | ↓ | Normal | Normal | Normal | Normal |
| Ferritin | ↓ | ↑ | Normal/↑ | ↑ | Normal | Normal |
| Transferrin sat | ↓ | ↓ | Normal | ↑ | Normal | Normal |
| Homocysteine | — | — | — | — | ↑ | ↑ |
| Methylmalonate | — | — | — | — | Normal | ↑ |
| Smear hallmark | Microcytic, hypochromic | Mild hypo/normo | Target cells, microcytic | Dimorphic, ring sideroblasts on BM | Macro-ovalocytes, hyperseg PMN | Macro-ovalocytes, hyperseg PMN |
| Neuro deficits | No | No | No | No (unless Cu deficiency) | No | YES (SCD) |
| BM finding | ↑ cellularity, no iron stores | ↑ iron in macrophages | Hyperplastic erythropoiesis | Ringed sideroblasts | Megaloblastic | Megaloblastic |
Sources: Robbins & Kumar Basic Pathology (ISBN 9780323790185), Ch. 10, pp. 393–397; Robbins, Cotran & Kumar Pathologic Basis of Disease (ISBN 9780443264528), Ch. 14, pp. 608–616; Goldman-Cecil Medicine, Ch. 145.
This is a shared conversation. Sign in to Orris to start your own chat.