Chronic ischemic changes in bilateral corona radiata
Reading File
Finding Sources
Finding Sources
Searching Images
chronic ischemic changes bilateral corona radiata MRI white matter
Reading File
Finding Sources
Loading Image
Reading File
Reading File
Here is a comprehensive overview of chronic ischemic changes in the bilateral corona radiata:
What Are They?
Chronic ischemic changes in the bilateral corona radiata — also called leukoaraiosis, microvascular ischemic changes, or chronic small vessel disease (SVD) — refer to areas of white matter injury within the corona radiata resulting from cumulative small-vessel infarction. They appear on imaging as diffuse or patchy T2/FLAIR hyperintensities in the deep periventricular and subcortical white matter.
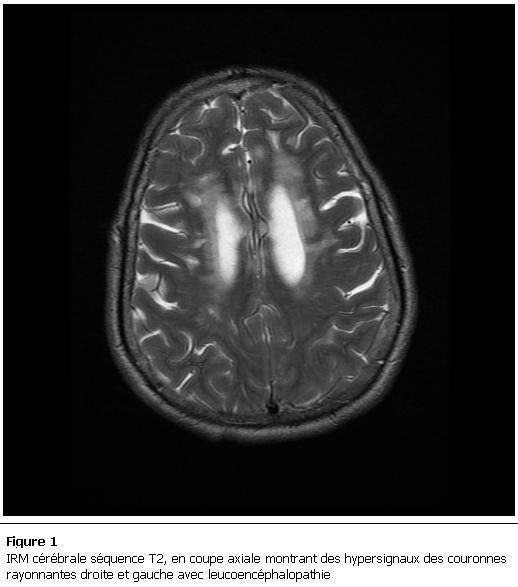
Anatomy
The corona radiata is a fan-shaped white matter tract that carries all motor and sensory fibers between the cerebral cortex and the internal capsule (and beyond). It receives its blood supply from small penetrating arteries (<200 µm diameter), making it vulnerable to small vessel disease.
Pathophysiology
The core mechanism is lipohyalinosis (and arteriosclerosis) of small penetrating arteries supplying the deep white matter — driven most commonly by chronic hypertension. The vessel walls thicken and narrow, leading to:
- Reduced perfusion → chronic ischemia → demyelination + gliosis
- Focal infarctions (lacunes) over time
- Axonal loss contributing to tract dysfunction
The vessel lumens may also show amyloid deposits in their walls. — Bradley and Daroff's Neurology in Clinical Practice
Imaging Characteristics
| Feature | Appearance |
|---|---|
| CT | Hypodense areas in periventricular white matter / corona radiata |
| MRI T2/FLAIR | Hyperintense (bright) — much more sensitive than CT |
| MRI T1 | May be hypointense (T1 "black holes" correlate with more severe damage) |
| DWI | No restricted diffusion (distinguishes chronic from acute) |
| Distribution | Periventricular, corona radiata, deep white matter; U-fibers typically spared |
| Morphology | Patchy, scattered, or confluent; no mass effect, no edema |
| Change over time | Very slow progression; minimal change on scans separated by several years |
On MRI, >1 white matter hyperintensity per decade of life is considered pathological. — Bradley and Daroff's Neurology in Clinical Practice
Risk Factors
- Hypertension (strongest modifiable risk factor)
- Diabetes mellitus
- Hypercholesterolemia
- Smoking
- Advanced age (incidence increases after age 40)
Clinical Consequences
Bilateral corona radiata involvement disrupts corticospinal, corticobulbar, and thalamocortical connections. Clinical manifestations depend on lesion burden and location:
| Domain | Manifestation |
|---|---|
| Cognition | Subcortical dementia, slowing of processing speed, executive dysfunction |
| Gait | Frontal/magnetic gait disorder, small shuffling steps |
| Motor | Mild pyramidal signs (brisk reflexes, spasticity) — less severe than capsular lesions |
| Bladder | Urgency incontinence (due to white matter disconnection) |
| Mood | Depression ("vascular depression" hypothesis — disruption of frontolimbic white matter) |
Periventricular white matter disease may lead to subcortical (vascular) dementia, which may be delayed with antihypertensive therapy. — Harrison's Principles of Internal Medicine, 22e
Differential Diagnosis of Bilateral White Matter Hyperintensities
| Condition | Distinguishing Features |
|---|---|
| Chronic SVD / leukoaraiosis | Age-related, vascular risk factors, symmetric, spares U-fibers |
| CADASIL | Anterior temporal pole + external capsule involvement; autosomal dominant (Notch-3 mutation); migraine with aura |
| CARASIL | Recessive; alopecia + spondylosis + early-onset dementia |
| Multiple sclerosis | Younger patients, perpendicular to ventricles (Dawson's fingers), infratentorial/spinal lesions |
| Vasculitis | Headache, CSF pleocytosis, enhancing lesions |
| PRES | Occipital predominance, hypertensive crisis, reversible |
Management
There is no treatment that reverses existing lesions, but progression can be slowed:
- Aggressive vascular risk factor control — especially hypertension (target BP <130/80 mmHg) and diabetes
- Antiplatelet therapy — if associated lacunar infarcts are present
- Statin therapy — for hyperlipidemia
- Lifestyle modification — smoking cessation, exercise
- Cognitive monitoring — neuropsychological assessment for early vascular cognitive impairment
If lesion burden is high and cognitive/gait symptoms are present, neurological follow-up with repeat MRI in 1–2 years is reasonable to assess progression.
Sources: Harrison's Principles of Internal Medicine 22e; Bradley and Daroff's Neurology in Clinical Practice; Adams and Victor's Principles of Neurology, 12e; Localization in Clinical Neurology, 8e
This is a shared conversation. Sign in to Orris to start your own chat.