Pyogenic liver abscess, Amoebic liver abscess .Clinical, course, diagnosis, differential diagnosis of methods of surgical treatment. Complications of parasitic diseases of the liver
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information to construct a thorough answer. Let me compile it.
Liver Abscess & Parasitic Liver Disease: Clinical Review
I. PYOGENIC LIVER ABSCESS (PLA)
Pathophysiology & Etiology
PLAs arise from five major routes:
- Biliary obstruction (cholelithiasis, malignancy, intrahepatic cholangiopathy post-transplant) — now the most frequent cause
- Portal vein pyemia from ascending enteric infection (appendicitis, diverticulitis)
- Trauma (blunt or penetrating)
- Hematogenous seeding via the hepatic artery from systemic bacteremia
- Cryptogenic (no identifiable source — frequently associated with Klebsiella pneumoniae)
Risk factors: age, diabetes mellitus, biliary malignancy or instrumentation, immunosuppression, liver transplantation, proton pump inhibitor use (loss of gastric acid barrier). Cryptogenic PLA has an association with occult colorectal malignancy — colonoscopy may be warranted.
Microbiology: Mostly polymicrobial. Biliary-source PLA: E. coli, Klebsiella spp., Enterococcus. Enteric-source PLA: Bacteroides fragilis and other anaerobes. Gram-negative organisms predominate in Europe and Asia; Streptococcus spp. predominate in US studies. Cryptogenic PLA is typically monomicrobial (K. pneumoniae).
Clinical Presentation
| Feature | Frequency |
|---|---|
| Fever | ~90% |
| Leukocytosis | ~90% |
| Elevated alkaline phosphatase | ~80% |
| Abdominal pain (RUQ) | ~60% |
| Hyperbilirubinemia | ~50% |
| Transaminitis | ~50% |
Non-specific symptoms (malaise, anorexia, weight loss, nausea) are common. Jaundice suggests biliary tract obstruction. Radiation of pain to the right shoulder indicates diaphragmatic irritation. Left untreated, mortality approaches 100%. Rupture into the peritoneum carries very high mortality without surgical intervention.
Diagnosis
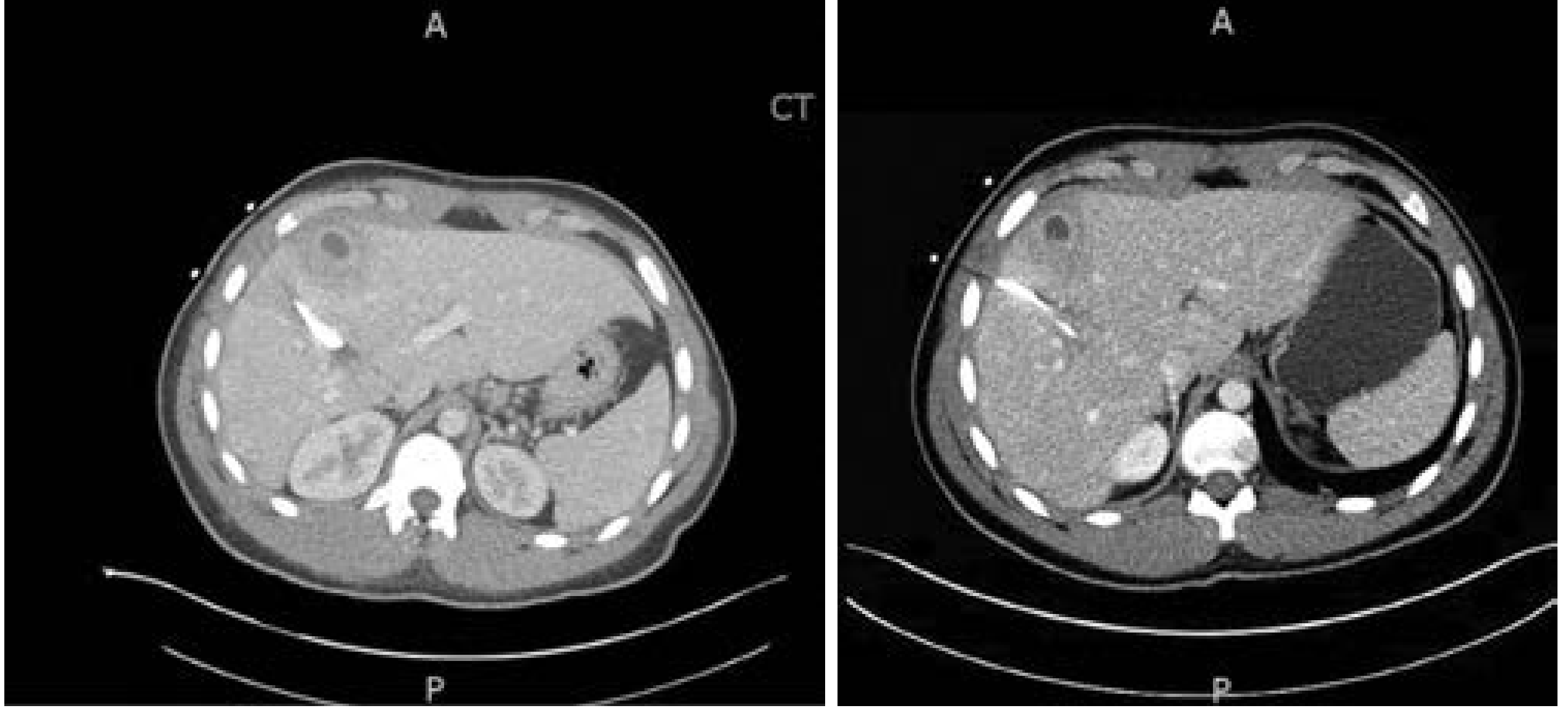
- CT with IV contrast — most sensitive; abscesses appear hypoattenuated with possible loculations and peripheral rim enhancement (uncommon but specific for abscess)
- Ultrasound — less sensitive; preferred initial test in pediatrics; abscesses appear hypoechoic with possible internal echoes
- Laboratory: leukocytosis, elevated ALP, elevated bilirubin, transaminitis
- Blood cultures (positive in >50% of cases) and abscess cultures are essential
- Local inflammation can cause portal or hepatic vein thrombosis → portal hypertension or Budd-Chiari syndrome
Treatment
Antibiotics
- Broad-spectrum empiric coverage targeting aerobic enteric gram-negatives (E. coli, Klebsiella), gram-positives (streptococci), and enteric anaerobes
- Post-transplant patients: consider carbapenems for ESBL coverage
- Duration: guided by clinical response + abscess resolution on follow-up imaging at 4–6 weeks
Percutaneous Drainage (standard of care)
- Recommended for any abscess >5 cm — superior outcomes vs needle aspiration alone
- Very large abscesses >10 cm may require multiple catheters but percutaneous drainage remains first-line
- Abscesses <3 cm: aspiration if technically feasible to guide antibiotic treatment
- Catheters remain until output is low and clear for several days
- Watch for bile in drain fluid (suggests biliary communication) → investigate with HIDA or MRCP/ERCP; may require sphincterotomy or percutaneous biliary drain
Surgical Drainage or Resection
- Reserved for cases with abscess rupture, concurrent intraabdominal abscess, or failure of minimally invasive management
- Open drainage carries higher morbidity than percutaneous approach
Prognosis: Mortality remains 4–10%. Independent risk factors for increased mortality: diabetes mellitus, underlying malignancy, cirrhosis, and large abscess size (>10 cm associated with pleural effusion and rupture risk).
II. AMOEBIC LIVER ABSCESS (ALA)
Pathophysiology
Caused by the protozoan Entamoeba histolytica. Transmission is fecal-oral via mature cysts in contaminated food or water → colitis → trophozoites invade intestinal mucosa → seed the liver via the portal circulation. This typically occurs 2–4 months after symptoms of colonic amebiasis.
- Abscesses contain necrotic hepatocytes and trophozoites in thick, proteinaceous brown fluid — classically described as "anchovy paste"
- Rates of ALA are less than 1% of symptomatic enteric amebiasis
Demographics: In the US, predominantly male patients (8:1 male:female ratio) with a history of travel to an endemic area (Central/South America, Africa, South Asia).
Clinical Presentation
Symptoms are similar to PLA and cannot be used to reliably differentiate the two entities:
- Fever (~80%), RUQ pain (~60%), leukocytosis, elevated ALP/bilirubin/transaminases
- Eosinophilia is not commonly seen despite parasitic cause
- Travel history and remote antecedent diarrheal illness are critical clues
Natural course: Without treatment, abscess enlarges and ruptures into peritoneal cavity or chest. Rupture into the right pleural space is uncommon (diaphragm fusion from inflammation). Rupture into the pericardium is rare but historically severe.
Diagnosis
- Serology (antibody or antigen): highly sensitive, essential for diagnosis
- Antigen testing is more specific than antibody serology
- ~⅓ of patients with prior E. histolytica infection retain antibodies without active disease
- >90% of patients with ALA have antibodies to E. histolytica in high titer
- Imaging (CT/US): cannot differentiate ALA from PLA
- Multiple abscesses suggest pyogenic infection; ALA tends to be solitary
- Aspiration: "anchovy paste" fluid; finding cysts/trophozoites in stool supports diagnosis
Differential Diagnosis: ALA vs PLA
| Feature | Pyogenic (PLA) | Amoebic (ALA) |
|---|---|---|
| Epidemiology | Any patient; biliary disease, immunosuppression | Travel to endemic area; young male |
| Number of abscesses | Often multiple | Usually solitary |
| Microbiology | Polymicrobial bacteria | E. histolytica |
| Serology | Negative | Anti-amoebic antibodies >90% |
| Aspiration fluid | Pus | "Anchovy paste" |
| Response to metronidazole | No | Yes (>90%) |
| Eosinophilia | No | No |
Differential Diagnosis (both types)
- Hepatic malignancy (necrotic center can mimic fluid-filled abscess)
- Cholangitis / cholecystitis
- Right lower lobe pneumonia
- Acute pancreatitis
- Perforated peptic ulcer
Treatment
Drug of choice: Metronidazole 500–750 mg orally three times daily for 7–10 days
- Clinical improvement (pain + fever reduction) in >90% of patients within 72 hours
- Longer-acting nitroimidazoles (tinidazole, ornidazole) effective as single-dose therapy
Mandatory luminal agent (to eradicate intestinal cysts and prevent recurrence/transmission):
- Paromomycin 10 mg/kg three times daily for 7 days, OR
- Diiodohydroxyquin (iodoquinol) 650 mg three times daily for 20 days
Aspiration / Drainage — indicated only when:
- Need to rule out pyogenic abscess (multiple lesions)
- No clinical response within 3–5 days
- Threat of imminent rupture
- Left-lobe abscess threatening pericardial rupture
- No evidence that aspiration accelerates healing, even for large (up to 10 cm) abscesses
- Percutaneous drainage can be successful even after rupture
Surgery reserved for: bowel perforation, rupture into the pericardium, failure of all other therapies.
Repeat imaging is recommended but abscess radiological resolution may take several months and is not required to stop antibiotics.
Prognosis: With early diagnosis and therapy, mortality from uncomplicated ALA is <1%.
III. PARASITIC LIVER DISEASES — COMPLICATIONS
Hydatid (Echinococcal) Cyst Disease
Caused by larvae of Echinococcus granulosus (cystic form, 55–80% in liver) or Echinococcus multilocularis (alveolar form — aggressive, "tumor-like," 99% hepatic).
Cyst structure: Two-layer wall — inner germinal layer (produces scolices and daughter cysts) + outer acellular pericyst (calcified in ~50%).
Complications:
- Rupture into the biliary tree (in ~⅓ of cases) — cholangitis, obstructive jaundice
- Rupture through the diaphragm into the pleural cavity
- Rupture into the peritoneum — anaphylaxis risk
- Secondary bacterial infection (pyogenic superinfection)
- Compression of hepatic vessels or bile ducts
Classification (WHO-IWGE, updated 2003): CL, CE1–CE5 based on ultrasound appearance and functional status (active, transitional, inactive). This guides treatment selection.
Treatment (surgical approach is primary):
- PAIR (Puncture-Aspiration-Injection-Re-aspiration) + albendazole for active CL/CE1/CE3 lesions; avoid for ALA due to anaphylaxis risk with echinococcal cysts
- Surgery + albendazole for complex, large, or complicated cysts
- Albendazole or mebendazole alone is less effective than combined with surgery
- Alveolar echinococcosis (E. multilocularis): 5-year mortality ~70% untreated; requires aggressive resection + long-term albendazole
ALA Complications
- Rupture into right pleural space, peritoneum, or pericardium
- Secondary bacterial superinfection
- Hepatobronchial fistula (rare)
- Portal hypertension (rare)
PLA Complications
- Rupture → purulent peritonitis (high mortality)
- Portal or hepatic vein thrombosis → portal hypertension, Budd-Chiari syndrome
- Pleural effusion / empyema (especially with abscesses >10 cm)
- Septicemia / septic shock
- Biliary fistula
Sources:
- Current Surgical Therapy, 14th ed. — pp. 440–443
- Yamada's Textbook of Gastroenterology, 7th ed. — pp. 2178–2181
- Harrison's Principles of Internal Medicine, 22nd ed. — p. 1801
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease
hydatid cyst
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material to write a thorough answer. Let me compile it.
Hydatid Cyst (Echinococcosis)
Etiology & Life Cycle
Hydatid disease (echinococcosis) is a zoonosis caused by the larval stage of the canine tapeworm Echinococcus granulosus (cystic form — most common) or Echinococcus multilocularis (alveolar form — rare, aggressive).
Life cycle:
- Definitive host: dogs (and other canids) — adult tapeworm lives attached to the ileum; sheds up to 500 ova per day into feces
- Intermediate hosts: sheep, goats, cattle — ingest ova from contaminated grass/farmland
- Humans: accidental intermediate hosts — dead-end host; no human-to-human transmission
After ingestion, gastric acid dissolves the chitinous envelope → the liberated oncosphere (containing hooklets) burrows through the intestinal mucosa → enters the portal vein → carried to the liver (70% of cysts form here). A few ova bypass the liver and lodge in the lungs (10–40%), or enter the systemic circulation (brain, bone, spleen, kidney).
Endemic areas: Mediterranean countries, Middle East, South America, Africa, Australia/New Zealand, Far East, Central Europe (for E. multilocularis: Russia, Turkey, China, Japan).
Cyst Structure & Pathology
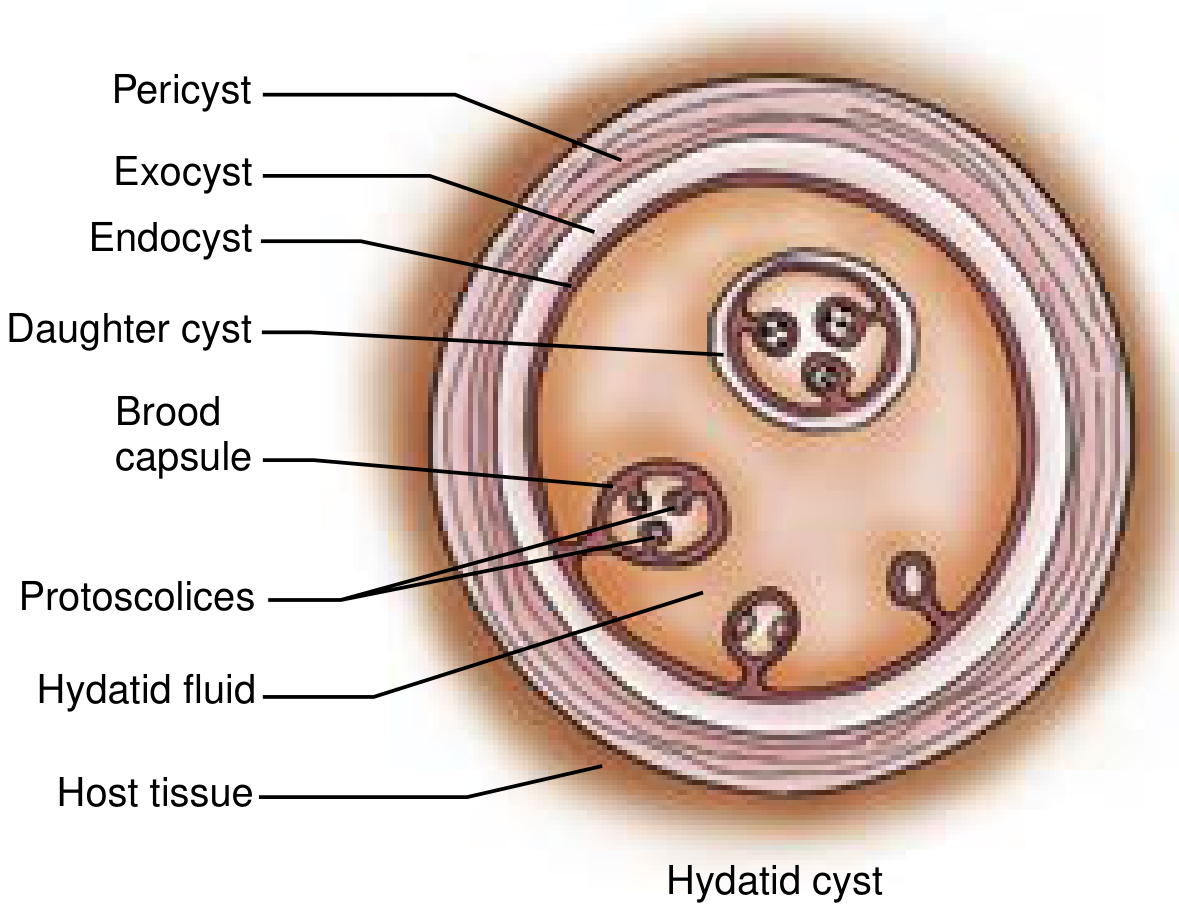
Three weeks post-infection, a visible cyst develops and slowly expands. The cyst has three distinct layers:
| Layer | Origin | Description |
|---|---|---|
| Pericyst (exocyst) | Host tissue reaction | Outer reactive fibrous capsule; calcified in ~50% of patients |
| Ectocyst | Parasite | Outer gelatinous (laminated) membrane |
| Endocyst (germinal layer) | Parasite | Inner single-cell germinal membrane — produces brood capsules, daughter cysts, and scolices |
- Hydatid sand: free-floating brood capsules and scolices in the cyst fluid
- Daughter cysts: true replicas of the mother cyst, developing from the germinal layer
- Calcification of the wall does not always imply the cyst is dead
- ~75% of cysts are located in the right lobe of the liver and are solitary
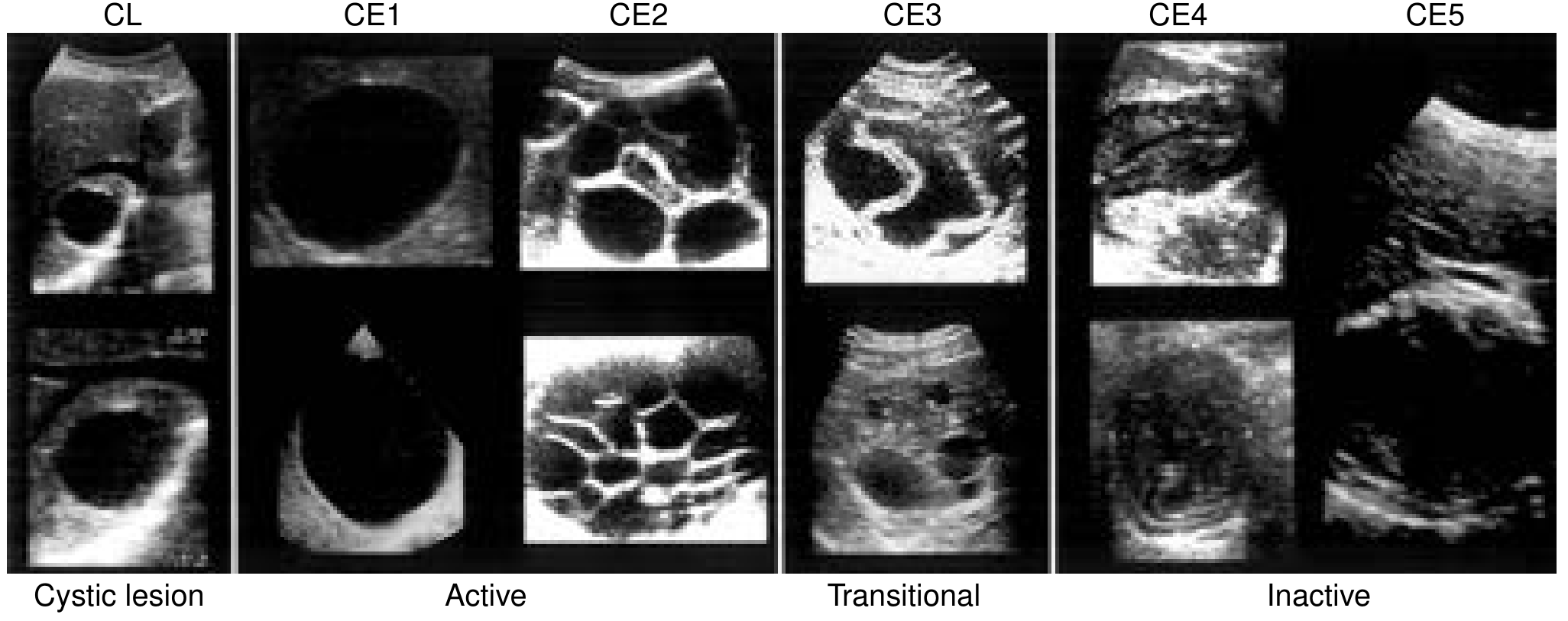
WHO-IWGE Classification (Ultrasound-Based)
| Cyst Type | Status | Ultrasound Features | Remarks | Treatment |
|---|---|---|---|---|
| CL | Active | Unilocular, anechoic; wall not visible | Early, not fertile | Needs diagnostic tests |
| CE1 (Gharbi I) | Active | Unilocular, anechoic; wall visible; hydatid sand ("snowflake") | Fertile — pathognomonic | <5 cm: ABZ alone; >5 cm: PAIR + ABZ |
| CE2 (Gharbi III) | Active | Multivesicular, septated; rosette/honeycomb | Fertile — pathognomonic | Percutaneous therapy or surgery + ABZ |
| CE3 (Gharbi II) | Transitional | Detached floating membrane; "water lily sign" | Degenerating — pathognomonic | PT, PAIR, or surgery + ABZ |
| CE4 (Gharbi IV) | Inactive | Heterogeneous, hypoechoic; "ball of wool" | Usually not fertile | Surgery + ABZ, or watchful waiting |
| CE5 (Gharbi V) | Inactive | Thick, calcified wall; arch-shaped with cone-shaped shadow | Usually not fertile; highly suggestive | Surgery + ABZ, or watchful waiting |
Clinical Presentation
Most patients have an extended asymptomatic period — cysts may be discovered incidentally on imaging. The clinical presentation depends on whether complications occur.
Uncomplicated disease:
- Dull RUQ pain / abdominal fullness
- Hepatomegaly
- Dyspepsia and nausea
- Abdominal distension
Complicated disease (presenting symptoms):
- Jaundice and cholangitis — rupture/communication with the biliary tree (occurs in up to ~⅓ of patients)
- Fever — bacterial superinfection mimicking a pyogenic abscess
- Productive cough / hemoptysis — rupture through the diaphragm into pleural cavity and lung
- Acute abdomen + anaphylactic shock — free rupture into the peritoneum (rare but life-threatening)
- Jaundice and fever each present in ~8% of patients overall
Diagnosis
Laboratory Tests
- Eosinophilia: ~30–40% of patients
- LFTs: normal or mildly elevated ALP/transaminases (unless biliary complication)
- ELISA (enzyme-linked immunosorbent assay): sensitivity 85–89% — has replaced older tests (Casoni skin test, complement fixation, indirect hemagglutination)
- Confirmatory: immunoblotting, antigen 5 and antigen B testing on hydatid cyst fluid
- Alveolar disease: Em2 antigen — 99% specificity for E. multilocularis; Em16/Em18 to distinguish active vs inactive
Note: Serology may be negative if the cyst has not leaked, contains no scolices, or if the parasite is no longer viable. Negative serology does not exclude disease.
Imaging
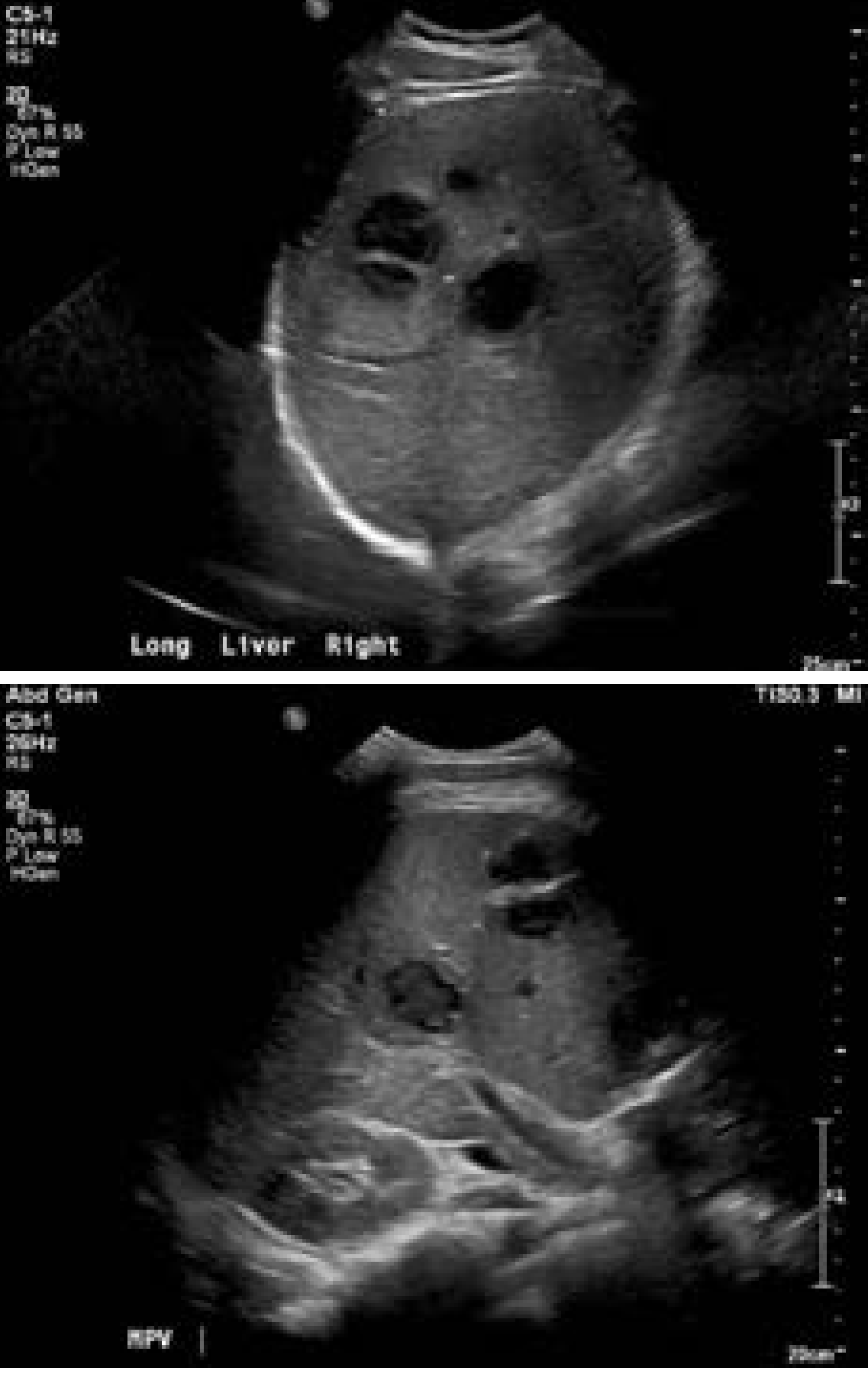
Ultrasound — first-line worldwide (low cost, 90% specificity):
- Simple cyst: well-circumscribed with budding signs; floating hyperechoic hydatid sand
- Daughter cysts: rosette/honeycomb appearance
- Calcifications: eggshell appearance — highly suggestive
- "Ball of wool" sign with degenerated membranes
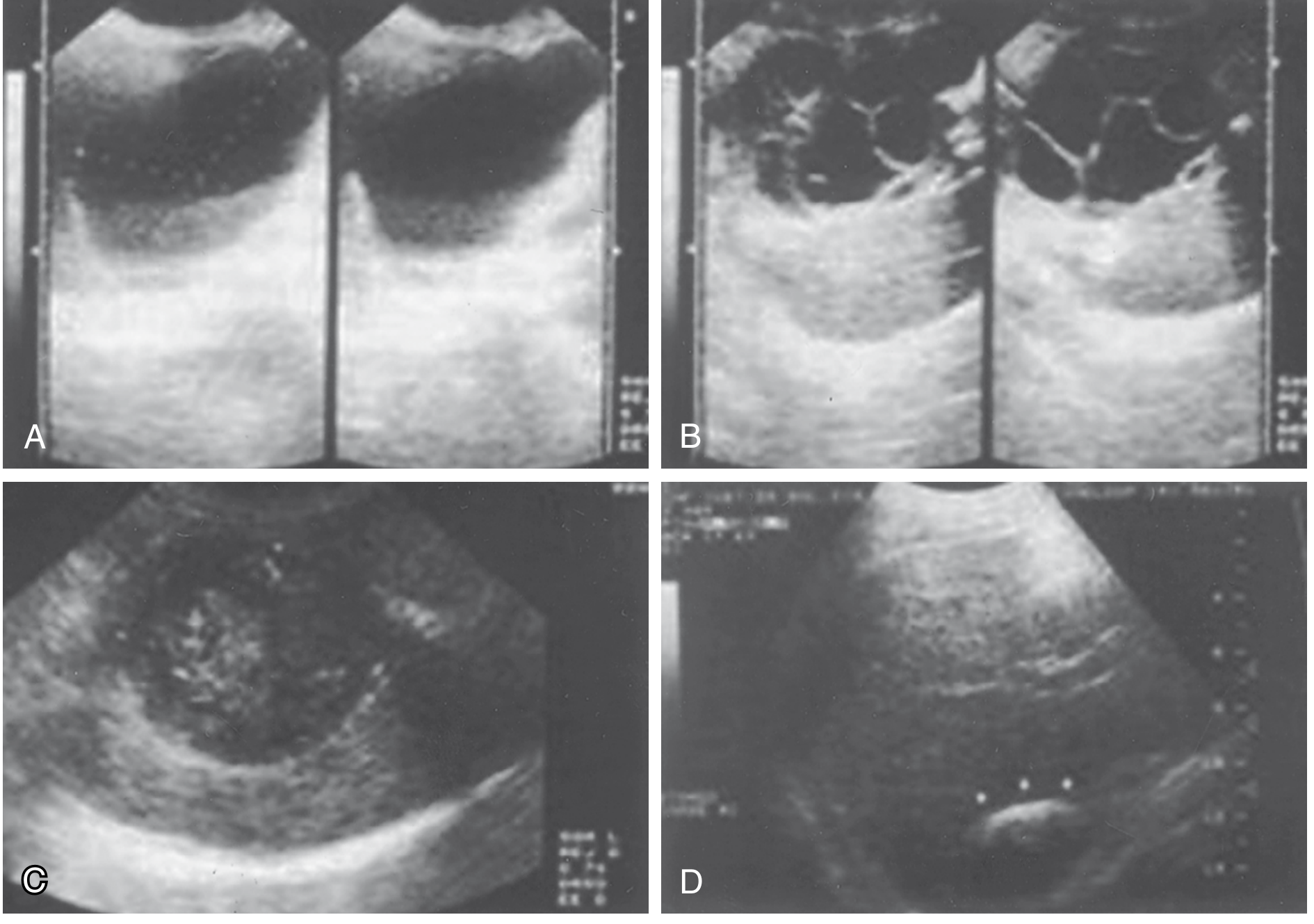
CT scan: more detail on location, depth, vascular/biliary relationships — essential for operative planning
MRI/MRCP: best for characterizing pericyst/cyst matrix and identifying cystobiliary fistulae
ERCP: biliary communication occurs in up to 25% of cases — ERCP delineates biliary anatomy, visualises fistulae, and allows biliary drainage before surgery
PET scan: useful for detecting metastatic lesions and active areas in alveolar echinococcosis
Differential Diagnosis
- Simple congenital hepatic cyst
- Amoebic abscess
- Pyogenic liver abscess
- Biliary cystadenoma / cystadenocarcinoma
- Hepatic metastasis with necrotic centre
- Polycystic liver disease
Treatment
Treatment aims to: eradicate the parasite, prevent secondary complications, prevent recurrence, and minimise morbidity.
Treatment Options Comparison
| Modality | Pros | Cons | Recurrence |
|---|---|---|---|
| Medical therapy alone | Non-invasive | Higher recurrence | 70–80% |
| PAIR + ABZ | Nonsurgical; fewer complications | Lower evidence base | ~10% |
| Open cyst evacuation | Definitive; lower risk than pericystectomy | Invasive; spillage risk | ~20% |
| Laparoscopic cyst evacuation | Shorter stay; faster recovery | Pneumoperitoneum increases spillage risk | ~20% |
| Pericystectomy | Less spillage; lower recurrence vs drainage | Bleeding/bile duct injury risk (20% major morbidity) | ~10% |
| Liver resection / transplantation | Lowest recurrence | Most invasive; 40% morbidity | ~10% |
1. Medical Management (Benzimidazoles)
Albendazole — drug of choice:
- Dose: 400 mg orally twice daily (≥60 kg); or 10–15 mg/kg/day divided twice daily (<60 kg), max 800 mg/day
- Better absorbed and concentrated in cyst fluid than mebendazole
- Duration: 3–6 months continuous (or perioperatively)
- Albendazole + praziquantel (25 mg/kg/day) is more effective than albendazole alone
- Side effects: neutropenia, hepatotoxicity, nausea, alopecia — monitor CBC and LFTs
- Monotherapy cure rate is low (cyst disappearance in well under 50%) — should be combined with procedural treatment
- Not indicated for inactive or densely calcified cysts (unless complicated)
2. PAIR (Percutaneous Aspiration-Injection-Reaspiration)
Used for carefully selected patients, especially high-risk operative candidates.
Procedure steps:
- Percutaneous puncture and aspiration under ultrasound guidance
- Aspiration of cyst contents
- Injection of scolicidal agent (20% hypertonic saline or absolute alcohol) for 10–30 minutes
- Re-aspiration and final irrigation with 0.9% saline
Indications: CE1/CE3 cysts >5 cm; patients refusing surgery; failed medical management; recurrence after surgery; infected cysts; high surgical risk
Contraindications: biliary fistulae, complicated cysts, inaccessible/superficial locations, CE4 or CE5 (inactive/calcified cysts), children <3 years
Albendazole must be given before PAIR and continued for up to 1 month after — combined PAIR + ABZ is superior to either alone.
3. Surgical Management
Indications for surgery:
- Large CE2–CE3 cysts with multiple daughter cysts
- Superficial cysts at risk of spontaneous or traumatic rupture
- Infected cysts or biliary communication (when PAIR unavailable)
- Mass effect on adjacent vital organs
- Failure of PAIR or medical management
Surgical principles (tenets):
- Complete inactivation of scolices with scolicidal agents
- Eliminate the parasite and viable cyst contents
- Prevent spillage and recurrence
- Manage the residual cyst cavity
- Pack the field with hypertonic saline–soaked sponges before opening
Scolicidal agents: 20% hypertonic saline is WHO-recommended (contact with germinal layer ≥15 minutes). Avoid in cystobiliary fistulae — risk of sclerosing cholangitis. Avoid formalin and cetrimide (unsafe).
Surgical techniques:
a) Open Cyst Evacuation (conservative, most common)
- Best for peripherally located cysts
- Aspiration → injection of scolicidal agent → unroofing → removal of daughter cysts → resection of active cyst lining → omentoplasty
- If biliary communication present: close bile duct openings with absorbable sutures; pack with omentum; external closed-suction drainage if fistula cannot be closed
b) Laparoscopic Cyst Evacuation
- Reduced hospital stay, shorter operating time, faster recovery
- Disadvantages: pneumoperitoneum increases spillage risk, limited manipulation, difficulty aspirating thick contents
- Conversion to open <5% in properly selected patients
c) Pericystectomy
- Complete resection of the cyst wall (open or closed technique)
- Closed pericystectomy: removes entire pericyst without entering cyst — lowest risk of spillage and recurrence
- Increased risk of bleeding and bile duct injury (~20% major morbidity)
- Best for peripheral cysts
d) Liver Resection / Transplantation
- For alveolar echinococcosis (E. multilocularis), massive or centrally located cysts
- Lowest recurrence (~10%) but highest morbidity (~40%)
- Transplantation reserved for end-stage alveolar disease with hepatic failure
Biliary complications at surgery: When bile duct communication is found, it must be meticulously repaired. Simple suture closure is often sufficient; major biliary reconstruction or postoperative ERCP may be needed.
Complications
| Complication | Details |
|---|---|
| Rupture into biliary tree | Most common (~⅓ of patients); cholangitis, obstructive jaundice, biliary colic |
| Rupture into peritoneum | Anaphylactic shock; disseminated echinococcosis (secondary implantation of scolices) |
| Rupture into pleural cavity | Via diaphragmatic erosion; hydatid empyema, bronchopleural fistula |
| Pulmonary rupture | Productive cough; expectoration of hydatid material ("grape skin") |
| Secondary bacterial infection | Pyogenic superinfection — mimics pyogenic abscess |
| Pericardial rupture | Very rare; cardiac tamponade |
| Compression | Portal hypertension, Budd-Chiari syndrome, IVC compression |
| Bone involvement | Pathological fractures (rare) |
| Anaphylaxis | During spontaneous or iatrogenic rupture |
Alveolar Echinococcosis (E. multilocularis)
A distinct and far more aggressive disease:
- Exclusively hepatic in 99% of cases
- Behaves like a malignant infiltrating tumour — no outer fibrous capsule, extends through liver parenchyma
- 5-year mortality ~70% untreated; 10-year mortality ~94%
- Requires radical surgical resection + long-term albendazole
- PNM staging system (analogous to TNM): P = parasite location/size in liver; N = neighbouring organ involvement; M = distant metastases (M0 vs M1)
- PET scan useful to identify active areas
Sources:
- Current Surgical Therapy, 14th ed. — pp. 398–404
- Sabiston Textbook of Surgery, pp. 1891–1892
- Schwartz's Principles of Surgery, 11th ed. — p. 1551
prevention
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
The textbooks in this library do not contain dedicated prevention chapters for these conditions beyond what was already retrieved in prior sessions. Based on all material already in context (Harrison's, Current Surgical Therapy, Yamada's, Sabiston), here is a complete prevention overview:
Prevention of Liver Abscess & Hydatid Cyst Disease
I. Prevention of Amoebic Liver Abscess (ALA)
Prevention targets Entamoeba histolytica transmission, which is exclusively fecal-oral.
Sanitation & Water Safety
- Provision of safe drinking water and proper sewage disposal is the single most effective public health measure
- E. histolytica cysts are resistant to standard chlorination — disinfection by iodination (tetraglycine hydroperiodide tablets) is recommended in high-risk settings
- Use bottled water when travelling to endemic areas (Central/South America, Africa, South Asia)
- Avoid unpeeled raw fruits and vegetables in endemic regions (cysts survive on surfaces)
- Avoid ice made from potentially contaminated water
Personal Hygiene
- Strict handwashing with soap and water — especially after defecation and before food handling
- Food handlers with known E. histolytica infection should be excluded from work until cyst-free
Eradication of Cyst Carriage
- An asymptomatic carrier can excrete up to 15 million cysts per day
- Treatment of asymptomatic cyst passers with a luminal agent (paromomycin or iodoquinol) reduces community transmission
- All patients treated for ALA must also receive a luminal agent to eradicate intestinal cysts and prevent further transmission and relapse
No Vaccine
- There is currently no effective prophylactic vaccine against E. histolytica
II. Prevention of Pyogenic Liver Abscess (PLA)
PLA is not a communicable disease — prevention focuses on managing underlying conditions and minimising modifiable risk factors.
Prompt Treatment of Source Infections
- Early diagnosis and treatment of biliary disease (cholelithiasis, cholangitis, cholecystitis) before progression to liver abscess
- Timely management of appendicitis and diverticulitis — before portal pyaemia develops
- Adequate treatment of periodontal / dental infections (haematogenous seeding)
- Effective management of inflammatory bowel disease and colorectal malignancy
Peri-procedural Measures
- Appropriate antibiotic prophylaxis before biliary instrumentation (ERCP, percutaneous biliary drainage, stent placement)
- Sterile technique during biliary and hepatic procedures
- Prompt biliary decompression when obstruction is identified
Risk Factor Control
- Tight glycaemic control in diabetic patients — diabetes is an independent risk factor
- Consideration of cryptogenic PLA: colonoscopy to exclude occult colorectal malignancy
- Caution with proton pump inhibitor use in patients with other risk factors — loss of gastric acid barrier may facilitate bacterial colonisation of the biliary tree
- Post-liver transplant patients: routine antifungal prophylaxis reduces fungal hepatic abscess risk
III. Prevention of Hydatid Cyst Disease (Echinococcosis)
Prevention of hydatid disease requires breaking the dog–livestock–human transmission cycle at multiple levels. This is fundamentally a One Health problem requiring coordinated veterinary and public health action.
Primary Prevention (Interrupting the Life Cycle)
| Target | Intervention |
|---|---|
| Dogs (definitive host) | Regular deworming with praziquantel (eliminates adult Echinococcus from the intestine); restrict dog access to slaughterhouses and offal |
| Livestock (intermediate host) | Proper disposal or destruction of infected animal offal — do not feed raw viscera to dogs; condemn and incinerate infected carcasses at abattoirs |
| Slaughterhouse control | Veterinary inspection of all slaughtered animals; hygienic disposal of condemned organs |
| Human exposure | Avoid close contact with dogs in endemic areas; wash hands thoroughly after handling dogs or contaminated soil |
Public Health & Community Measures
- Mass dog treatment programmes with praziquantel in endemic communities — the most effective population-level intervention
- Health education in endemic communities: explain the dog–sheep–human cycle and discourage feeding of raw offal to dogs
- Stray dog population control
- Surveillance and reporting of human cases to track endemicity
Personal Precautions (for Travellers / Exposed Individuals)
- Wash hands after contact with dogs, dog faeces, or contaminated soil
- Do not consume unwashed raw vegetables or herbs from areas where dogs defecate
- Drink safe water in endemic regions
Vaccination of Livestock
- EG95 vaccine for sheep (E. granulosus) — proven effective in reducing prevalence in intermediate hosts; used in national control programmes in New Zealand, Australia, and parts of South America. Vaccination of the main intermediate host (sheep) combined with dog deworming has achieved significant reductions in transmission in controlled programmes.
No Human Vaccine
- No licensed vaccine exists for humans. Prevention relies entirely on environmental and veterinary control.
Summary Table
| Disease | Key Transmission Route | Primary Prevention Strategy |
|---|---|---|
| Amoebic liver abscess | Fecal-oral (water/food contamination) | Safe water, sanitation, handwashing, iodination of water, treat cyst carriers |
| Pyogenic liver abscess | Endogenous (biliary, portal, haematogenous) | Treat biliary disease early, antibiotic prophylaxis, glycaemic control |
| Hydatid cyst | Fecal-oral (dog ova in contaminated environment) | Dog deworming (praziquantel), destroy infected offal, livestock vaccination (EG95), hygiene |
Sources: Harrison's Principles of Internal Medicine 22e; Current Surgical Therapy 14e; Yamada's Textbook of Gastroenterology 7e; Sabiston Textbook of Surgery
This is a shared conversation. Sign in to Orris to start your own chat.