Acute inflammation of the middle ear. Mastoiditis. Chronic inflammation of the middle ear (mesotympanitis. epitympanitis)
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
I now have sufficient information to compose a comprehensive answer covering all three topics. Let me synthesize:
Ear Infections: Acute Otitis Media, Mastoiditis, and Chronic Otitis Media
I. Acute Otitis Media (AOM)
Definition & Epidemiology
AOM is a bacterial/viral infection of the middle ear characterized by the presence of a middle ear effusion with signs and symptoms of acute infection. It is the most common bacterial disease of childhood (80% of children aged 1–6 years experience at least one episode) and the leading driver of pediatric antibiotic prescriptions in the United States. Peak incidence is between 6–15 months of age.
Risk factors: daycare attendance, male sex, parental smoking, pacifier/bottle use, family history of recurrent OM, cleft palate, Down syndrome. Breastfeeding is protective.
— Rosen's Emergency Medicine, p. 908
Pathophysiology
The fundamental mechanism is Eustachian tube dysfunction:
- Viral URI causes Eustachian tube inflammation → mechanical or functional obstruction
- Negative middle ear pressure develops → fluid accumulates
- Nasopharyngeal bacteria enter the middle ear cavity → inflammation, pus, increased pressure
AOM is commonly a co-infection: viruses are isolated from middle ear aspirates in ~70% of cases.
Common bacterial pathogens:
- Streptococcus pneumoniae (most virulent; commonest cause)
- Haemophilus influenzae (non-typeable; increasing in frequency post-pneumococcal vaccination)
- Moraxella catarrhalis
- Rarer: S. pyogenes, S. aureus, Mycobacterium tuberculosis, gram-negatives
- In neonates: additionally Group B Streptococcus and gram-negatives
— Rosen's Emergency Medicine, p. 908–909
Clinical Features
- Otalgia (most important symptom — unilateral or bilateral)
- Fever, irritability, pulling at ears (non-specific in children)
- Otoscopy: erythematous, bulging tympanic membrane (TM) with decreased mobility (by pneumatic otoscopy) — the hallmark finding
- Bullous myringitis (bullae on TM) may be seen; treatment is the same as standard AOM
- Middle ear effusion present
- Conductive hearing loss
Examination of the TM is essential — no constellation of symptoms alone reliably predicts AOM.
Management
"Watch and wait" (observation): Appropriate for selected children ≥2 years with mild symptoms for 2–3 days before prescribing antibiotics. Not validated in adults (antibiotics recommended for all adults).
Antibiotic therapy:
| Scenario | First-line |
|---|---|
| Standard AOM, no PCN allergy | Amoxicillin 80–90 mg/kg/day ÷ BID |
| AOM within 30 days / concurrent conjunctivitis / β-lactamase coverage needed | Amoxicillin-clavulanate 90 mg/kg/day |
| Non-severe PCN allergy | Cefdinir, cefuroxime, cefpodoxime, or ceftriaxone IM |
| Severe PCN allergy | Azithromycin or clindamycin (less coverage) |
| Treatment failure at 3 days | Amoxicillin-clavulanate or IM ceftriaxone × 3 days |
Duration: 10 days for children <2 years, TM perforation, or chronic/recurrent disease; shorter courses acceptable in mild cases in older children.
Analgesia: NSAIDs (ibuprofen) or acetaminophen — antibiotics do NOT provide pain relief in the first 24 hours.
AOM with perforation: Treat identically to AOM without perforation; small perforations typically heal spontaneously.
AOM with tympanostomy tubes: Ototopical fluoroquinolone drops (e.g., ofloxacin, ciprofloxacin/dexamethasone) — not aminoglycosides (ototoxic with non-intact TM).
— Rosen's Emergency Medicine, p. 909–910
Complications of AOM
- Persistent otitis media with effusion (OME) — leading cause of childhood hearing loss
- Acute mastoiditis (most common suppurative complication)
- TM perforation, ossicular erosion, tympanosclerosis
- Petrous apicitis (Gradenigo's syndrome)
- Facial nerve palsy
- Labyrinthitis
- Intracranial extension (meningitis, brain abscess, lateral sinus thrombosis)
— Cummings Otolaryngology, p. 2681–2682
II. Mastoiditis
Definition & Pathogenesis
Mastoiditis is inflammation of the mastoid air cells and the most common suppurative complication of AOM, though its incidence has fallen dramatically with antibiotic use. It may occur with or without a known preceding AOM episode.
Anatomic basis: The middle ear communicates with the mastoid air cells via the aditus ad antrum — a narrow passage. If this becomes obstructed:
- Pus accumulates in a closed space
- Infection spreads via venous channels → periosteal inflammation
- Destruction of bony trabeculae → coalescent mastoiditis (acute mastoid osteitis)
- Subperiosteal abscess may form, pushing the auricle anteroinferiorly
Pathogens:
- Streptococcus pneumoniae (leading cause)
- Group A Streptococcus
- P. aeruginosa, S. aureus, Fusobacterium necrophorum (particularly after prior antibiotic use)
— Rosen's Emergency Medicine, p. 910–912
Clinical Features
- Fever, headache, otalgia, hearing loss
- Postauricular erythema, edema, and tenderness (classic)
- Protrusion (displacement) of the auricle anteroinferiorly — highly characteristic
- Abnormal TM (erythema, bulging) — may be normal in ~10% of cases
- Symptoms of AOM lasting >2 weeks should raise suspicion
Differential Diagnosis
- Severe AOM / otitis externa
- Skull fracture
- Posterior auricular lymphadenitis
- Deep space neck infection (Box 58.4)
Investigations
- Clinical diagnosis is possible with classic findings
- CT scan of temporal bones: indicated when neurologic symptoms suggest intracranial spread, or failure to improve with conservative therapy. Shows opacification of mastoid air cells, bony destruction (coalescence), subperiosteal abscess
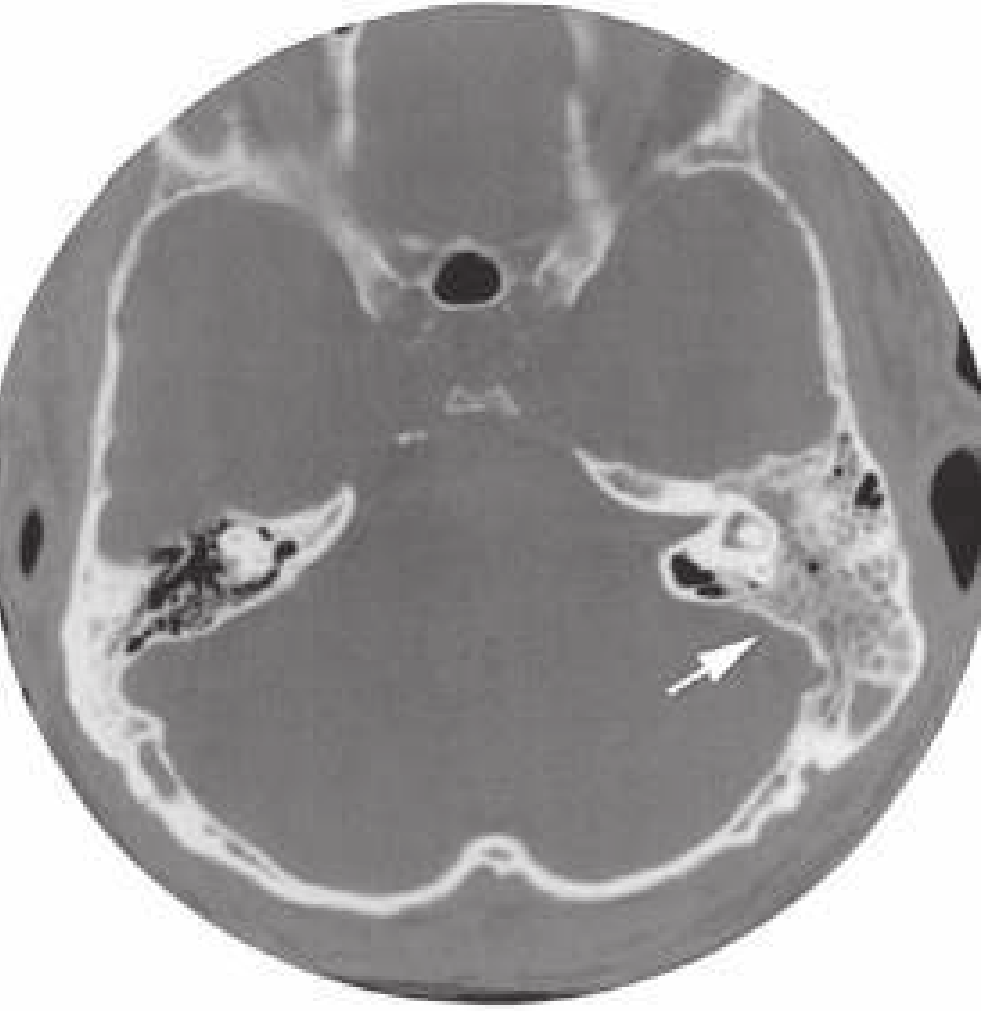
Management
Hospitalization required for IV antibiotics.
| Scenario | Antibiotic |
|---|---|
| No recent AOM or antibiotics | Vancomycin 15 mg/kg IV |
| Recent AOM (within 6 months) or prior antibiotics | Vancomycin + anti-pseudomonal agent (e.g., cefepime 50 mg/kg IV in children; 2 g IV in adults) |
Surgical options (determined with ENT):
- Myringotomy ± tympanostomy tube placement (to drain middle ear)
- Mastoidectomy for coalescent mastoiditis, subperiosteal abscess, or failure of medical therapy
Early ENT referral is mandatory.
Complications of untreated mastoiditis: subperiosteal abscess, Bezold's abscess (spread along sternocleidomastoid), petrous apicitis, sigmoid sinus thrombosis, meningitis, subdural empyema, brain abscess.
— Rosen's Emergency Medicine, p. 912–913
III. Chronic Otitis Media (COM)
Chronic otitis media encompasses prolonged inflammatory disease of the middle ear and mastoid, classified into two main forms based on the location of TM involvement:
A. Mesotympanitis (Tubotympanic Disease / "Safe" COM)
Definition: Chronic inflammation involving the central (pars tensa) portion of the TM with a central perforation. This form does not involve the attic and is considered "safe" because it carries a much lower risk of serious complications.
Features:
- Central TM perforation (pars tensa), with intact annulus
- Persistent or recurrent mucoid/mucopurulent otorrhea through the perforation
- Conductive hearing loss proportional to perforation size and ossicular involvement
- Mucosa of the middle ear is inflamed (granulation tissue in 97% of operated cases — Cummings, Table 140.3)
- No cholesteatoma (distinguishes it from epitympanitis)
Bacterial pathogens in chronic otorrhea:
- P. aeruginosa (most common in chronic disease)
- S. aureus
- Gram-negative rods
- Anaerobes in long-standing cases
Pathology (temporal bone findings with perforated TM):
- Granulation tissue: 97.4%
- Ossicular changes: 90.5%
- Tympanosclerosis: 19.8%
- Cholesterol granuloma: 12.1%
- Cholesteatoma: 4.3%
Treatment:
- Conservative (dry ear): Aural toilet, topical quinolone drops (e.g., ofloxacin — avoids ototoxicity risk of aminoglycosides)
- Systemic antibiotics for acute exacerbations guided by culture
- Surgery (tympanoplasty / myringoplasty): TM repair once ear is dry; ossiculoplasty if ossicular chain is disrupted. Goal: eliminate infection, restore hearing, prevent recurrence
— Cummings Otolaryngology, p. 2683–2685
B. Epitympanitis (Atticoantral Disease / "Unsafe" COM)
Definition: Chronic inflammation involving the pars flaccida (attic) or pars tensa posterior-superior quadrant, characteristically associated with a marginal (attic/retraction pocket) perforation and cholesteatoma formation. This form is "unsafe" due to the risk of bone erosion and serious complications.
Cholesteatoma — central to epitympanitis:
A cholesteatoma is an epidermal inclusion cyst of the middle ear or mastoid, filled with keratinous debris. Classified as:
- Congenital: develops behind an intact TM from embryonic epithelial rests
- Acquired (primary): most common — a retraction pocket in the pars flaccida or posterosuperior pars tensa deepens due to Eustachian tube dysfunction, accumulates keratin, and forms a cyst
- Acquired (secondary): keratinizing epithelium migrates through a pre-existing TM perforation
Mechanism of bone erosion:
- Osteoclastic activity driven by inflammatory cytokines, pressure, keratin, bacterial toxins
- The attic communicates with the antrum only through two narrow openings (between the tensor tympani tendon and stapes; and between the short process of incus and stapedial tendon) — blockage by edema/granulation tissue promotes chronic infection and bone erosion
— Cummings Otolaryngology, p. 2685–2690
Clinical features of epitympanitis:
- Attic (marginal/posterosuperior) perforation of TM
- Scanty, foul-smelling otorrhea (keratin debris; typically less profuse than mesotympanitis)
- Conductive hearing loss
- Otoscopy: white pearly debris or crusting in the attic area (Shrapnell's membrane region)
- Aural polyp may protrude through the perforation
- Potential for serious complications due to bone erosion
Complications specific to epitympanitis/cholesteatoma:
- Ossicular erosion → conductive hearing loss
- Labyrinthine fistula → sensorineural hearing loss, vertigo
- Facial nerve canal erosion → facial palsy
- Tegmen erosion → meningitis, brain abscess
- Sigmoid sinus erosion → lateral sinus thrombosis
- Petrous apicitis (Gradenigo's syndrome: trigeminal pain, abducens palsy, otorrhea)
Treatment:
- Surgery is mandatory — cholesteatoma cannot be eradicated medically
- Canal wall up (CWU) tympanomastoidectomy: preserves the posterior canal wall; better hearing outcome; requires "second-look" surgery at 12 months to check for residual cholesteatoma
- Canal wall down (CWD) mastoidectomy / modified radical mastoidectomy: removes the posterior canal wall, creating an open mastoid cavity; better visualization and lower recurrence, but creates a permanent "mastoid bowl" requiring lifelong care
— Cummings Otolaryngology, p. 2690–2695
Summary Comparison
| Feature | AOM | Mastoiditis | Mesotympanitis | Epitympanitis |
|---|---|---|---|---|
| Duration | Acute | Acute complication | Chronic | Chronic |
| TM finding | Bulging, intact | Bulging/abnormal | Central perforation | Attic/marginal perforation |
| Cholesteatoma | No | No | No | Yes |
| Otorrhea | None/after rupture | None/with AOM | Mucoid, profuse | Scanty, fetid |
| Bone destruction | No | Mastoid trabeculae | Uncommon | Characteristic |
| Risk of complications | Low-moderate | Moderate | Low ("safe") | High ("unsafe") |
| Primary treatment | Antibiotics | IV antibiotics ± surgery | Topical Abx ± tympanoplasty | Surgery mandatory |
Sources: Rosen's Emergency Medicine: Concepts and Clinical Practice; Cummings Otolaryngology: Head and Neck Surgery
Acute inflammation of the middle ear. Mastoiditis. Chronic inflammation of the middle ear (mesotympanitis. epitympanitis)
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have rich textbook content and clinical images to compile a thorough, well-structured answer. Here it is:
Middle Ear Infections: Acute Otitis Media, Mastoiditis, and Chronic Otitis Media
I. Acute Otitis Media (AOM)
Definition & Epidemiology
AOM is a bacterial infection of the middle ear space characterized by vascular dilation, mucosal edema, exudation, bacterial proliferation, white blood cell infiltration, and pus formation. It arises de novo in a previously normal middle ear — this distinction from acute exacerbations of chronic disease is important because bacteriology, treatment, and complication patterns differ.
AOM is the most common bacterial infection of childhood: ~85% of children have at least one episode. Peak incidence is at 6–24 months of age. By age 6, approximately 60% of children will have had at least one episode. It is the primary driver of pediatric antibiotic prescriptions.
Predisposing factors: young age, male sex, bottle-feeding, daycare attendance, crowded living conditions, parental smoking, cleft palate, Down syndrome, cystic fibrosis, ciliary dyskinesia, immunodeficiency. Breastfeeding is protective.
— Cummings Otolaryngology, p. 2702; Harrison's Principles of Internal Medicine 22E, p. 297
Pathophysiology
A viral upper respiratory infection causes Eustachian tube inflammation and obstruction → negative middle ear pressure → fluid accumulation → bacteria ascend from the nasopharynx → suppurative infection. Viruses are co-isolated from middle ear aspirates in ~70% of AOM cases. The Eustachian tube is shorter, more horizontal, and more prone to dysfunction in infants, explaining peak childhood incidence.
Causative bacteria (roughly by frequency):
| Organism | Notes |
|---|---|
| Streptococcus pneumoniae | ~30–50%; most virulent; most common in mastoiditis |
| Haemophilus influenzae (non-typeable) | ~20–45%; increasing post-PCV13; β-lactamase in 33–50% |
| Moraxella catarrhalis | ~10%; >90% β-lactamase-producing |
| S. pyogenes, S. aureus | Less common but important in neonates and complications |
| Gram-negatives, Group B Strep | Neonatal AOM specifically |
Despite routine pneumococcal conjugate vaccination, S. pneumoniae remains one of the most commonly isolated pathogens.
— Cummings Otolaryngology, p. 2702–2703; Harrison's 22E, p. 297
Clinical Features
- Otalgia — the cardinal symptom (unilateral or bilateral)
- Fever, irritability, poor feeding, ear-pulling (nonspecific in infants)
- Otoscopy (essential for diagnosis):
- Bulging, erythematous, opacified tympanic membrane (TM)
- Obscured bony landmarks
- Absent or reduced TM mobility on pneumatic otoscopy — the most reliable sign
- Conductive hearing loss
- Severe AOM criteria: otalgia ≥48 hours, temperature >39°C (102.2°F), moderate-to-severe bulging of TM
AOM should not be diagnosed without evidence of middle ear effusion. Bullae on the TM (bullous myringitis) may occur; treatment is identical.
— Harrison's 22E, p. 297–298
Management
Analgesia first: NSAIDs (ibuprofen) or acetaminophen for pain relief. Topical benzocaine or lidocaine may provide brief added relief. Antibiotics do NOT relieve pain in the first 24 hours.
Observation vs. antibiotics: Up to 80% of cases resolve without antibiotics. Watchful waiting (48–72 hrs) is appropriate for:
- Children ≥6 months with mild, unilateral AOM
- Children ≥2 years with nonsevere symptoms
Antibiotics are indicated immediately for:
- Age <6 months (all cases)
- Bilateral AOM in children 6–24 months
- AOM with otorrhea (any age >6 months)
- Severe otalgia, fever >39°C, or symptoms >48 hours (any age)
- Adults: treat all with antibiotics
Antibiotic regimens:
| Situation | Regimen |
|---|---|
| First-line (no PCN allergy) | Amoxicillin 80–90 mg/kg/day ÷ BID (max 3 g/day) |
| Prior amoxicillin in 30 days / concurrent conjunctivitis / β-lactamase coverage | Amoxicillin-clavulanate 90/6.4 mg/kg/day ÷ TID |
| Non-severe PCN allergy | Cefdinir, cefuroxime, cefpodoxime, or ceftriaxone IM |
| Treatment failure at 3 days | Amoxicillin-clavulanate or IM ceftriaxone × 3 days |
| AOM with tympanostomy tubes | Ototopical fluoroquinolone drops (e.g., ofloxacin) — aminoglycosides are ototoxic through non-intact TM |
Duration: 10 days for children <2 years, TM perforation, or severe/recurrent disease; 5–7 days for milder cases in older children.
The number needed to treat to prevent one case of mastoiditis is approximately 5,000 — antibiotics provide modest benefit but are warranted in higher-risk patients.
— Harrison's 22E, p. 297–298; K.J. Lee's Essential Otolaryngology, p. 216
II. Mastoiditis
Definition & Pathogenesis
Mastoiditis is inflammation of the mastoid air cells and is the most common suppurative (extracranial) complication of AOM. Its incidence has fallen sharply with antibiotics but it remains the most feared acute complication. It can occur without preceding clinically apparent AOM, and in older children with acute mastoiditis, an underlying cholesteatoma should be suspected.
Anatomic key: The middle ear communicates with the mastoid via the aditus ad antrum — a narrow passage. Obstruction creates a closed-space infection:
- Incipient mastoiditis — fluid fills mastoid cells, no bony destruction yet
- Coalescent mastoiditis (acute mastoid osteitis) — progressive demineralization and destruction of bony septa between air cells → coalescence into a single cavity
- Subperiosteal abscess — infection spreads through the mastoid cortex to the subperiosteal space, displacing the auricle
Infection can also spread via venous channels from mastoid veins. In infants, open cribriform channels near the spine of Henle allow direct passage to the subperiosteal space before bony fusion.
Pathogens: S. pneumoniae (leading cause), S. pyogenes, H. influenzae, S. aureus (including MRSA), P. aeruginosa (especially after prior antibiotic use), Fusobacterium necrophorum.
— Cummings Otolaryngology, p. 2703; Harrison's 22E, p. 298
Clinical Features
- Fever, otalgia, hearing loss, headache
- Postauricular erythema, tenderness, and swelling — most consistent finding
- Protrusion of the auricle downward and laterally — due to abscess displacing the upper mastoid → highly characteristic
- TM typically shows AOM changes (bulging, erythema); may be normal in ~10%
- Superoposterior protrusion of the posterior bony canal wall on otoscopy
- Symptoms of AOM persisting >2 weeks should raise suspicion
Masked Mastoiditis
A distinct variant where chronic granulation and bone erosion occur without otorrhea and with a near-normal TM — typically in patients receiving repeated antibiotic courses that partially suppress (but do not eliminate) the mastoid infection. Presents with chronic postauricular pain and mastoid tenderness. CT shows a localized area of mastoid opacification. Treatment is surgical.
Investigations
- Clinical diagnosis is sufficient with classic findings
- CT temporal bones (HRCT): Indicated for neurologic symptoms, failure to improve, or uncertain diagnosis. Shows:
- Opacification of mastoid air cells
- Loss/destruction of bony septa (coalescence)
- Subperiosteal abscess
- Erosion of tegmen, sigmoid sinus plate, labyrinth
- Sensitivity 97%, positive predictive value 94% for AOM complications
- MRI with gadolinium: Preferred for intracranial complications (meningitis, brain abscess, sigmoid sinus thrombosis) — better soft tissue detail
Management
Hospitalization and IV antibiotics are required for all patients.
| Patient | Antibiotic |
|---|---|
| No recent AOM / no prior antibiotics | Vancomycin 15 mg/kg IV (covers resistant S. pneumoniae and MRSA) |
| Recurrent AOM or prior antibiotic use | Vancomycin + anti-pseudomonal agent (cefepime 50 mg/kg IV in children; 2 g IV in adults; or ceftazidime) |
IV therapy for 7–10 days, then complete a 4-week oral antibiotic course. Culture from myringotomy/abscess drainage guides de-escalation.
Surgical management (ENT referral essential):
- Myringotomy ± tympanostomy tube: First step — decompresses middle ear, provides culture material
- Mastoidectomy: If no improvement within 48 hours of IV antibiotics, coalescent mastoiditis confirmed on CT, or complications present
- Abscess drainage: Subperiosteal or Bezold's abscess requires direct surgical drainage
Complications (Box 141.1)
| Extracranial | Intracranial |
|---|---|
| Coalescent mastoiditis | Meningitis |
| Subperiosteal abscess | Brain abscess |
| Bezold's abscess (tracks along sternocleidomastoid) | Epidural abscess |
| Temporal/zygomaticroot abscess | Subdural empyema |
| Petrous apicitis (Gradenigo's syndrome) | Lateral sinus thrombosis |
| Labyrinthine fistula | Otitic hydrocephalus |
| Facial nerve paralysis | |
| Acute suppurative labyrinthitis |
— Cummings Otolaryngology, p. 2703–2715; Harrison's 22E, p. 298–299
III. Chronic Otitis Media (COM)
Definition: COM reflects inflammation and infection of the middle ear and mastoid persisting longer than 3 weeks (the usual resolution time of AOM in a previously healthy ear). COM can occur with or without cholesteatoma, and a third form occurs in children with patent tympanostomy tubes.
When infection does not resolve, mucosal edema and exudation increase, mucous glands proliferate, and blockage of the aditus/epitympanum prevents drainage of the antrum and mastoid, leading to "irreversible" mucosal and bony changes.
COM is divided into two clinical forms based on TM involvement:
A. Mesotympanitis (Tubotympanic COM / "Safe" Disease)
Location: Central/pars tensa of the TM.
Defining feature: A central (safe) perforation of the TM — the annulus fibrosus remains intact. Because the perforation is away from the attic, it does not carry the same risk of cholesteatoma formation as attic disease.
Pathological findings (temporal bone histopathology with perforated TM):
| Finding | Frequency |
|---|---|
| Granulation tissue | 97.4% |
| Ossicular changes | 90.5% |
| Tympanosclerosis | 19.8% |
| Cholesterol granuloma | 12.1% |
| Cholesteatoma | 4.3% |
Data: da Costa et al., Laryngoscope 102:1229, 1992 — cited in Cummings Otolaryngology
Clinical features:
- Persistent or intermittent otorrhea — mucoid or mucopurulent, typically odourless or mildly malodorous, often profuse
- Central TM perforation visible on otoscopy
- Conductive hearing loss — proportional to perforation size and ossicular involvement
- Recurrent episodes often triggered by upper respiratory infections (water entering through the perforation)
- Middle ear mucosa appears thickened, edematous, with granulation tissue
Bacteriology in chronic otorrhea:
- Pseudomonas aeruginosa (most common in chronic disease)
- Staphylococcus aureus
- Mixed gram-negatives, anaerobes
- Biofilms are common and contribute to antibiotic resistance
Treatment:
- Aural toilet — meticulous cleaning (microsuction) to remove debris and secretions
- Topical therapy: Fluoroquinolone ear drops (ciprofloxacin/dexamethasone, ofloxacin) — avoid aminoglycosides due to ototoxicity risk through perforated TM; acetic acid/alcohol solutions have antiseptic utility
- Systemic antibiotics for acute exacerbations guided by culture/sensitivity
- Surgery (definitive): Tympanoplasty (myringoplasty) once the ear is dry — to close the perforation and restore the sound-conducting mechanism. Ossiculoplasty if the ossicular chain is disrupted. Goal: dry ear, hearing rehabilitation, prevention of recurrence.
B. Epitympanitis (Atticoantral COM / "Unsafe" Disease)
Location: Pars flaccida (Shrapnell's membrane) or posterosuperior pars tensa — the attic (epitympanum).
Defining feature: An attic (marginal) perforation or retraction pocket, invariably associated with cholesteatoma — which is the key feature distinguishing this form and making it "unsafe."
Cholesteatoma
A cholesteatoma is an epidermal inclusion cyst of the middle ear or mastoid filled with desquamating keratin debris. Classified as:
- Congenital: Develops behind an intact TM from embryonic epithelial rests; found in children
- Primary acquired (most common): A retraction pocket in the pars flaccida deepens progressively due to chronic Eustachian tube dysfunction and negative middle ear pressure. Keratin accumulates, and the self-cleaning mechanism of the skin fails, creating an expanding cyst
- Secondary acquired: Keratinizing squamous epithelium migrates through an existing TM perforation (typically in mesotympanitis), invades the middle ear/mastoid, and forms a cholesteatoma
Why cholesteatoma erodes bone:
The attic communicates with the mastoid antrum through only two constant narrow openings (between the tensor tympani tendon and the stapes; and between the short process of the incus and the stapedial tendon). Edema/granulation tissue blocks these, causing persistent infection. Bone erosion is driven by:
- Osteoclastic activation by inflammatory cytokines (IL-1, TNF-α), localized pressure, keratin, bacterial toxins
- Fibroblasts in the subepithelium with invasive phenotype
— Cummings Otolaryngology, p. 2685–2690
Clinical features of epitympanitis:
- Attic/marginal or posterosuperior perforation on otoscopy — white pearly debris, crust, or granulation in the attic (Shrapnell's membrane area)
- Scanty but foul-smelling (fetid) otorrhea — keratin debris; less voluminous than mesotympanitis
- Conductive hearing loss (progressive with ossicular erosion)
- Aural polyp may prolapse through the perforation
- High risk of serious complications from bone destruction
Complications specific to epitympanitis/cholesteatoma:
- Ossicular erosion → conductive hearing loss (incus long process most commonly eroded)
- Labyrinthine fistula (most often horizontal SCC) → sensorineural hearing loss, vertigo; positive fistula test (Hennebert's sign)
- Facial nerve canal erosion → peripheral facial palsy
- Tegmen plate erosion → meningitis, brain abscess, CSF otorrhea
- Sigmoid sinus erosion → lateral sinus thrombosis
- Petrous apicitis → Gradenigo's syndrome (trigeminal pain, abducens palsy, otorrhea)
Investigations:
- High-resolution CT temporal bones: Reveals the extent of cholesteatoma, ossicular chain erosion, tegmen/sigmoid sinus/labyrinthine erosion. Guides surgical planning.
- MRI (non-EPI DWI): Diffusion-weighted MRI is now the gold standard for detecting residual/recurrent cholesteatoma post-surgery without the need for a "second-look" operation.
Treatment — Surgery is mandatory:
Cholesteatoma cannot be eradicated by medical therapy alone.
| Technique | Description | Advantage/Disadvantage |
|---|---|---|
| Canal wall-up (CWU) tympanomastoidectomy | Removes disease while preserving posterior canal wall | Better hearing; more physiological; requires second-look at 12 months to detect residual disease |
| Canal wall-down (CWD) / modified radical mastoidectomy | Removes posterior canal wall, creating open mastoid cavity | Lower recurrence risk; better visualization; permanent mastoid bowl requires lifelong aural toilet |
| Radical mastoidectomy | Exenteration of all middle ear contents | Reserved for extensive disease/complications; abolishes hearing |
Labyrinthine fistula at surgery: the cholesteatoma matrix is carefully peeled from the endosteum under high magnification; the fistula site is addressed last and covered with fascia/bone.
Summary Comparison
| Feature | Acute Otitis Media | Mastoiditis | Mesotympanitis | Epitympanitis |
|---|---|---|---|---|
| Nature | Acute | Acute complication | Chronic | Chronic |
| TM finding | Bulging, intact, erythematous | Bulging / abnormal | Central perforation (pars tensa) | Attic / marginal perforation (pars flaccida) |
| Otorrhea | None (unless TM ruptures) | With AOM or absent | Mucoid, profuse, odourless | Scanty, fetid (keratin) |
| Cholesteatoma | No | No | No (rarely) | Yes — defining feature |
| Bone destruction | No | Mastoid trabeculae | Uncommon | Characteristic, progressive |
| Safety classification | — | — | "Safe" | "Unsafe" |
| Hearing loss | Mild conductive (effusion) | Conductive | Conductive | Conductive → sensorineural |
| Complication risk | Low-moderate | Moderate-high | Low | High |
| Primary treatment | Antibiotics ± observation | IV antibiotics + surgery | Aural toilet + topical Abx ± tympanoplasty | Surgery mandatory |
Sources: Cummings Otolaryngology: Head and Neck Surgery, 7th ed. — K.J. Lee's Essential Otolaryngology, 11th ed. — Harrison's Principles of Internal Medicine, 22nd ed.
Labyrinthitis. Otogenic intracranial complications (meningitis. meningoencephalitis. arachnoiditis. abscess of the brain and cerebellum. thrombophlebitis of the sigmoid sinus).
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
Reading File
I now have all the material needed. Let me compose the comprehensive answer:
Labyrinthitis and Otogenic Intracranial Complications
Routes of Spread from the Middle Ear to Intracranial Structures
Before addressing each complication individually, it is essential to understand how infection spreads from the temporal bone to the cranium. Four main pathways exist:
- Direct extension through bone eroded by cholesteatoma, granulation tissue, or coalescent mastoiditis
- Venous propagation — thrombophlebitis of diploic veins and mastoid emissary veins carries septic thrombus to adjacent sinuses and intracranial vessels
- Labyrinthine pathway — through the foramina of the internal auditory canal, the cochlear aqueduct, or labyrinthine windows, connecting the middle ear/mastoid with the CSF
- Preformed pathways — congenital bony dehiscences, prior surgical defects, tegmen perforations, perilymph fistulas
Critical diagnostic point: The TM and middle ear may appear normal or near-normal even when serious complications are developing in the mastoid — particularly in masked mastoiditis. The mastoid does not necessarily respond in parallel with the middle ear under antibiotic treatment.
— Cummings Otolaryngology, p. 2703
I. Labyrinthitis
Labyrinthitis refers to inflammation of the membranous labyrinth (cochlea and vestibular apparatus). It is classified into serous and suppurative forms based on whether bacteria physically invade the inner ear.
A. Serous (Toxic) Labyrinthitis
Definition: Inflammation of the labyrinth in the absence of bacteria within the inner ear. Bacterial toxins and inflammatory mediators enter through intact membranes.
Pathophysiology:
- Bacterial toxins/inflammatory mediators enter the labyrinth via the oval window, round window membrane, or labyrinthine fistula
- Round window membrane permeability is significantly increased during middle ear inflammation
- Labyrinthine dysfunction results from changes in ionic potentials caused by inflammatory mediators rather than neuroepithelial destruction
- Because the neuroepithelium is not destroyed, cochlear/vestibular function can return to normal after the inflammatory state resolves
Most common in children due to high incidence of AOM.
Pathogens: S. pneumoniae, H. influenzae, M. catarrhalis
Clinical features:
- Typically unilateral
- Dizziness (not required for diagnosis)
- Hearing loss (mild to severe sensorineural) — typically reversible
- Symptoms resolve over time as inflammation subsides
Histopathology: Loss of outer hair cells primarily in basal turn, with significant incidence of endolymphatic hydrops.
Diagnosis:
- Audiogram (SNHL)
- Culture of middle ear effusion when present
- Vestibular testing when necessary
- Imaging only if other complications suspected
Treatment:
- Directed at the infectious source — oral antibiotics typically effective
- Steroids (oral and/or intratympanic) may improve hearing outcomes but evidence is limited
- Myringotomy for unresolved middle ear infection
- Tympanomastoidectomy if cholesteatoma is present
— K.J. Lee's Essential Otolaryngology, p. 460
B. Suppurative (Purulent) Labyrinthitis
Definition: Bacterial invasion of the labyrinth itself, causing irreversible destruction of cochlear and vestibular neuroepithelium. Uncommon in the antibiotic era; most commonly associated with cholesteatoma.
Routes of bacterial entry into the labyrinth:
- From the subarachnoid space (meningitis) — bacteria enter the scala tympani via the patent cochlear aqueduct or travel up the IAC through perineural/perivascular spaces
- From the temporal bone (osteomyelitis) — direct erosion
- From the middle ear (AOM/COM ± cholesteatoma):
- Through a weakened or dehiscent oval window membrane (especially in Mondini deformity, enlarged vestibular aqueducts, or post-stapes surgery)
- Through a cholesteatomatous lateral semicircular canal fistula — infected granulation tissue lies directly on the endosteal membrane; bacteria contact underlying perilymph
- Through foramina of the internal auditory canal
Both directions of spread are possible: suppurative labyrinthitis can cause meningitis, and meningitis can cause suppurative labyrinthitis. This bidirectional relationship is clinically important, particularly in children with congenital labyrinthine anomalies.
Pathogens: Those of the underlying AOM or cholesteatoma — S. pneumoniae, S. pyogenes, H. influenzae, P. aeruginosa, gram-negative rods (polymicrobial in otogenic cases).
Clinical features (classic progression):
- Tinnitus and dizziness → rapidly progressing to whirling vertigo, pallor, diaphoresis, nausea, and vomiting
- Brisk nystagmus directed toward the opposite (unaffected) ear
- Over first several hours: spontaneous vertigo and nystagmus begin to abate
- Over 2–3 weeks: central compensation restores near-normal balance
- Complete, permanent, unilateral sensorineural hearing loss (total cochlear destruction)
- If due to meningitic spread: bilateral profound deafness (10% average; range 2–20%)
- Fever, meningeal signs if associated meningitis; cranial neuropathies if disease spreads beyond the otic capsule
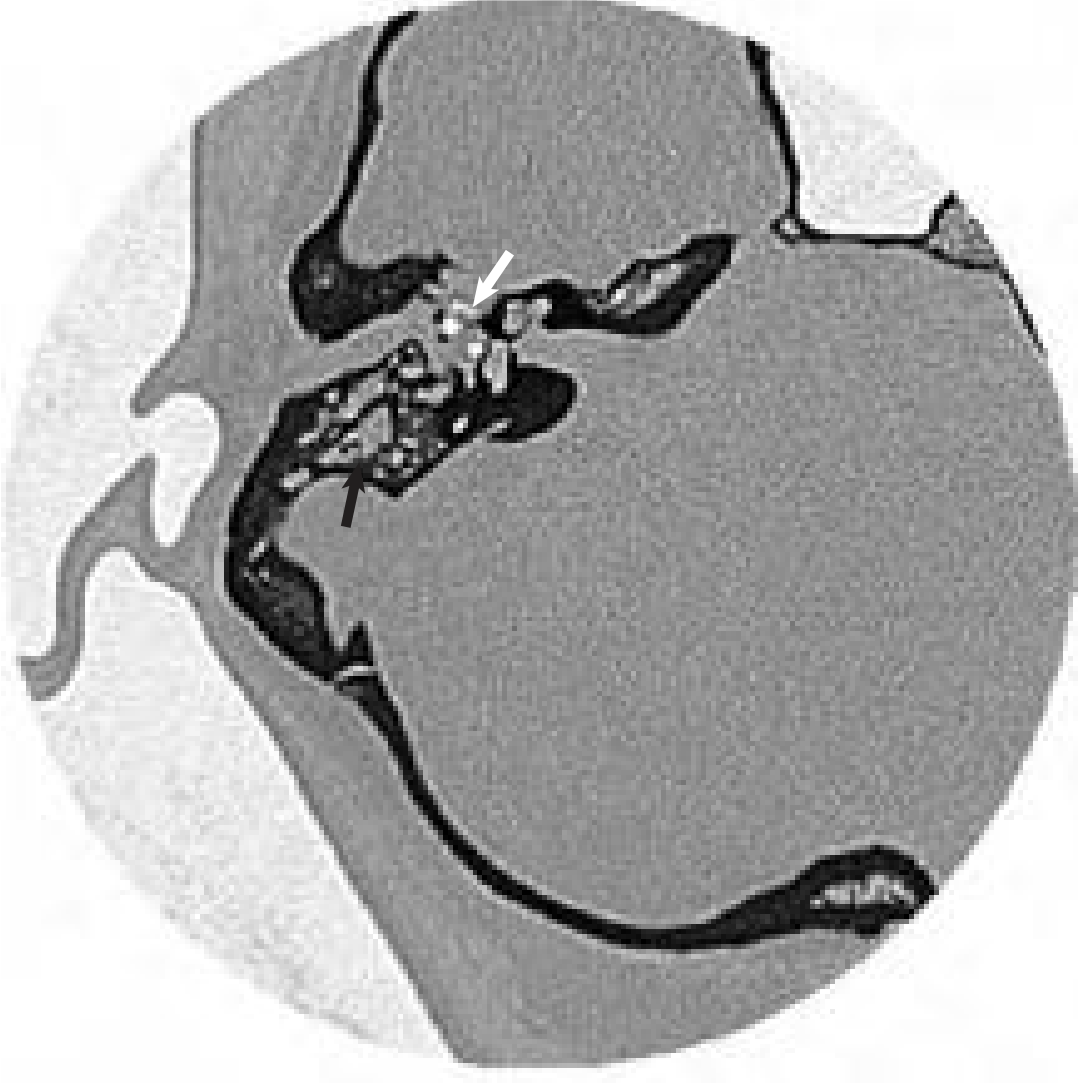
Imaging findings:
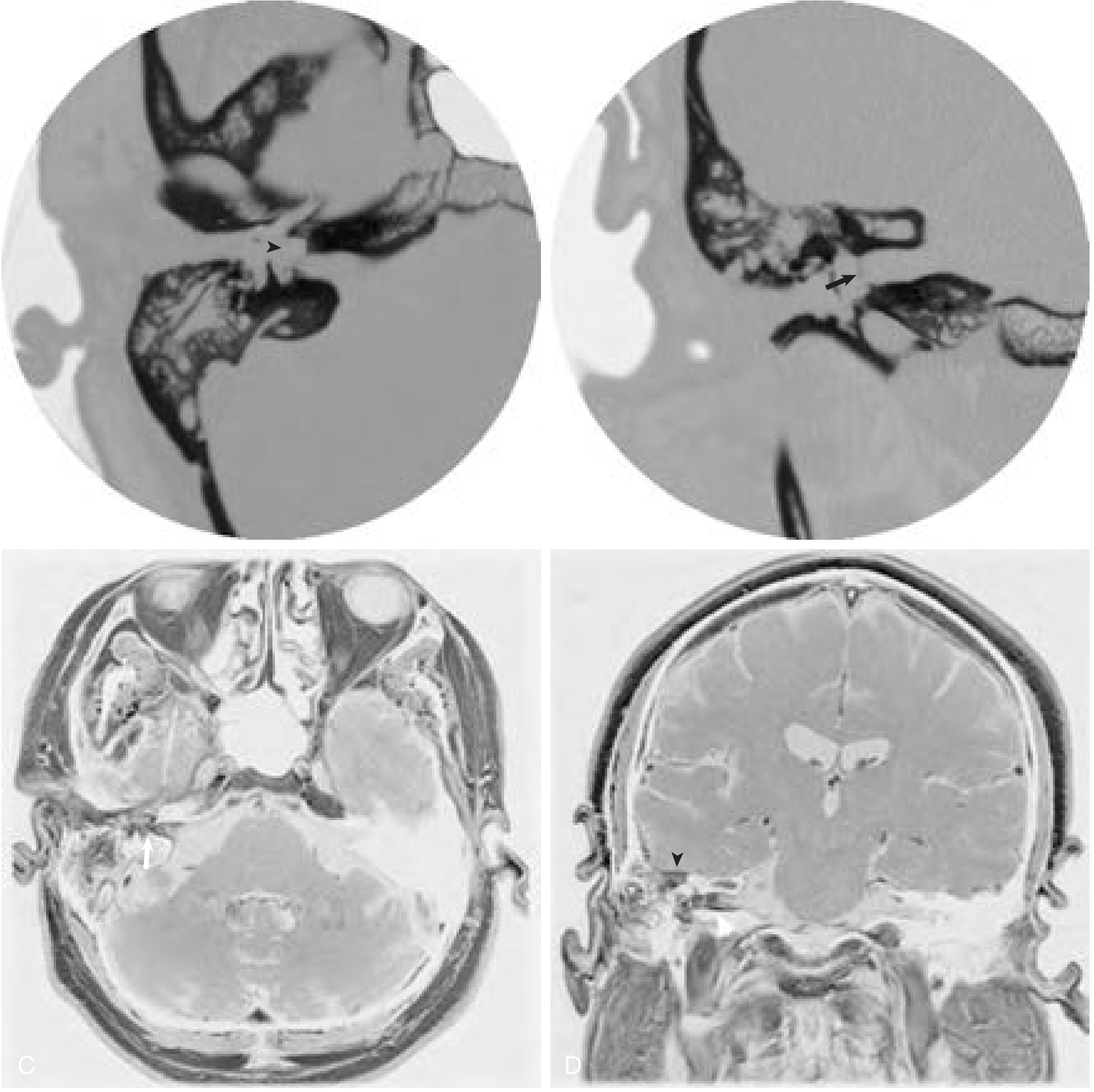
Complications of suppurative labyrinthitis:
- Labyrinthitis ossificans — ossification of the cochlea following inflammation; detected by MRI and HRCT; if bilateral SNHL is present, early cochlear implantation is indicated before the cochlear duct is obliterated
- Meningitis (via cochlear aqueduct/IAC spread)
Treatment:
- Parenteral antibiotics with CSF penetration for 10 days (to eradicate infection and prevent meningeal spread)
- Directed at underlying cause (AOM or COM/cholesteatoma)
- Myringotomy ± ventilation tube for non-cholesteatoma otogenic infection
- Tympanomastoidectomy if cholesteatoma present (labyrinthectomy is NOT necessary in cases secondary to AOM)
- Dexamethasone improves hearing outcomes in meningitic labyrinthitis due to H. influenzae and S. pneumoniae
- Serial CT/MRI for labyrinthitis ossificans if SNHL present; early cochlear implant referral for bilateral cases
— Cummings Otolaryngology, p. 2716–2717; K.J. Lee's Essential Otolaryngology, p. 460–461
II. Otogenic Intracranial Complications
Despite widespread antibiotic use and vaccination, intracranial complications of otitis media continue to occur and carry high morbidity and mortality. Complications from COM + cholesteatoma are generally more dangerous than those from AOM.
Classification (Box 141.1):
| Extracranial | Intracranial |
|---|---|
| Coalescent/masked mastoiditis | Meningitis |
| Subperiosteal/Bezold/temporal abscess | Brain abscess (temporal lobe, cerebellar) |
| Petrous apicitis (Gradenigo's) | Subdural empyema |
| Labyrinthine fistula | Epidural abscess |
| Acute suppurative labyrinthitis | Lateral sinus thrombosis |
| Facial nerve paralysis | Otitic hydrocephalus |
Nearly all patients with lateral sinus thrombosis have one or more additional intracranial complications. Multiple simultaneous complications are common in COM.
— Cummings Otolaryngology, p. 2703–2722
1. Otogenic Meningitis
Epidemiology: Historically the most common intracranial complication of otitis media. Dramatically reduced by H. influenzae type B vaccination (1980s) and PCV13 pneumococcal vaccination. Incidence of otogenic meningitis is now inversely proportional to vaccination prevalence in a given country.
Pathways from ear to meninges:
- Hematogenous dissemination (most common in children <2 years with AOM)
- Extension through the tegmen tympani (bony plate separating middle ear from middle cranial fossa)
- Via the labyrinthine route — cochlear aqueduct, foramina of the IAC
- Congenital anomalies (Mondini dysplasia, enlarged vestibular aqueduct, CSF fistulas)
- After stapes surgery (perilymph gusher/fistula)
- Traumatic temporal bone fracture with dural laceration
Pathogens: Previously H. influenzae and S. pneumoniae (responsible for nearly all otogenic cases). Post-vaccination era: S. pneumoniae dominant; gram-negative rods and polymicrobial organisms in COM-related cases.
Clinical features:
- Severe, generalized headache — earliest and most persistent symptom
- Photophobia, general hyperesthesia, social withdrawal
- High, sustained fever (universal)
- Nuchal rigidity — pain with neck flexion, most consistent meningeal sign
- Brudzinski sign — passive neck flexion causes involuntary bilateral hip and knee flexion
- Kernig sign — with thigh flexed at 90°, passive knee extension meets resistance and pain from hamstring spasm
- Altered level of consciousness (ranges from normal to unresponsive)
- Papilledema on fundoscopy
Investigations:
- CT scan first (before lumbar puncture) — to rule out brain abscess, cerebritis, subdural empyema; assesses safety of LP
- Lumbar puncture: elevated CSF pressure; early: protein ↑ and glucose ↓ vs. serum; bacteria absent until late; cells: neutrophilic pleocytosis
- MRI with gadolinium: shows dural enhancement; superior for detecting labyrinthine route of infection and additional complications
Treatment:
- Parenteral antibiotics with CSF penetration — directed at S. pneumoniae (consider resistance), gram-negatives
- Dexamethasone adjunct — evidence suggests decreased adverse outcomes in community-acquired pneumococcal meningitis; likely beneficial in otogenic cases
- When AOM/suppurative labyrinthitis is source: myringotomy + antibiotics; no surgery on mastoid required
- When COM/mastoiditis is source: mastoidectomy is mandatory as soon as patient is neurologically stable — the infected mastoid must be exenterated to eliminate the source
- If profound ipsilateral SNHL develops: route through labyrinth is confirmed; if SNHL is unchanged from baseline: route was extralabyrinthine
Otogenic meningitis is more lethal when caused by COM/mastoiditis than when it occurs as a hematogenous complication of AOM in infancy.
2. Meningoencephalitis and Arachnoiditis
Meningoencephalitis represents extension of bacterial meningitis into the brain parenchyma (cerebritis). Features overlap with meningitis but include:
- Focal neurological deficits
- Altered sensorium progressing to obtundation/coma
- Seizures
- MRI shows parenchymal edema and enhancement adjacent to the infected temporal bone
Otogenic arachnoiditis (leptomeningitis) refers to chronic adhesive inflammation of the arachnoid membrane following bacterial meningitis. Consequences:
- CSF outflow obstruction → communicating or non-communicating hydrocephalus
- Cranial nerve entrapment (particularly CN VIII → sensorineural hearing loss; CN VI → diplopia)
- Chronic headaches
Arachnoiditis is a late consequence and is managed with neurosurgical CSF diversion when indicated (shunting).
3. Epidural (Extradural) Abscess
Pathogenesis: Bone erosion from cholesteatoma, granulation tissue, or coalescent mastoiditis creates an abscess between the temporal bone and the outer surface of the dura mater. The dura thickens (pachymeningitis) in response to direct contact with granulation tissue. Frequently associated with lateral sinus thrombophlebitis, meningitis, and cerebritis.
Clinical features:
- Deep mastoid pain — the dominant symptom; may be the only symptom
- No specific signs or symptoms attributable to epidural abscess alone — many are discovered only at surgery
- Signs of underlying otomastoiditis (ear discharge, postauricular swelling)
- If large: can be detected on contrast-enhanced CT/MRI as a fluid collection between temporal bone and enhanced dura
Treatment:
- Surgery is the only recommended treatment
- Type of mastoidectomy appropriate to underlying otitis (canal wall-up or canal wall-down)
- Surgical approach: progressive exenteration from lateral to medial, from less diseased to more diseased regions; careful removal of overlying bone until healthy dura is evident on all margins; granulation tissue scraped from abscess cavity with a blunt elevator
4. Brain Abscess (Temporal Lobe and Cerebellar)
Epidemiology: Incidence and mortality have decreased substantially in developed countries. Otogenic causes are the third most common cause of intraparenchymal brain abscess (after cyanotic congenital heart disease and post-traumatic/neurosurgical). More than 75% in male patients, predominantly lower socioeconomic classes.
Location: Almost always ipsilateral to the otitis. Occur nearly equally in the temporal lobe (adjacent to the tegmen) and the cerebellum (adjacent to the cerebellar plate of the mastoid). ~75% are secondary to cholesteatoma.
Pathogenesis:
- Bacteria propagate in and around venous channels from the mastoid into adjacent brain parenchyma → cerebritis (stage 1)
- Cerebritis spreads to form a necrotic center of liquefied pus surrounded by cerebral edema (stage 2)
- A fibrous capsule forms from the surrounding gliosis (encapsulation, stage 3)
- Further spread/rupture (stage 4) — intraventricular rupture is catastrophic (mortality >80%)
Clinical features:
- Early (cerebritis phase): Headache, low-grade fever, malaise — often subtle and non-specific
- Established abscess:
- Increasing headache, high fever, signs of raised ICP (nausea, vomiting, papilledema, altered consciousness)
- Temporal lobe abscess: Dysphasia (dominant side), contralateral hemiparesis, visual field defects, focal seizures
- Cerebellar abscess: Ataxia, nystagmus, ipsilateral limb dysmetria (past-pointing), intention tremor, gait disturbance; may mimic labyrinthitis early
- Symptoms evolve over days to weeks (contrast with subdural empyema which can evolve in hours)
- Patients appear severely systemically ill; obtundation may be present
Imaging:
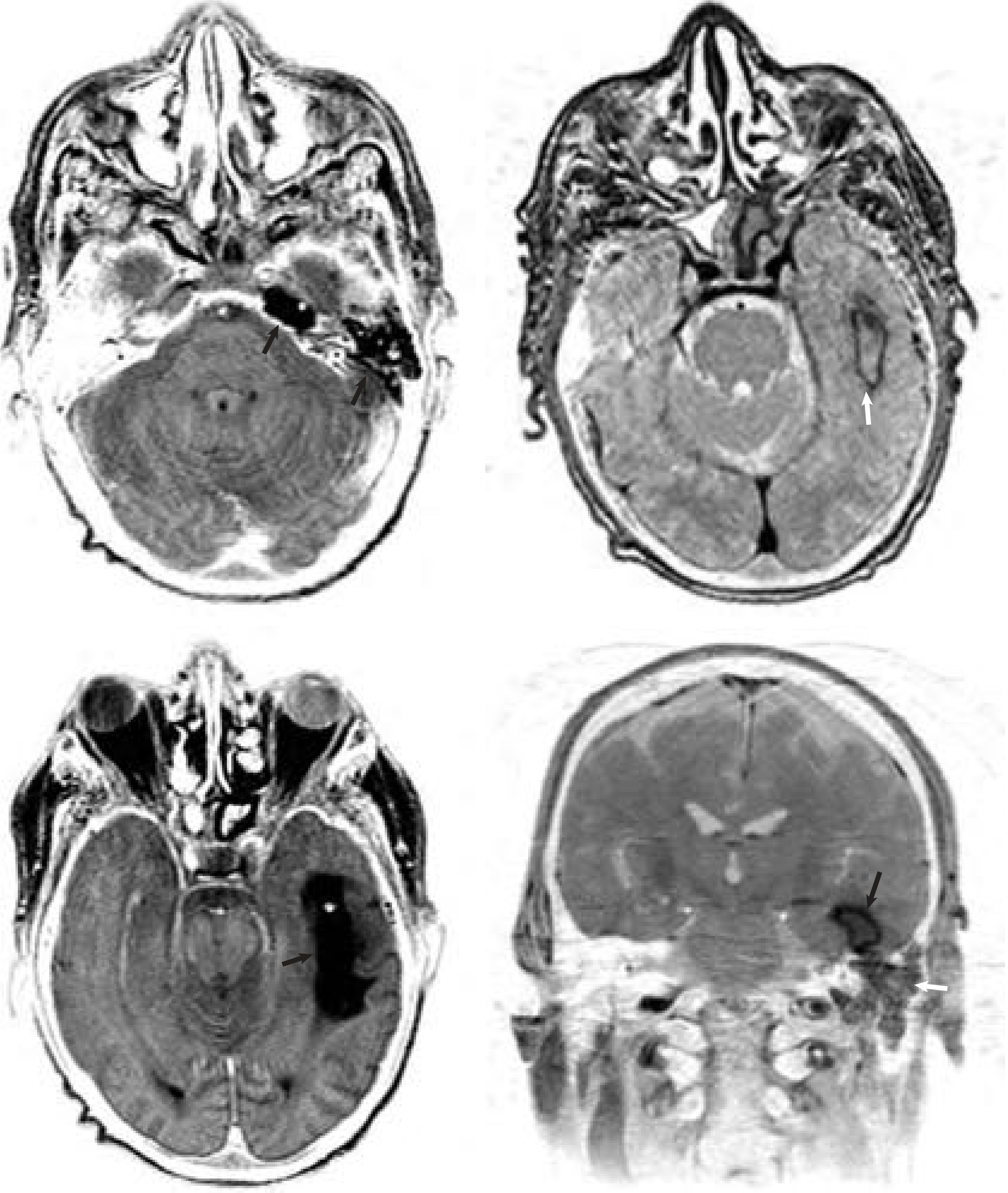
- CT with contrast: Ring-enhancing lesion with central hypodensity; surrounding edema; mass effect; midline shift if large
- MRI with gadolinium: More sensitive; defines extent, multiplicity, and capsule formation; diffusion-weighted imaging (DWI) confirms restricted diffusion in abscess cavity (distinguishes from necrotic tumor)
- CT/MRI of temporal bones: establishes underlying otologic diagnosis
Treatment:
- Neurosurgical drainage of the abscess takes precedence over mastoid surgery if both are present — drain the abscess first, then immediately proceed to mastoidectomy to eliminate the source
- Methods: stereotactic aspiration (for deep/eloquent areas), or craniotomy with excision (for superficial, encapsulated, posterior fossa lesions)
- Intravenous antibiotics covering gram-positives, gram-negatives, and anaerobes for extended duration (6–8 weeks)
- Mastoidectomy (canal wall-up or canal wall-down) to eliminate underlying COM/cholesteatoma
- Dexamethasone may be used to reduce cerebral edema
5. Thrombophlebitis of the Sigmoid (Lateral) Sinus
Anatomy: The sigmoid sinus is a continuation of the transverse sinus that drains into the internal jugular vein at the jugular bulb. It lies in close proximity to the posterior wall of the mastoid. The lateral sinus system includes both the sigmoid and transverse sinuses.
Pathogenesis:
- Bone erosion from cholesteatoma, granulation tissue, or coalescent mastoiditis creates a perisinus abscess
- Pressure on the outer dural wall → necrosis → extension to the intima
- Fibrin, blood cells, and platelets aggregate → mural thrombus forms
- Thrombus becomes infected, enlarges, and occludes blood flow
- Infected clot propagates in either direction:
- Anteriorly: to the transverse sinus, torcular herophili, superior sagittal sinus, cavernous sinus
- Inferiorly: to the jugular bulb and internal jugular vein in the neck
- Infected clot showers bacteria into the bloodstream → septicemia → metastatic abscesses (most commonly pulmonary)
Lateral sinus thrombosis can also occur via osteothrombophlebitis during AOM, even without gross bone erosion (sinus plate intact at surgery).
Clinical features:
Classic triad (may be attenuated in the modern antibiotic era):
- "Picket fence" fever pattern — diurnal temperature spikes exceeding 39.4°C (103°F), with intervening afebrile periods; characteristic of septicemia from infected thrombus shedding bacteria intermittently
- Severe headache and evidence of raised ICP
- Signs of otomastoiditis (ear pain, discharge, postauricular swelling)
Additional features:
- Neck pain — from sternocleidomastoid inflammation (tenderness to percussion of mastoid tip distinguishes this from meningitis-induced neck stiffness)
- Diplopia — from CN VI palsy with raised ICP (increasingly the presenting sign in the antibiotic era)
- Jugular foramen syndrome (if thrombosis extends to jugular bulb): palsies of CN IX, X, XI → hoarseness, dysphagia, shoulder weakness
- Progressive obtundation, worsening headache → bilateral involvement of venous outflow → cerebral edema → otitic hydrocephalus
In patients previously treated with antibiotics, the classic picket fence fever pattern is less common. Diplopia or other neurological signs may be the first manifestation. History of recent OM symptom resolution following antibiotics followed by new neurological symptoms should trigger investigation.
Griesinger's sign — edema of the skin over the mastoid emissary vein and postauricular region from thrombosis of the mastoid emissary vein.
Investigations:
- MRI/MRV (gold standard): Demonstrates absent flow in sigmoid/transverse sinus; extent of thrombus; associated complications (subdural empyema, cerebritis, cerebellar abscess)
- CT with contrast: The "delta sign" — absence of contrast within the vessel lumen with enhancement of the sinus wall; sensitive but less so than MRI
- Blood cultures (may be positive during septic emboli)
- CT/MRI temporal bones: cholesteatoma or mastoiditis as underlying cause
Treatment:
- Mastoidectomy — mandatory in all cases to eliminate underlying mastoid disease
- IV antibiotics — broad spectrum, covering gram-positives, gram-negatives, and anaerobes; prolonged course
- Anticoagulation — controversial; not routinely indicated given risks vs. unproven benefit in most cases; strongly considered when:
- Clot extends to transverse sinus or cavernous sinus
- Septic embolic events are present
- Clot evacuation from the sinus — considered if the sinus wall appears disrupted during exploration; a needle aspirates the sinus, and if pus is present, a linear incision is made for drainage; recent studies show no significant mortality benefit, but may shorten hospital stay
- Internal jugular vein ligation in the neck — considered if clot extends into the neck or septic emboli are present; not routinely performed
- For associated otitic hydrocephalus: urgent neurosurgical/neurological consultation; serial lumbar punctures or lumbar drain to reduce ICP; consider acetazolamide
— Cummings Otolaryngology, p. 2499–2542; Adams & Victor's Principles of Neurology 12E, p. 2137–2145
6. Otitic Hydrocephalus
A specific sequela of lateral sinus thrombosis (or transverse sinus occlusion) in which diminished venous drainage of the cranium raises intracranial pressure — not a true hydrocephalus (ventricles not enlarged), but a pseudotumor cerebri state caused by impaired venous outflow.
The likelihood of developing symptomatic intracranial hypertension depends on:
- The relative size of the occluded sinus vs. the opposite sinus (right > left in 60%)
- Adequacy of the collateral venous network (cavernous sinus, opposite inferior petrosal sinus)
- Whether propagation affects additional venous outflow (superior sagittal sinus)
Clinical features:
- Diffuse severe headache and nausea — dominant early symptoms
- With inadequate collateral drainage: dulling of sensorium, decreased visual acuity (retinal vein occlusion), papilledema
- Progression to coma and death possible if untreated
- CN VI palsy (diplopia) from raised ICP stretching the abducens nerve over the petrous tip
Treatment: Address the underlying otologic disease (mastoidectomy); serial LPs or lumbar drainage to reduce ICP; urgent neurosurgical consultation.
Summary: Routes, Complications, and Key Distinguishing Features
| Complication | Route | Key Sign | Investigation of Choice | Essential Treatment |
|---|---|---|---|---|
| Serous labyrinthitis | Toxins via round/oval window | Reversible SNHL + vertigo | Audiogram | Treat AOM; ± steroids |
| Suppurative labyrinthitis | Bacterial invasion (oval window, fistula, IAC) | Total irreversible ipsilateral deafness + violent vertigo | CT + MRI temporal bone | Parenteral Abx ± mastoidectomy |
| Otogenic meningitis | Hematogenous / tegmen / labyrinthine route | Nuchal rigidity, Kernig/Brudzinski signs | CT → LP; MRI gadolinium | Abx + dexamethasone; mastoidectomy if COM source |
| Epidural abscess | Direct bone erosion | Deep mastoid pain (often no other signs) | Contrast CT/MRI | Mastoidectomy with abscess drainage |
| Subdural empyema | Direct / retrograde venous | Rapid ↓ consciousness, contralateral hemiparesis, seizures | MRI (CT may miss early) | Emergency neurosurgical drainage + Abx |
| Temporal lobe abscess | Venous from tegmen area | Dysphasia, focal seizures, headache | MRI gadolinium + DWI | Neurosurgical aspiration/excision + mastoidectomy |
| Cerebellar abscess | Venous from cerebellar plate | Ipsilateral ataxia, nystagmus, dysmetria | MRI gadolinium + DWI | Neurosurgical drainage + mastoidectomy |
| Sigmoid sinus thrombosis | Perisinus abscess → thrombus | Picket fence fever + headache + diplopia | MRI/MRV; "delta sign" on CT | Mastoidectomy + IV Abx ± anticoagulation |
| Otitic hydrocephalus | Venous outflow obstruction | Papilledema + severe headache, ↑ ICP | MRI/MRV; LP shows raised pressure | Treat sinus thrombosis + LP/lumbar drain |
Sources: Cummings Otolaryngology: Head and Neck Surgery, 7th ed. — K.J. Lee's Essential Otolaryngology, 11th ed. — Adams & Victor's Principles of Neurology, 12th ed.
This is a shared conversation. Sign in to Orris to start your own chat.