Cardiac excitability disorders
Reading File
search_medical_text
cardiac excitability disorders arrhythmia pathophysiology classification
search_medical_image
cardiac arrhythmia ECG excitability disorders
GradeResponse
GradeResponse
Here is a comprehensive overview of cardiac excitability disorders.
Cardiac Excitability Disorders
Definition and Concept
Cardiac excitability disorders (also called cardiac channelopathies or ion channel disorders) are conditions in which dysfunction of ion channels — proteins governing transmembrane transit of Na⁺, K⁺, and Ca²⁺ — disrupts the electrical excitability of cardiomyocytes, leading to arrhythmias without necessarily causing structural myocardial disease.
The 2006 AHA Scientific Statement classified ion channel disorders as primary genetic cardiomyopathies, recognizing the pivotal role of pathogenic variants in ion channel genes (Evaluation, Risk Stratification, and Management of Arrhythmogenic Cardiomyopathy, p. 37).
Pathophysiology
Normal cardiac excitability depends on the action potential (AP), which proceeds through five phases:
| Phase | Event | Key Channels |
|---|---|---|
| 0 | Rapid depolarization | Fast Na⁺ inward current (INa) — SCN5A |
| 1 | Early repolarization | Transient outward K⁺ current (Ito) |
| 2 | Plateau | L-type Ca²⁺ inward current (ICaL) balanced by K⁺ |
| 3 | Rapid repolarization | Delayed rectifier K⁺ currents (IKr, IKs) |
| 4 | Resting potential / automaticity | IK1, pacemaker funny current (If) |
Disruption at any phase produces abnormal excitability through three main mechanisms:
- Abnormal automaticity — spontaneous depolarization outside the SA node
- Triggered activity — early (EAD) or delayed (DAD) afterdepolarizations
- Re-entry — unidirectional conduction block + slow conduction creating a circus loop
Major Disorders
1. Long QT Syndrome (LQTS)
- Mechanism: Reduced repolarizing K⁺ current (IKr/IKs) or enhanced Na⁺/Ca²⁺ inward current → prolonged AP plateau → EADs → torsades de pointes (TdP)
- Genetics: >17 subtypes; most common:
- LQT1 (KCNQ1, IKs ↓) — triggered by exercise/swimming
- LQT2 (KCNQ2/KCNH2, IKr ↓) — triggered by auditory stimuli/emotions
- LQT3 (SCN5A, INa ↑ — gain-of-function) — events at rest/sleep
- ECG: QTc ≥ 480 ms (symptomatic) or ≥ 500 ms (high risk); T-wave morphology varies by subtype
- Management: β-blockers (esp. LQT1/2), avoid QT-prolonging drugs, ICD for high-risk patients, mexiletine in LQT3
2. Short QT Syndrome (SQTS)
- Mechanism: Gain-of-function K⁺ channel variants → accelerated repolarization → shortened refractory period → AF and VF
- Genetics: KCNH2, KCNQ1, KCNJ2
- ECG: QTc ≤ 340 ms; tall, narrow, symmetrical T-waves
- Management: ICD; quinidine (increases QT)
3. Brugada Syndrome (BrS)
- Mechanism: Loss-of-function SCN5A mutations (INa ↓) → heterogeneous repolarization in RV epicardium → phase 2 re-entry → VF
- ECG: Type 1 pattern — coved ST elevation ≥ 2 mm in V1–V2 (spontaneous or sodium-channel-blocker-provoked)
- Triggers: Fever, large carbohydrate meals, vagotonia, alcohol
- Management: Avoid Na⁺-channel blockers, treat fever aggressively; ICD for symptomatic patients; quinidine, catheter ablation (epicardial RV outflow tract)
4. Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT)
- Mechanism: Abnormal Ca²⁺ release from SR during adrenergic stimulation → DADs → triggered VT
- Genetics:
- RYR2 (ryanodine receptor) — autosomal dominant, ~60%
- CASQ2 (calsequestrin) — autosomal recessive
- ECG (at rest): Normal QT, no structural disease
- Hallmark: Bidirectional or polymorphic VT precipitated by exercise or emotion
- Management: β-blockers (non-selective, e.g., nadolol), flecainide (blocks RyR2), ICD + β-blockers, left cardiac sympathetic denervation (LCSD)
5. Early Repolarization Syndrome (ERS)
- Mechanism: Enhanced Ito in inferior/lateral leads → J-point elevation → dispersion of repolarization → VF
- ECG: J-wave/notching ≥ 1 mm in ≥ 2 inferior or lateral leads
- Management: ICD; quinidine or isoproterenol for electrical storm
6. Progressive Cardiac Conduction Disease (Lenègre–Lev Disease)
- Mechanism: SCN5A loss-of-function → progressive fibrosis and conduction slowing in His-Purkinje system
- ECG: Progressive bundle branch block, high-degree AV block
- Management: Pacemaker
Genetic Overlap and SCN5A Spectrum
The SCN5A gene (encoding Nav1.5) exemplifies how a single gene can produce multiple phenotypes depending on whether the mutation is a gain- or loss-of-function:
| SCN5A Variant | Phenotype |
|---|---|
| Gain-of-function | LQT3 |
| Loss-of-function | BrS, PCCD, sick sinus syndrome |
| Combined | Overlap syndrome |
Pathogenic SCN5A variants also intersect with structural cardiomyopathies — DCM, ARVC, and LVNC — blurring the boundary between pure channelopathy and cardiomyopathy (Arrhythmogenic Cardiomyopathy Guidelines, p. 37).
Diagnostic Approach
Unexplained syncope / resuscitated SCA / family history of SCD
↓
12-lead ECG (resting)
↓
┌─────────────────────────────────┐
│ QTc prolonged? → LQTS workup │
│ QTc short? → SQTS workup │
│ BrS pattern? → BrS confirmed │
│ Normal? → Exercise test │
│ (CPVT, LQTS) │
└─────────────────────────────────┘
↓
Genetic panel (multigene NGS)
↓
Family cascade screening
Key tests: Resting ECG, exercise stress test, sodium-channel provocation (ajmaline/flecainide for BrS), Holter, loop recorder, electrophysiological study (selected cases), cardiac MRI (rule out structural disease), genetic testing.
Risk Stratification
| Disorder | High-Risk Features |
|---|---|
| LQTS | QTc > 500 ms, prior TdP/syncope, LQT2/3, female sex |
| BrS | Spontaneous type 1 ECG, prior VF/syncope, male sex |
| CPVT | Prior cardiac arrest, non-sustained VT on treatment |
| SQTS | Prior VF, family history of SCD |
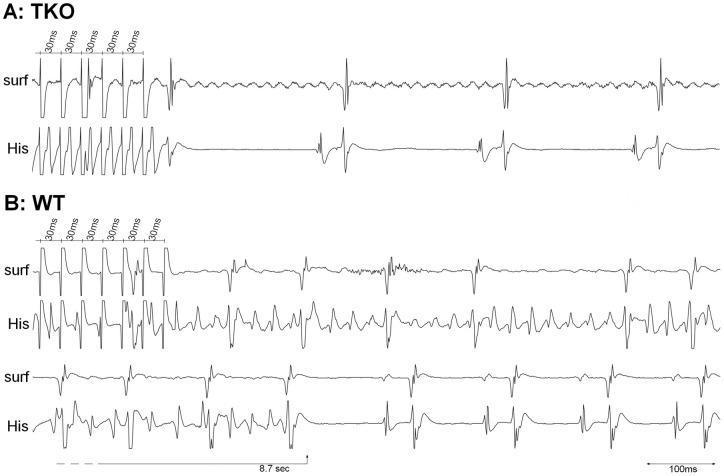
ECG Illustration: Cardiac Excitability and Refractory Periods
The following experimental ECG tracings illustrate differential excitability responses to atrial burst stimulation, with wild-type (WT) mice showing sustained AF while triple knockout (TKO) models revert rapidly to sinus rhythm — demonstrating the role of specific ion channel genotypes in maintaining tachyarrhythmias and modulating refractory periods:
General Management Principles
- Avoid triggers: QT-prolonging drugs (CredibleMeds database), fever, specific sports by disorder
- β-blockers: First-line in LQTS (LQT1/2) and CPVT
- Quinidine: Useful in BrS, SQTS, ERS (Ito blocker)
- Flecainide: Adjunct in CPVT (RyR2 blocker); contraindicated in BrS
- ICD: For secondary prevention (post-arrest) universally; primary prevention based on risk stratification
- Catheter ablation: Epicardial ablation in BrS; trigger ablation in CPVT
- Left cardiac sympathetic denervation (LCSD): CPVT, LQTS refractory to medical therapy
- Genetic counseling + cascade family screening: Essential in all confirmed channelopathies
Key Takeaways
- Cardiac excitability disorders are primarily genetic ion channel diseases causing life-threatening arrhythmias in structurally normal hearts
- The main culprits are Na⁺ (SCN5A), K⁺ (KCNQ1, KCNH2, KCNJ2), and Ca²⁺ (RYR2, CACNA1C) channel genes
- Arrhythmia mechanism (EAD vs. DAD vs. re-entry) dictates both phenotype and treatment choice
- All patients warrant genetic testing and family screening
- Management pivots on trigger avoidance + pharmacotherapy ± ICD, tailored to the specific disorder
This is a shared conversation. Sign in to Orris to start your own chat.