explain anemia with classification (morphological & etiological)
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
anemia classification morphological microcytic normocytic macrocytic RBC
Loading Image
Loading Image
Here is a comprehensive explanation of anemia with both classification systems:
Anemia — Definition & Classification
Definition
Anemia is defined as blood counts (hemoglobin/hematocrit/RBC count) below normal for a given population. Standard WHO thresholds: Hb < 13 g/dL in adult males, < 12 g/dL in adult females, < 11 g/dL in pregnant women.
(Harrison's Principles of Internal Medicine 22E, p. 492)
Two Major Classification Systems
There are two general approaches to classifying anemia, each with distinct clinical utility:
| Approach | Based on | Best use |
|---|---|---|
| Morphological | Red cell size (MCV) | Initial workup, guides differential |
| Etiological/Pathophysiological | Underlying mechanism | Definitive diagnosis, treatment planning |
I. MORPHOLOGICAL CLASSIFICATION (by MCV)
Pioneered by hematologist Max Wintrobe. Groups anemia by mean corpuscular volume (MCV). Less hemoglobin → smaller red cells; thus morphology reflects the synthesis defect.
A. Microcytic Anemia (MCV < 80 fL)
Caused by any process that interferes with hemoglobin production. All four causes affect either heme synthesis or globin synthesis:
| Cause | Mechanism | Key Features |
|---|---|---|
| Iron Deficiency Anemia | Insufficient iron → impaired heme synthesis | Most common worldwide; ↑TIBC, ↓ferritin, ↓serum iron |
| Thalassemia | Defective globin chain synthesis (α or β) | ↑RBC count, targeting on smear, abnormal Hb electrophoresis |
| Anemia of Chronic Disease | Cytokine-mediated iron sequestration (hepcidin↑) | Often normocytic; ↓TIBC, ↑ferritin |
| Sideroblastic Anemia | Defects of heme synthesis (pyridoxine pathway) | Ring sideroblasts on bone marrow; ↑serum iron, ↑ferritin |
| Lead Poisoning | Inhibits δ-aminolevulinic acid dehydratase | Basophilic stippling; ↑erythrocyte protoporphyrin |
(Goldman-Cecil Medicine, Table 144-9)
Differential diagnosis of microcytic anemia by lab:
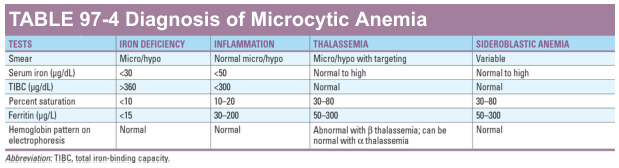
B. Normocytic Anemia (MCV 80–100 fL)
This is the broadest category — a drawback of the morphological scheme. It includes all anemias where RBC production fails without a size change:
| Cause | Notes |
|---|---|
| Aplastic anemia | Marrow failure; pancytopenia |
| Anemia of chronic kidney disease | ↓Erythropoietin production |
| Anemia of inflammation (most cases) | Cytokine-mediated; normocytic in majority |
| Endocrinopathies | Hypothyroidism, adrenal insufficiency, androgen deficiency |
| Marrow invasion/infiltration | Leukemia, myeloma, myelofibrosis |
| Pure red cell aplasia | Selective erythroid suppression |
| Early iron deficiency | Before cells become microcytic |
| Sickle cell disease | Normocytic with sickle forms on smear |
| Physiologic anemia of pregnancy | Dilutional |
C. Macrocytic Anemia (MCV > 100 fL)
Due to defects in DNA synthesis (leading to oval macrocytes) or membrane/metabolic defects (leading to round macrocytes):
Megaloblastic (Oval Macrocytes) — impaired DNA synthesis:
- Vitamin B₁₂ deficiency (pernicious anemia, dietary)
- Folate deficiency
- Medications (methotrexate, hydroxyurea, certain anticonvulsants)
- Myelodysplastic syndrome (MDS)
Non-Megaloblastic (Round Macrocytes) — membrane defects/other:
- Alcohol use disorder (most common cause of macrocytosis)
- Liver disease
- Hypothyroidism
- Reticulocytosis (young RBCs are large)
- Dysproteinemia / hyperlipidemia
- Smoking, hypoxia
(Harrison's Principles of Internal Medicine 22E, p. 492)
II. ETIOLOGICAL / PATHOPHYSIOLOGICAL CLASSIFICATION
Based on the underlying mechanism. The reticulocyte count is the critical first step — it separates hypoproliferative (low retic) from hyperproliferative (high retic) causes.
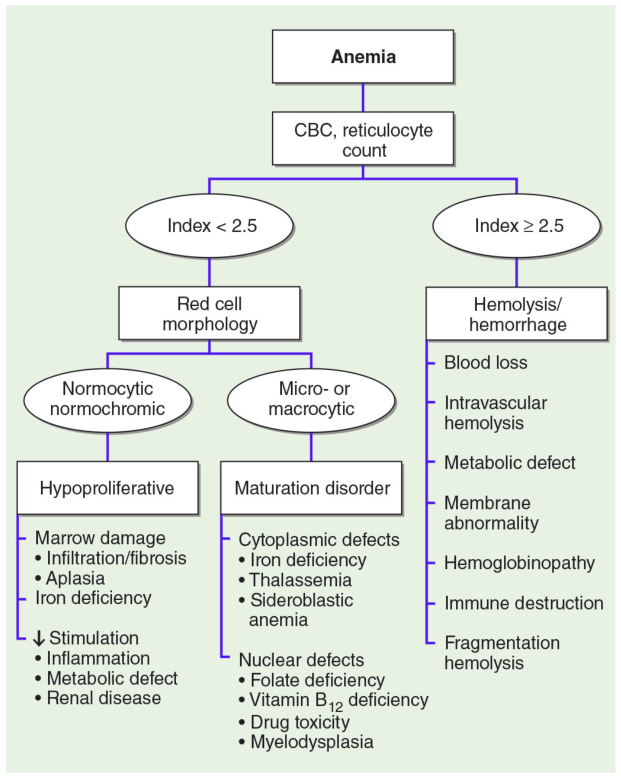
A. BLOOD LOSS
| Type | Examples |
|---|---|
| Acute | Trauma, surgery, GI bleed, rupture |
| Chronic | GI tract lesions (ulcer, carcinoma), gynecologic (menorrhagia), hookworm |
Chronic blood loss eventually leads to iron deficiency.
B. INCREASED RBC DESTRUCTION (Hemolytic Anemias)
Characterized by ↑reticulocyte count, ↑LDH, ↑indirect bilirubin, ↓haptoglobin. Subdivided into intrinsic (inherited/acquired defect within the RBC) vs. extrinsic (external destructive forces):
1. Intrinsic (Intracorpuscular) Defects
a) Red Cell Membrane Disorders
- Hereditary Spherocytosis — mutations in ankyrin, band 3, spectrin, or band 4.2; spherocytes trapped in spleen; splenectomy is curative
- Hereditary Elliptocytosis — spectrin dimer self-association defects
b) Enzyme Deficiencies
- Hexose Monophosphate Shunt: G6PD deficiency (most common; X-linked; episodic hemolysis with oxidant stress), Glutathione synthetase deficiency
- Glycolytic Pathway: Pyruvate kinase (PK) deficiency, Hexokinase deficiency
c) Hemoglobin Abnormalities
- Deficient globin synthesis: Thalassemia syndromes (α and β)
- Structurally abnormal globins (Hemoglobinopathies): Sickle cell disease (HbS), unstable hemoglobins
d) Acquired Intracorpuscular Defect
- Paroxysmal Nocturnal Hemoglobinuria (PNH) — somatic mutation in PIG-A gene → deficiency of GPI-anchored complement regulatory proteins (CD55, CD59) → complement-mediated hemolysis
2. Extrinsic (Extracorpuscular) Defects
a) Immune-Mediated Destruction
- Autoimmune hemolytic anemia (AIHA): warm (IgG, spleen-mediated) and cold (IgM, complement-mediated)
- Hemolytic disease of the newborn (Rh/ABO incompatibility)
- Transfusion reactions
- Drug-induced (hapten, immune complex, or autoantibody mechanisms)
b) Mechanical Trauma
- Microangiopathic hemolytic anemias (MAHA): HUS, TTP, DIC — fibrin strands shear RBCs → schistocytes on smear
- Cardiac traumatic hemolysis: Defective prosthetic valves
- March hemoglobinuria: Repetitive physical trauma (marathon running, karate)
c) Infections of Red Cells
- Malaria (Plasmodium species invade and destroy RBCs)
- Babesiosis
d) Toxic/Chemical Injury
- Oxidant drugs, snake venoms, clostridial toxins, arsenic
e) Sequestration
- Hypersplenism (pooling and destruction in enlarged spleen)
C. DECREASED RBC PRODUCTION (Hypoproliferative/Maturation Defects)
Reticulocyte count is low (inappropriately for degree of anemia).
1. Nutritional Deficiencies
| Deficiency | Effect | Result |
|---|---|---|
| Iron | Impaired heme synthesis | Microcytic hypochromic anemia |
| Vitamin B₁₂ | Impaired DNA synthesis | Megaloblastic macrocytic anemia |
| Folate | Impaired DNA synthesis | Megaloblastic macrocytic anemia |
| Copper | Myelopathy + anemia | Normocytic or macrocytic |
2. Erythropoietin (EPO) Deficiency
- Chronic kidney disease — most important cause; loss of peritubular EPO-producing cells
- Anemia of endocrine deficiency (hypothyroidism, adrenal insufficiency)
3. Bone Marrow Failure / Stem Cell Defects
- Aplastic anemia — immune-mediated destruction of hematopoietic stem cells; pancytopenia; can be congenital (Fanconi anemia, telomerase defects) or acquired
- Pure red cell aplasia (PRCA) — selective suppression of erythroid progenitors (parvovirus B19, thymoma, autoimmune)
- Diamond-Blackfan syndrome — congenital PRCA
4. Marrow Infiltration / Replacement
- Leukemia, lymphoma, multiple myeloma
- Myelofibrosis
- Metastatic carcinoma
- Granulomatous disease (TB, sarcoid)
5. Inflammation-Mediated Iron Sequestration
- Anemia of chronic disease/inflammation — hepcidin ↑ → ferroportin degradation → iron trapped in macrophages → unavailable for erythropoiesis
6. Primary Hematopoietic Neoplasms
- Myelodysplastic syndrome (MDS) — dysplastic erythropoiesis, ineffective hematopoiesis
(Robbins, Cotran & Kumar — Pathologic Basis of Disease, Table 14.1; Harrison's 22E)
Summary Comparison Table
| Feature | Morphological | Etiological |
|---|---|---|
| Basis | MCV (red cell size) | Mechanism / pathophysiology |
| Categories | Microcytic / Normocytic / Macrocytic | Blood loss / Hemolysis / Decreased production |
| Clinical utility | Guides initial workup rapidly | Directs definitive treatment |
| Key tool | MCV on CBC | Reticulocyte count + peripheral smear |
| Limitation | Normocytic group is very broad | Requires more tests to classify |
Key takeaway: In clinical practice, both systems are used together. The MCV gives an immediate morphological direction; the reticulocyte count then separates the etiological mechanism. The peripheral blood smear integrates both — it confirms RBC size/shape and hints at the underlying cause (spherocytes → HS or AIHA; schistocytes → MAHA; hypersegmented neutrophils → megaloblastic; target cells → thalassemia or liver disease).
Make a presentation on hemolytic anemias with mechanisms
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
hereditary spherocytosis peripheral blood smear spherocytes
Searching Images
sickle cell disease blood smear sickle shaped cells
Searching Images
G6PD deficiency Heinz bodies oxidative hemolysis mechanism
Searching Images
microangiopathic hemolytic anemia schistocytes fragmentation TTP HUS
Reading File
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.