Write a notes on hypoxic pulmonary vasoconstiction and effect of Anesthetic agent in it
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have all the content needed for comprehensive notes. Here are the complete notes on Hypoxic Pulmonary Vasoconstriction and the effects of anesthetic agents:
Hypoxic Pulmonary Vasoconstriction (HPV) & Effects of Anesthetic Agents
1. Definition
Hypoxic Pulmonary Vasoconstriction (HPV) is the process by which pulmonary vascular smooth muscle contracts in response to alveolar hypoxia, raising local vascular resistance and redirecting blood flow away from poorly ventilated (hypoxic) lung regions toward better-ventilated ones. This improves ventilation-perfusion (V/Q) matching and preserves arterial oxygenation.
HPV is unique to the pulmonary circulation — in contrast, systemic vascular beds (coronary, cerebral) dilate in response to hypoxia.
— Miller's Anesthesia, 10e
2. Physiological Significance
- Optimizes gas exchange by diverting blood flow from hypoxic to well-ventilated regions
- Critically important in: atelectasis, pneumonia, ARDS, reactive airway disease, and one-lung ventilation (OLV)
- Does not significantly contribute to pulmonary blood flow heterogeneity under normal conditions in supine humans
- In the fetal period, HPV is the primary driver of high pulmonary vascular resistance (PVR), shunting blood away from non-gas-exchanging fetal lungs
- HPV can reduce blood flow to a nonventilated lung by up to 50% during OLV
— Miller's Anesthesia, 10e; Murray & Nadel's Textbook of Respiratory Medicine
3. Stimulus for HPV
| Trigger | Detail |
|---|---|
| Primary stimulus | Alveolar O₂ tension (PAO₂) — NOT arterial PO₂ |
| Threshold | HPV begins when alveolar PO₂ falls below ~60 mmHg |
| Maximum response | At alveolar PO₂ of approximately 30 mmHg |
| Secondary stimulus | Mixed venous PO₂ (P(v̄O₂)) — weaker than PAO₂ |
| Augmenting factors | Hypercapnia, local acidosis (both increase PVR, especially during hypoxia) |
Key point: Perfusing a lung with high-PO₂ blood while keeping alveolar PO₂ low still produces vasoconstriction — confirming alveolar gas is the primary trigger.
4. Temporal (Biphasic) Response
HPV has a biphasic temporal pattern:
- Phase 1 (Rapid): Begins immediately upon hypoxia; plateaus by 20–30 minutes
- Phase 2 (Delayed): Begins after ~40 minutes; reaches maximum plateau after ~2 hours
The offset is also biphasic — PVR may not return to baseline for several hours after prolonged OLV. This may worsen desaturation during bilateral thoracic procedures when the second lung is collapsed.
HPV also exhibits preconditioning: the response to a second hypoxic challenge is greater than to the first.
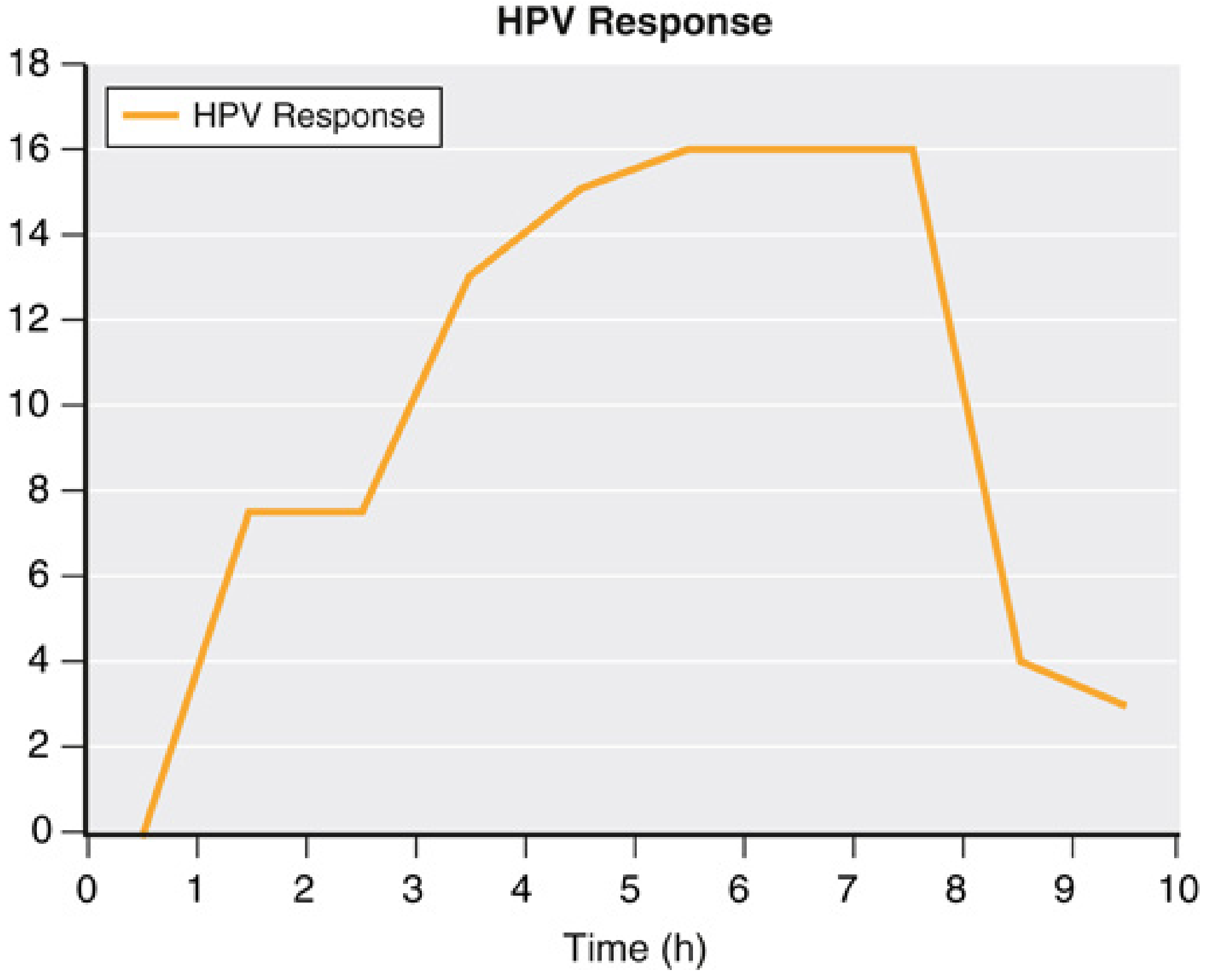
5. Mechanism of HPV
The precise oxygen-sensing mechanism is still incompletely understood but involves multiple pathways:
Cellular Mechanisms
- Voltage-gated K⁺ channel inhibition in pulmonary artery (PA) smooth muscle → membrane depolarization → Ca²⁺ influx
- Ca²⁺ release from sarcoplasmic reticulum (SR) via ryanodine receptors
- Enhanced Ca²⁺ sensitization of contractile proteins
- Connexin 40-mediated retrograde endothelial signal conduction for O₂ sensing
- TRP channel V4 (TRPV4) Ca²⁺ influx at pulmonary arterioles — required for HPV
Endothelial Mediators
- Nitric Oxide (NO): Endothelium-derived relaxing factor; inhibitors of NO synthesis augment HPV; inhaled NO (at ~20 ppm) reduces HPV. High CO₂ reduces NO levels, potentially augmenting HPV.
- Endothelins: Vasoconstrictor peptides released by pulmonary vascular endothelial cells; may participate in hypoxic vasoconstriction
- The primary endothelial hypoxia-mediated vasoconstrictor has not yet been definitively identified
Primary Site
The small pulmonary arteries are the primary site of vasoconstriction. Because smooth muscle is unevenly distributed in normal human lungs, vasoconstriction during global hypoxia (e.g., high altitude) is also uneven — explaining the uneven distribution of high-altitude pulmonary edema.
— Miller's Anesthesia, 10e; Murray & Nadel's Respiratory Medicine
6. Clinical Demonstration of HPV
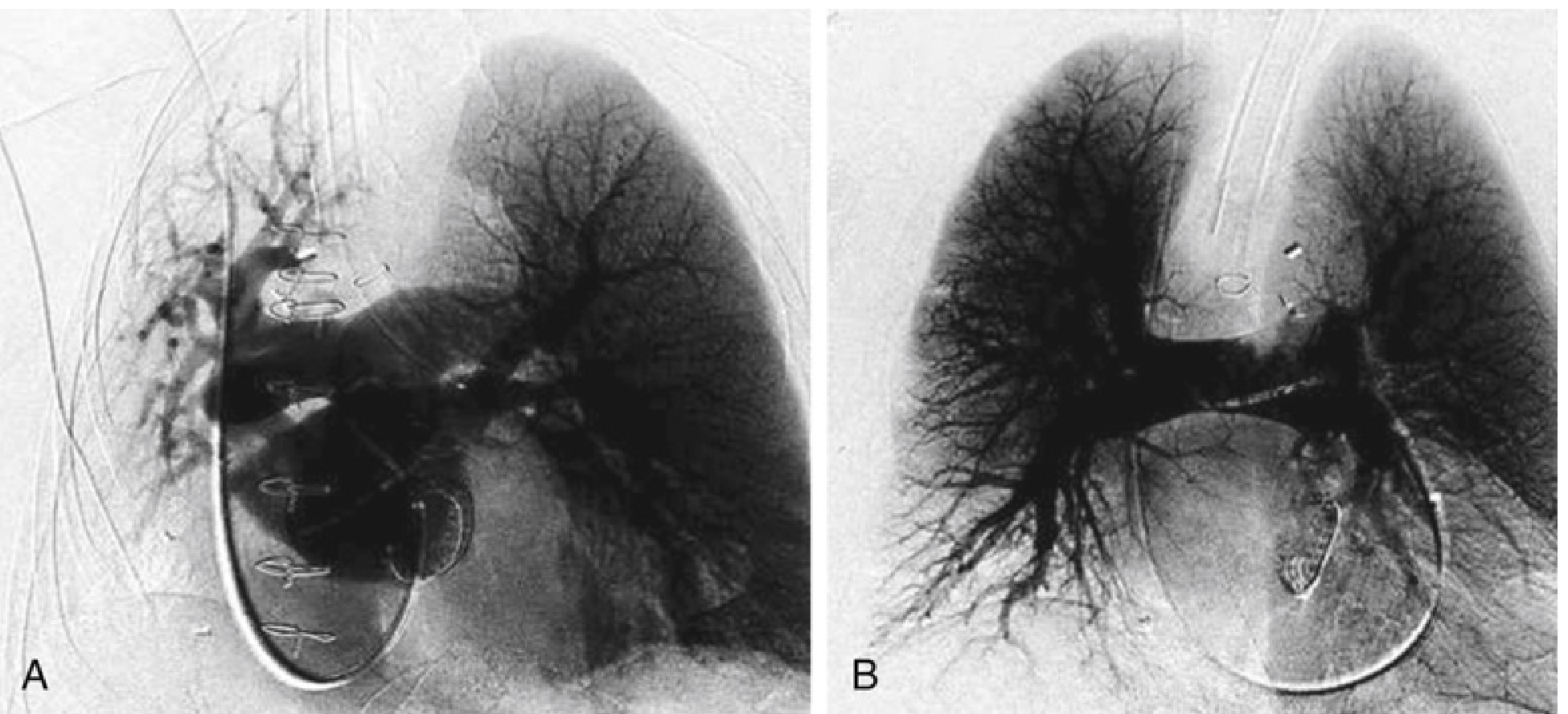
7. Factors That Modify HPV
| Factor | Effect on HPV |
|---|---|
| Hypercapnia / acidosis | Augments HPV |
| Hypothermia | Attenuates HPV |
| Vasodilators (nitroglycerin, nitroprusside) | Inhibit HPV → worsen PaO₂ during OLV |
| Surgical trauma | May oppose HPV via local vasoactive metabolite release |
| Mechanical interference with PA/PV | Can dramatically reduce blood flow to nonventilated lung |
| Thoracic epidural blockade | No direct effect; indirect effect if cardiac output falls |
| Size of hypoxic lung segment | Larger hypoxic areas → greater HPV response |
| Degree of hypoxia | More profound hypoxia → stronger HPV |
8. Effect of Anesthetic Agents on HPV
8.1 Volatile (Inhaled) Anesthetics — General Principle
All volatile anesthetics dilate pulmonary vasculature and attenuate HPV, primarily through:
- Reduction in free cytosolic Ca²⁺
- Inhibition of myofilament Ca²⁺ sensitivity
- Modulation of TASK-1 K⁺ channels in smooth muscle
- Effects via arachidonic acid metabolites
- Endothelium-derived vasodilating factors
In normal lungs, the vasodilatory effect is modest and the small reduction in PVR is usually offset by a concomitant fall in cardiac output — resulting in minor change in pulmonary arterial pressure.
However, when HPV is needed (atelectasis, OLV, pneumonia), inhibition by volatile agents impairs V/Q matching and worsens arterial oxygenation.
— Miller's Anesthesia, 10e; Barash Clinical Anesthesia, 9e
8.2 Individual Volatile Agents
| Agent | Effect on HPV | Notes |
|---|---|---|
| Halothane | Attenuates HPV | Inhibits cGMP accumulation; inhibits KATP channel-mediated endothelium-dependent vasodilation |
| Isoflurane | Attenuates HPV | Acts via Ca²⁺-activated and voltage-sensitive K⁺ channels; reduces HPV and oxygenation during OLV in animal models |
| Sevoflurane | Attenuates HPV (mild) | Attenuation occurs independent of K⁺ channel function; clinical studies in OLV patients show minimal effect on PaO₂ and shunt fraction |
| Desflurane | Attenuates HPV | Reduces mixed venous O₂ saturation and cardiac output during OLV; clinical impact similar to isoflurane |
| Nitrous Oxide (N₂O) | Attenuates HPV | Early studies showed attenuation in animal models; reduces mixed venous O₂ saturation and cardiac output during OLV |
| Xenon | Does not significantly attenuate HPV | Does not reduce mixed venous O₂ saturation or oxygenation during OLV — distinct from other inhaled agents |
Combined volatile + Ca²⁺ channel blocker: Additive inhibition of HPV by up to 40% compared to either drug alone — suggesting different mechanisms of inhibition.
8.3 Intravenous Anesthetics
In contrast to volatile agents, most intravenous anesthetics do not attenuate HPV. This is a clinically important distinction during one-lung ventilation.
| Agent | Effect on HPV |
|---|---|
| Propofol | Does not significantly inhibit HPV; preferred TIVA agent for OLV |
| Ketamine | Does not inhibit HPV |
| Opioids | No direct inhibitory effect on HPV |
One observational study in children with pulmonary hypertension found no significant difference in pulmonary hemodynamics between propofol-based and isoflurane-based anesthesia — suggesting that at clinical doses, the difference may be smaller than animal data implies.
8.4 Why the Clinical Effect Is Smaller Than Expected
Despite animal data showing clear HPV inhibition, clinical studies of patients under OLV during thoracic surgery show only minimal effects of volatile anesthetics on PaO₂ and intrapulmonary shunt (Qs/Qt). This is because:
- Multiple non-pharmacologic confounders impair HPV independently: surgical trauma, temperature changes, pH, PaCO₂, size of the hypoxic segment
- Indirect cardiovascular effects of volatile agents (↓ cardiac output, ↓ mixed venous PO₂) also alter V/Q — not just direct vascular effects
- Elevated PA pressures from other causes can passively overdistend constricted vascular beds and reverse HPV
- Volatile anesthetics have paradoxical initial vasoconstrictor effects (dose-dependent Ca²⁺ release from SR) before the predominant vasodilatory phase
9. Clinical Implications
- One-lung ventilation (OLV): HPV is the primary protective mechanism limiting shunt. Volatile anesthetics may mildly worsen oxygenation. TIVA with propofol is often preferred for thoracic procedures requiring OLV, though modern volatile agents at ≤1 MAC have a small impact in practice.
- Vasodilators (nitroglycerin, nitroprusside, phosphodiesterase inhibitors) given systemically inhibit HPV and can significantly worsen PaO₂ during OLV — use with caution.
- Epidural analgesia: No direct HPV inhibition, but hemodynamic effects (hypotension, ↓ CO) can indirectly worsen oxygenation.
- Bronchodilators in asthma: Some bronchodilators inhibit HPV and may paradoxically lower PaO₂ by increasing blood flow to poorly ventilated areas.
- High altitude/ARDS/Pulmonary hypertension: HPV plays a critical role; anesthetic choices that minimize HPV inhibition are preferred.
Summary Table
| Feature | Detail |
|---|---|
| Primary trigger | Alveolar PO₂ < 60 mmHg |
| Maximum response | Alveolar PO₂ ~30 mmHg |
| Temporal pattern | Biphasic: rapid (20–30 min) + delayed (2 h) |
| Primary site | Small pulmonary arteries |
| Key mechanism | ↓K⁺ channel activity → depolarization → Ca²⁺ influx + SR release |
| Volatile anesthetics | All attenuate HPV (isoflurane > sevoflurane/desflurane > xenon) |
| IV anesthetics | Minimal/no HPV inhibition (propofol, ketamine) |
| Clinical relevance | Especially important during OLV, atelectasis, ARDS |
Sources: Miller's Anesthesia, 2-Volume Set, 10e | Murray & Nadel's Textbook of Respiratory Medicine, 2-Volume | Barash, Cullen & Stoelting's Clinical Anesthesia, 9e | Morgan & Mikhail's Clinical Anesthesiology, 7e
This is a shared conversation. Sign in to Orris to start your own chat.