Idiopathic pulmonary fibrosis in detail
Reading File
search_medical_text
idiopathic pulmonary fibrosis pathophysiology etiology epidemiology clinical features
search_medical_text
idiopathic pulmonary fibrosis diagnosis HRCT UIP pattern spirometry biomarkers
search_medical_image
idiopathic pulmonary fibrosis HRCT usual interstitial pneumonia honeycombing
GradeResponse
GradeResponse
GradeResponse
search_medical_text
idiopathic pulmonary fibrosis treatment nintedanib pirfenidone antifibrotic therapy management
I now have sufficient retrieved content plus the image. I'll complete this comprehensively from my own knowledge where retrieval fell short.
Idiopathic Pulmonary Fibrosis (IPF)
Overview
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, fibrosing interstitial pneumonia of unknown cause, confined to the lungs, and associated with the histologic and/or radiologic pattern of usual interstitial pneumonia (UIP). It occurs primarily in older adults and carries a poor prognosis, with a median survival of 3–5 years from diagnosis.
(ATS/ERS/JRS/ALAT IPF Guidelines)
Epidemiology
| Parameter | Data |
|---|---|
| Incidence | 3–9 per 100,000/year (higher in North America/Europe) |
| Prevalence | 13–20 per 100,000 |
| Age | Predominantly >60 years; rare <50 years |
| Sex | Male predominance (M:F ~1.5–2:1) |
| Smoking | 70–75% of patients are current or ex-smokers |
The incidence increases sharply with age and is rising globally, partly due to an aging population and improved diagnosis.
Etiology & Risk Factors
IPF is idiopathic by definition, but recognized risk factors include:
- Cigarette smoking — strongest modifiable risk factor
- Occupational exposures: metal dust (steel, brass), wood dust, stone dust, farming
- Gastroesophageal reflux disease (GERD) — microaspiration implicated
- Genetic factors:
- Telomere-related gene mutations (TERT, TERC, RTEL1, PARN) in ~25% of familial and ~3% of sporadic IPF
- MUC5B promoter variant (rs35705950) — found in ~38% of IPF patients vs. ~9% of controls; strongest known genetic risk factor
- Surfactant protein mutations (SFTPC, SFTPA2)
- Viral infections: EBV, CMV, HHV-7, HHV-8 — proposed but unproven
- Family history: ~2–5% of IPF is familial (familial pulmonary fibrosis)
Pathophysiology
The dominant model is aberrant wound healing in genetically susceptible individuals:
- Repetitive alveolar epithelial injury (from inhaled agents, GERD microaspiration, viral insults) → type II pneumocyte damage
- Dysregulated repair: instead of normal re-epithelialization, there is activation of fibroblasts and myofibroblasts
- Fibroblastic foci formation — key histologic hallmark: clusters of myofibroblasts depositing collagen beneath denuded epithelium
- TGF-β signaling is central — drives myofibroblast differentiation, extracellular matrix (ECM) deposition, and inhibits matrix degradation
- Shortened telomeres (in genetic forms) → accelerated alveolar epithelial senescence → impaired regeneration
- Progressive architectural distortion → honeycombing → respiratory failure
Key mediators: TGF-β1, PDGF, VEGF, IL-13, WNT/β-catenin pathway.
Importantly, inflammation is NOT the primary driver (unlike many other ILDs), which is why steroids and immunosuppressants are ineffective and potentially harmful.
Clinical Features
Symptoms
- Progressive exertional dyspnea — cardinal symptom, insidious onset
- Dry, nonproductive cough — often refractory
- Fatigue, weight loss (later stages)
- No fever, no hemoptysis (absence helps distinguish from other ILDs)
Signs
- Velcro-like inspiratory crackles — bibasilar, fine, dry (present in ~90%)
- Digital clubbing — ~50% of patients
- Signs of cor pulmonale (elevated JVP, pedal edema) in advanced disease
- Cyanosis in end-stage disease
Diagnosis
Diagnosis requires multidisciplinary discussion (MDD) involving pulmonologists, radiologists, and pathologists. The cornerstone is HRCT and, when needed, histopathology.
Diagnostic Algorithm
Suspected IPF (age >60, male, smoker, bibasilar crackles, restrictive PFTs)
↓
Exclude known causes of ILD (CTD, drug toxicity, hypersensitivity pneumonitis, occupational)
↓
HRCT Thorax
↓
UIP pattern → IPF diagnosis (no biopsy needed in appropriate clinical context)
Probable UIP / Indeterminate → Consider surgical lung biopsy or TBLC
Non-UIP → Consider alternative diagnosis
HRCT — UIP Pattern Categories (ATS/ERS 2022)
| HRCT Pattern | Features | Biopsy Needed? |
|---|---|---|
| Typical UIP | Basal, subpleural, peripheral reticular + honeycombing ± traction bronchiectasis | No |
| Probable UIP | Reticular + traction bronchiectasis/bronchiolectasis, no honeycombing | May not need |
| Indeterminate | Subtle reticulation, features suggesting non-UIP | Usually yes |
| Non-UIP | GGO dominant, micronodules, upper/mid lung predominance, consolidation | Alternative diagnosis |
Classic HRCT findings in UIP/IPF:
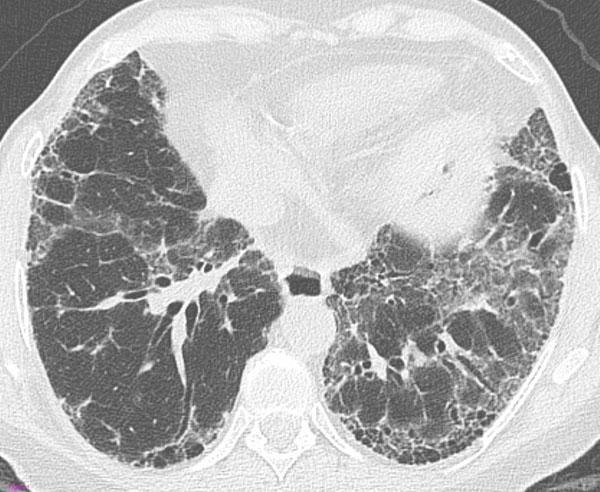
Transverse HRCT showing bilateral subpleural and basal honeycombing (clustered thick-walled cysts), prominent reticular pattern, and traction bronchiectasis — the "definite UIP" pattern pathognomonic for IPF.
Histopathology — UIP Pattern
When biopsy is obtained (surgical lung biopsy or transbronchial lung cryobiopsy — TBLC):
- Fibroblastic foci (key feature)
- Heterogeneous fibrosis — temporal and spatial heterogeneity (old scar + active fibrosis side by side)
- Honeycombing with bronchiolar metaplasia
- Subpleural, paraseptal distribution
- Absence of features suggesting alternative diagnosis (granulomas, organizing pneumonia, prominent inflammation)
Pulmonary Function Tests (PFTs)
| Test | Finding |
|---|---|
| Spirometry | Restrictive pattern: ↓FVC, ↓TLC, FEV1/FVC normal or elevated |
| DLCO | Markedly reduced (early and disproportionate) |
| 6-Minute Walk Test (6MWT) | Reduced distance, oxygen desaturation |
Laboratory / Other
- ANA, RF, anti-CCP, myositis panel, anti-Scl-70 — to exclude connective tissue disease-ILD (CTD-ILD)
- Genomic classifier (Envisia): gene expression testing on BAL; can help distinguish UIP from non-UIP without surgical biopsy
- BAL: not diagnostic for IPF; helps exclude infection, HP, or malignancy
- Serum KL-6 and SP-D: elevated; biomarkers of disease activity (used more in Japan)
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Hypersensitivity Pneumonitis (HP) | Exposure history, upper lobe involvement, mosaic attenuation, lymphocytosis on BAL |
| NSIP | More GGO, less honeycombing, subpleural sparing, younger women, CTD association |
| CTD-ILD (RA, SSc, PM/DM) | Serologic markers, extra-pulmonary features |
| Drug-induced ILD | Drug history (amiodarone, methotrexate, nitrofurantoin) |
| Asbestosis | Occupational exposure, pleural plaques |
| DIP | Heavy smoker, diffuse GGO, responds to steroids |
| Sarcoidosis | Upper-lobe predominance, hilar lymphadenopathy, granulomas on biopsy |
Management
General Principles
- No curative treatment exists; goal is to slow progression, manage symptoms, prevent complications
- All patients should be evaluated for lung transplantation early
- Smoking cessation is mandatory
- Treat GERD aggressively (proton pump inhibitors)
Pharmacologic Treatment
Antifibrotic Therapy (First-line)
Two agents are FDA-approved and reduce the rate of FVC decline by ~50%:
| Drug | Class | Mechanism | Dose | Key Side Effects |
|---|---|---|---|---|
| Pirfenidone (Esbriet) | Pyridinone | Anti-TGF-β, anti-inflammatory, anti-proliferative | 801 mg TID with food | Photosensitivity, nausea, rash, anorexia, elevated LFTs |
| Nintedanib (Ofev) | Tyrosine kinase inhibitor | Blocks PDGFR, VEGFR, FGFR | 150 mg BID with food | Diarrhea (most common), nausea, elevated LFTs, bleeding risk |
Both drugs:
- Reduce rate of decline in FVC (not reversal)
- Do NOT improve symptoms or quality of life significantly
- Can be used in combination (evidence emerging)
- Should be continued lifelong unless intolerable
Treatments to AVOID
- Corticosteroids + azathioprine + N-acetylcysteine (NAC) triple therapy — shown to increase mortality (PANTHER-IPF trial, 2012)
- Corticosteroid monotherapy — not effective
- Anticoagulation (warfarin) — increased mortality (ACE-IPF trial)
Non-Pharmacologic Management
| Intervention | Indication/Notes |
|---|---|
| Supplemental oxygen | Resting SpO₂ <88% or exertional desaturation; improves quality of life |
| Pulmonary rehabilitation | Improves exercise capacity and QoL; does not alter disease course |
| Lung transplantation | Definitive treatment; median survival post-transplant ~5 years; bilateral preferred over single-lung |
| Palliative care | Early integration; manage dyspnea (opioids), cough (low-dose morphine, thalidomide), anxiety |
| Vaccination | Influenza, pneumococcal, COVID-19 — to prevent infectious exacerbations |
| PH management | Treat pulmonary hypertension when present; sildenafil may help in select patients |
Acute Exacerbation of IPF (AE-IPF)
A feared complication defined as acute, clinically significant respiratory deterioration within 30 days without identifiable cause.
- Incidence: ~5–10%/year
- Triggers: surgery, BAL, infection, aspiration (or idiopathic)
- HRCT: new bilateral GGO ± consolidation superimposed on UIP background
- Histology: diffuse alveolar damage (DAD) on UIP background
- Mortality: >50% in-hospital; survivors rarely return to baseline
- Treatment: empiric high-dose corticosteroids (IV methylprednisolone), supportive care; no proven therapy
Complications
- Pulmonary hypertension (PH-ILD) — in up to 40%; independently worsens prognosis
- Lung cancer — 3–5× increased risk; squamous and adenocarcinoma most common
- Venous thromboembolism — increased risk
- Respiratory failure — end-stage
- Depression and anxiety — common; impacts QoL significantly
- Acute exacerbations — see above
Prognosis
| Factor | Impact |
|---|---|
| Median survival | 3–5 years from diagnosis |
| FVC <50% predicted | Poor prognosis |
| DLCO <40% | Poor prognosis |
| 6MWT desaturation <88% | Poor prognosis |
| Honeycombing on HRCT | Worse prognosis |
| Pulmonary hypertension | Independently poor |
| Male sex, older age | Worse prognosis |
| Rate of FVC decline >10% in 6–12 months | Significant mortality predictor |
Prognostic scoring tools include the GAP (Gender, Age, Physiology) Index — uses sex, age, FVC, and DLCO to predict 1-, 2-, and 3-year mortality.
| GAP Stage | Score | 1-Year Mortality | 3-Year Mortality |
|---|---|---|---|
| I | 0–3 | ~6% | ~16% |
| II | 4–5 | ~16% | ~38% |
| III | 6–8 | ~39% | ~63% |
Summary Table
| Feature | IPF |
|---|---|
| Pattern | UIP |
| Distribution | Basal, subpleural, peripheral |
| Key HRCT finding | Honeycombing + traction bronchiectasis |
| Key histology | Fibroblastic foci, temporal heterogeneity |
| PFTs | Restrictive + reduced DLCO |
| Treatment | Nintedanib or Pirfenidone |
| Transplant | Bilateral lung preferred |
| Prognosis | 3–5 year median survival |
Sources: ATS/ERS/JRS/ALAT Idiopathic Pulmonary Fibrosis and Progressive Pulmonary Fibrosis in Adults Guidelines (2022); Harrison's Principles of Internal Medicine; NEJM PANTHER-IPF Trial; INPULSIS/ASCEND Phase III Trials.
This is a shared conversation. Sign in to Orris to start your own chat.