Cirrhosis of the Liver: (Etiology and pathogenesis of liver cirrhosis. Methods for examining patients. Clinical presentation, complications. Surgical treatment
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Loading Image
Loading Image
Loading Image
I now have comprehensive material from Robbins Pathology covering all the requested topics. Let me compile a thorough, well-structured answer.
Cirrhosis of the Liver
1. Definition
Cirrhosis is the diffuse transformation of the entire liver into regenerative parenchymal nodules surrounded by fibrous bands. It is the morphologic endpoint most often associated with chronic liver disease and represents a continuous process involving both progressive fibrosis and, in some cases, partial regression.
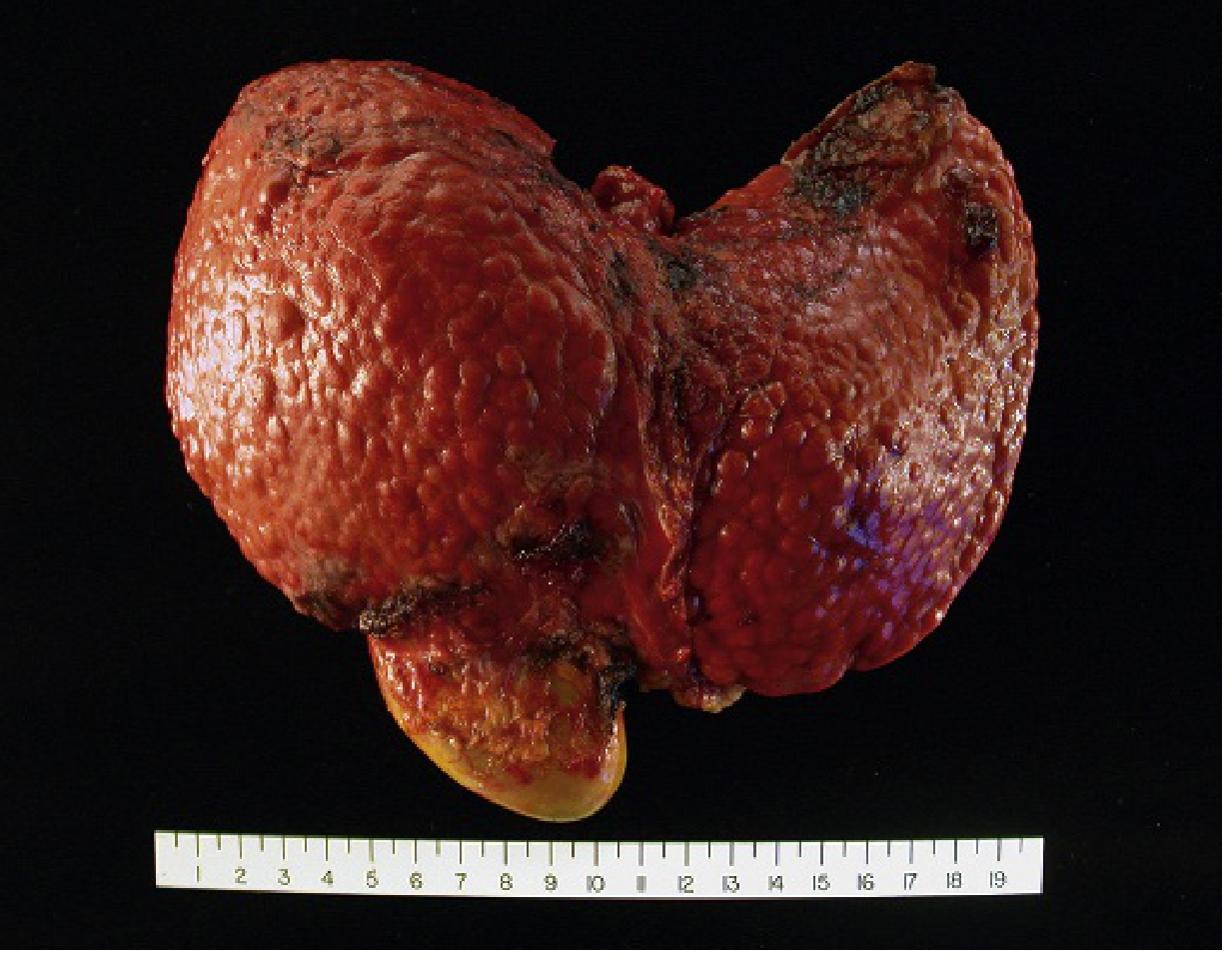
2. Etiology
The leading causes worldwide:
| Cause | Notes |
|---|---|
| Chronic Hepatitis B | Major global cause, especially in Asia and sub-Saharan Africa |
| Chronic Hepatitis C | Leading cause in Western countries prior to direct-acting antivirals |
| Alcohol-related liver disease | Dose-dependent toxicity; requires years of heavy use |
| Non-alcoholic fatty liver disease (NAFLD) | Increasingly prevalent; linked to metabolic syndrome, obesity, T2DM |
| Autoimmune hepatitis | Progressive chronic inflammation |
| Biliary obstruction | Primary biliary cholangitis, primary sclerosing cholangitis, biliary atresia |
| Metabolic/genetic diseases | Hemochromatosis, Wilson disease, α1-antitrypsin deficiency |
| Cryptogenic cirrhosis | No identifiable cause; some may be "burnt-out" NAFLD |
"There are also some instances in which cirrhosis arises without any clear cause; the term cryptogenic cirrhosis is sometimes applied to such cases." — Robbins & Kumar Basic Pathology, p. 598
3. Pathogenesis
3.1 Core Mechanism: Hepatic Stellate Cell Activation
The central event is activation of hepatic stellate cells (Ito cells) in the space of Disse. Under normal conditions these cells store vitamin A and produce small amounts of extracellular matrix. In response to chronic injury they transform into myofibroblasts that:
- Synthesize large amounts of type I and III collagen
- Produce TGF-β, which amplifies fibrogenesis
- Contract, increasing sinusoidal vascular resistance
3.2 Steps in Fibrosis Progression
- Chronic hepatocellular injury (by viral proteins, alcohol/acetaldehyde, lipotoxicity, bile acids, immune-mediated damage)
- Kupffer cell and macrophage activation → cytokine release (TGF-β, PDGF, TNF)
- Stellate cell activation and proliferation → excessive ECM deposition
- Ductular reaction — stem/progenitor cell activation gives rise to ductular structures, particularly prominent in advanced fibrosis
- Disruption of normal lobular architecture → formation of regenerative nodules separated by fibrous septa (portal-to-portal, or portal-to-central bridging)
3.3 Nodule Classification
- Micronodular cirrhosis (nodules < 3 mm): typical of alcohol and biliary disease
- Macronodular cirrhosis (nodules > 3 mm, up to several cm): typical of viral hepatitis
3.4 Reversibility
Cirrhosis is not always irreversible. After disease remission or cure (e.g., cured HCV, abstinence from alcohol), fibrotic bands can thin, compact, and fragment. Adjacent regenerating nodules may coalesce. However, even regressed cirrhosis may leave behind irreversible portosystemic shunts.
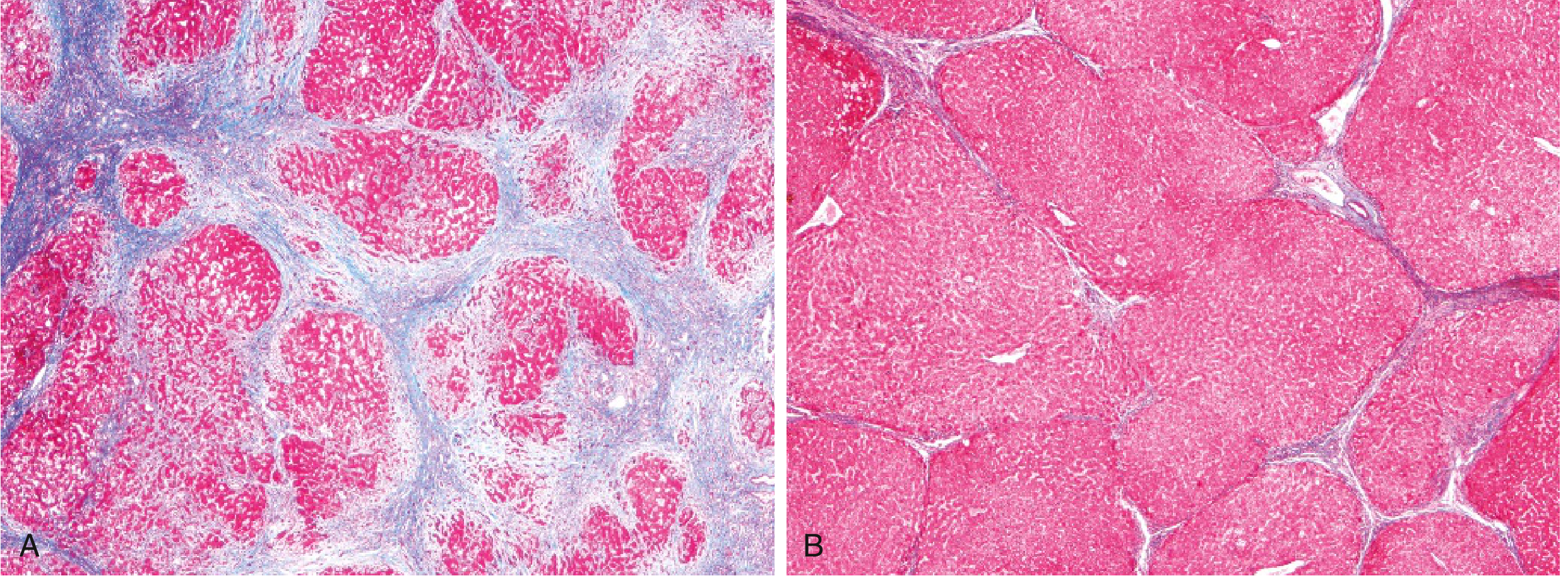
4. Methods for Examining Patients
4.1 History
- Duration and quantity of alcohol consumption
- Risk factors for viral hepatitis (IV drug use, transfusions, sexual contacts)
- Metabolic risk factors (obesity, diabetes, dyslipidaemia)
- Family history (Wilson disease, haemochromatosis, α1-AT deficiency)
- Drug and herbal supplement use
4.2 Physical Examination
- Hepatomegaly (early) or small, shrunken liver (late)
- Splenomegaly (portal hypertension)
- Ascites (shifting dullness, fluid thrill)
- Jaundice, scleral icterus
- Spider angiomas (>5 is significant), palmar erythema
- Caput medusae (dilated periumbilical veins)
- Gynaecomastia, testicular atrophy (hyperestrogenism)
- Dupuytren's contracture (alcohol)
- Asterixis (flapping tremor — hepatic encephalopathy)
- Fetor hepaticus (sweet, musty breath — encephalopathy)
4.3 Laboratory Investigations
| Test | Findings in Cirrhosis |
|---|---|
| LFTs (AST, ALT) | Elevated (may be normal in advanced disease) |
| Bilirubin | Elevated |
| Albumin | Reduced (impaired synthesis) |
| PT/INR | Prolonged (reduced clotting factor synthesis) |
| Platelet count | Low (hypersplenism) |
| FBC | Anaemia, leukopenia, thrombocytopenia |
| Serum Na | Hyponatraemia (dilutional) |
| Creatinine/BUN | Elevated if hepatorenal syndrome |
| AFP | Elevated in HCC |
| Viral serology | HBsAg, anti-HCV, anti-HBc |
| Specific tests | Ferritin/transferrin saturation (haemochromatosis), ceruloplasmin (Wilson), IgG/ANA/ASMA (AIH), AMA (PBC) |
Non-invasive fibrosis scores:
- FIB-4 index (age × AST / platelets × √ALT)
- APRI (AST-to-platelet ratio)
- FibroScan (transient elastography) — liver stiffness measurement in kPa
4.4 Imaging
- Ultrasound (first-line): nodular liver surface, increased echogenicity, splenomegaly, ascites, portal vein diameter, Doppler flow assessment
- CT (triphasic): nodular morphology, varices, HCC detection
- MRI/MRCP: superior HCC characterisation, biliary anatomy
- Endoscopy (EGD): grading of oesophageal/gastric varices — mandatory in all cirrhotic patients at diagnosis
4.5 Liver Biopsy
- Still the gold standard for staging fibrosis (METAVIR, Ishak scales)
- Used when non-invasive testing is inconclusive
- Provides aetiology, activity grade, and fibrosis stage simultaneously
5. Clinical Presentation
5.1 Compensated Cirrhosis
- ~40% of patients are asymptomatic at diagnosis
- Non-specific: fatigue, anorexia, weight loss, malaise
- Detected incidentally on imaging or blood tests
5.2 Decompensated Cirrhosis
Defined by the appearance of one or more complications:
6. Complications
6.1 Portal Hypertension
The central complication driving most others. Results from:
- Increased sinusoidal resistance: contraction of stellate cell myofibroblasts, fibrotic scarring, nodule formation
- Increased portal inflow: arterial vasodilation (NO-mediated) → increased splanchnic blood flow → increased venous return to portal system
Normal portal pressure: 5–10 mmHg. Portal hypertension defined as ≥12 mmHg (below this, varices do not bleed).
6.2 Oesophageal/Gastric Varices
- Develop in ~40% of patients with advanced cirrhosis
- Result from portosystemic collateralisation (left gastric/coronary vein → oesophageal plexus → azygous system)
- Variceal haemorrhage is the most immediately life-threatening complication: massive haematemesis with mortality 15–20% per episode
6.3 Ascites
- Present in ~85% of cases of ascites overall; most common decompensating event in cirrhosis
- Mechanism: portal hypertension → splanchnic vasodilation → reduced effective arterial blood volume → RAAS and SNS activation → Na and water retention → fluid transudation into peritoneum
- Fluid is a transudate: protein < 30 g/L, serum-to-ascites albumin gradient (SAAG) ≥ 1.1 g/dL
- Complicated by spontaneous bacterial peritonitis (SBP): fever, abdominal pain, ascitic PMN count > 250/mm³ — a life-threatening infection (E. coli, Klebsiella most common)
6.4 Hepatic Encephalopathy (HE)
- Spectrum from subtle cognitive impairment (minimal HE) to coma
- Caused by gut-derived toxins (particularly ammonia) reaching systemic circulation via portosystemic shunts and bypassing hepatic detoxification
- Precipitants: GI bleeding, infection, constipation, hyponatraemia, sedatives, large protein load
6.5 Hepatorenal Syndrome (HRS)
- Functional renal failure in the absence of intrinsic renal pathology
- Pathogenesis: hepatic failure → vasodilators (nitric oxide) → splanchnic vasodilation → reduced effective renal perfusion → RAAS/SNS-mediated afferent arteriolar vasoconstriction → GFR falls
- Rising creatinine, oliguria — HRS-1 (rapid, formerly "type 1") carries ~50% 30-day mortality without treatment
6.6 Hypersplenism
- Congestive splenomegaly from portal hypertension → splenic sequestration of blood cells
- Thrombocytopenia, anaemia, leukopenia (pancytopenia)
6.7 Coagulopathy
- Reduced synthesis of factors II, V, VII, IX, X, fibrinogen (all except factor VIII, produced by endothelium)
- Prolonged PT/INR — increased bleeding risk
6.8 Hepatopulmonary Syndrome (HPS)
- Intrapulmonary vascular dilatations → right-to-left shunting → hypoxaemia
- Classic finding: platypnoea (dyspnoea worsening when upright, improving supine)
6.9 Hepatocellular Carcinoma (HCC)
- Most chronic liver diseases predispose to HCC development
- Risk particularly high in HBV (even without cirrhosis), HCV, alcohol-related, NAFLD-related cirrhosis, haemochromatosis
- Surveillance: AFP + liver ultrasound every 6 months in all cirrhotic patients
6.10 Hepatic Hydrothorax
- Transudative pleural effusion (usually right-sided) due to passage of ascitic fluid through diaphragmatic defects
7. Staging and Prognosis
Child-Pugh Score
| Parameter | 1 point | 2 points | 3 points |
|---|---|---|---|
| Bilirubin (µmol/L) | < 34 | 34–51 | > 51 |
| Albumin (g/L) | > 35 | 28–35 | < 28 |
| PT prolongation (s) | < 4 | 4–6 | > 6 |
| Ascites | None | Mild | Tense |
| Encephalopathy | None | Grade 1–2 | Grade 3–4 |
- Class A (5–6): compensated; 1-year survival ~100%
- Class B (7–9): significant compromise
- Class C (10–15): decompensated; 1-year survival ~45%
MELD Score
- MELD = 9.57 × ln(creatinine) + 3.78 × ln(bilirubin) + 11.2 × ln(INR) + 6.43
- Used for liver transplant waitlist prioritisation (higher score = higher urgency)
- MELD ≥ 15: benefit from transplantation outweighs risk
8. Surgical Treatment
Surgical and interventional approaches address both complications and the underlying disease.
8.1 Management of Variceal Haemorrhage
Acute bleed:
- Haemodynamic resuscitation, blood transfusion (target Hb 70–80 g/L)
- Vasopressin analogues (terlipressin) or somatostatin analogues (octreotide) to reduce portal inflow
- Prophylactic antibiotics (norfloxacin or ceftriaxone) to prevent SBP and reduce mortality
- Urgent endoscopic variceal ligation (EVL) — first-line endoscopic treatment, superior to sclerotherapy
- Balloon tamponade (Sengstaken-Blakemore tube) as bridge to definitive treatment
Refractory/recurrent haemorrhage:
Transjugular Intrahepatic Portosystemic Shunt (TIPS)
- Interventional radiology procedure: metallic stent placed between hepatic vein and portal vein via transjugular approach
- Reduces portal pressure directly
- Highly effective for refractory variceal bleeding and refractory ascites
- Main complication: worsening hepatic encephalopathy (blood bypasses liver)
- Acts as a bridge to liver transplantation
Surgical Portosystemic Shunts (when TIPS not available or fails)
- Distal splenorenal shunt (Warren shunt): selective decompression of gastrosplenic varices while preserving mesenteric flow to the liver — lower encephalopathy rate than total shunts
- Mesocaval shunt: superior mesenteric vein to inferior vena cava
- Portocaval shunt (end-to-side or side-to-side): high effectiveness but high encephalopathy rate; largely replaced by TIPS
- Shunt surgery is generally reserved for Child-Pugh Class A/B patients with adequate hepatic reserve; contraindicated in advanced disease
8.2 Surgical Management of Ascites
- Peritoneovenous shunts (LeVeen/Denver shunt): now rarely performed due to complications (DIC, shunt thrombosis)
- Large-volume paracentesis (LVP): with albumin infusion (8 g/L ascites removed) — standard of care for tense/refractory ascites
- TIPS: effective for refractory ascites — reduces need for repeated paracentesis
8.3 Splenectomy / Partial Splenic Embolisation
- Occasionally performed for severe hypersplenism causing refractory cytopenias
- Usually avoided as a standalone procedure in cirrhosis due to operative risk and potential acceleration of portal hypertension
8.4 Liver Transplantation — Definitive Treatment
The only curative intervention for end-stage cirrhosis.
Indications:
- Decompensated cirrhosis (Child-Pugh C or MELD ≥ 15)
- Complications refractory to medical management (SBP, HRS, HE)
- HCC within Milan criteria (single nodule ≤ 5 cm, or up to 3 nodules none > 3 cm, no macrovascular invasion, no extrahepatic spread)
Absolute contraindications:
- Active extrahepatic malignancy
- Uncontrolled infection/sepsis (except SBP)
- Severe cardiac or pulmonary disease
- Active alcohol/drug use (requires documented abstinence period)
- Irreversible neurological disease
Types:
- Orthotopic liver transplantation (OLT) from deceased donor — standard
- Living donor liver transplantation (LDLT) — right lobe donation; reduces waitlist time
- Split liver transplantation — one donor liver for two recipients
Outcomes: 1-year survival ~85–90%; 5-year survival ~70–80% in most aetiologies.
Post-transplant immunosuppression: Tacrolimus ± mycophenolate ± corticosteroids; lifelong.
Summary Table: Surgical Options by Complication
| Complication | First-line Intervention | Surgical/Procedural Option |
|---|---|---|
| Variceal haemorrhage (acute) | EVL + vasoactive drugs | TIPS (refractory); portosystemic shunt |
| Refractory ascites | Repeated LVP + albumin | TIPS; peritoneovenous shunt (rarely) |
| Hypersplenism | Observation | Partial splenic embolisation; splenectomy |
| End-stage liver disease | Medical optimisation + MELD listing | Liver transplantation |
| HCC in cirrhosis | Ablation/TACE (bridge) | Resection (Child A); transplantation (Milan criteria) |
Sources: Robbins & Kumar Basic Pathology (9780323790185), pp. 597–602; Brenner and Rector's The Kidney (9780323532655); Goldman-Cecil Medicine (9780323930345)
This is a shared conversation. Sign in to Orris to start your own chat.