12 Surgical Diseases of the Lungs and Pleura: (Lung abscess and gangrene. Bronchiectasis. (Classification, clinical presentation, diagnosis, and treatment). Spontaneous pneumothorax. Acute purulent pleuritis. Pyoneumothorax. Chronic empyema of the pleura. (Clinical presentation, diagnosis, treatment). 4 Survey. Patient demonstration. Case st
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Here is a comprehensive structured review of all topics in this surgical lecture:
Surgical Diseases of the Lungs and Pleura
1. Lung Abscess
Definition
A lung abscess is a localized area of pulmonary parenchymal necrosis caused by an infectious organism, resulting in a solitary or dominant cavity ≥2 cm in diameter. Necrosis with multiple smaller cavities (<2 cm) is termed necrotizing pneumonia. An abscess present >6 weeks is considered chronic. — Schwartz's Principles of Surgery, 11e
Lung Gangrene
Lung gangrene (pulmonary gangrene) represents the most severe form — massive necrosis of an entire lobe or lung, usually caused by highly virulent organisms (e.g., Klebsiella, S. aureus, mixed anaerobes) and associated with vascular thrombosis. It is distinguished from simple abscess by its extent, the presence of sloughing necrotic tissue within a large cavity, and profound systemic toxicity.
Classification
| Type | Features |
|---|---|
| Primary | No underlying structural disease; arises from aspiration (most common), necrotizing pneumonia, or immunodeficiency |
| Secondary | Occurs in context of bronchial obstruction (neoplasm, foreign body), septic pulmonary emboli, or pulmonary infarct |
| Acute | <6 weeks duration |
| Chronic | >6 weeks; associated with thick wall, poor response to medical therapy |
Primary causes:
- Aspiration (anesthesia, stroke, alcohol/drugs, esophageal disease — achalasia, GERD, Zenker's diverticulum)
- Necrotizing pneumonia (S. aureus, Klebsiella, Pseudomonas, Bacteroides, Fusobacterium, Actinomyces)
- Immunodeficiency (cancer, diabetes, transplant, steroids)
Secondary causes: bronchial obstruction by neoplasm or foreign body; septic emboli; trauma.
Microbiology
Most are polymicrobial and anaerobic. Dominant organisms (aspiration-related): Peptostreptococcus, Prevotella, Bacteroides (not B. fragilis), Fusobacterium. Aerobic pathogens: Streptococcus anginosus, S. aureus, Klebsiella pneumoniae, H. influenzae type b. In immunocompromised patients: Pseudomonas aeruginosa, Nocardia, Aspergillus, Cryptococcus. — Sabiston Textbook of Surgery
Clinical Presentation
- Insidious onset mimicking pneumonia (weeks to months)
- Fever, cough, purulent foul-smelling sputum (often large volume — "mouthful of pus" when abscess drains into a bronchus)
- Systemic features: night sweats, weight loss, anemia, malaise
- Hemoptysis (may be severe)
- Pleuritic chest pain
- Clubbing (in chronic cases)
- On examination: dullness, bronchial breathing, amphoric breath sounds over the cavity
Diagnosis
- CXR: density/mass with a thin-walled cavity ± air-fluid level (indicates communication with the tracheobronchial tree); typically in dependent segments (superior segment of lower lobe, posterior segment of upper lobe)
- CT chest: confirms cavity, distinguishes parenchymal from pleural collections, identifies associated mass, endobronchial obstruction, or adjacent pathology — preferred imaging
- Bronchoscopy: essential to rule out endobronchial obstruction (tumor, foreign body); bronchoalveolar lavage for culture
- Sputum culture (limited by oropharyngeal contamination)
- Percutaneous transthoracic FNA (ultrasound or CT-guided): for culture when bronchoscopy not feasible
Differential diagnosis: Cavitating carcinoma (most important), loculated empyema, infected lung cyst/bulla, tuberculosis, bronchiectasis, fungal infection, Wegener's granulomatosis.
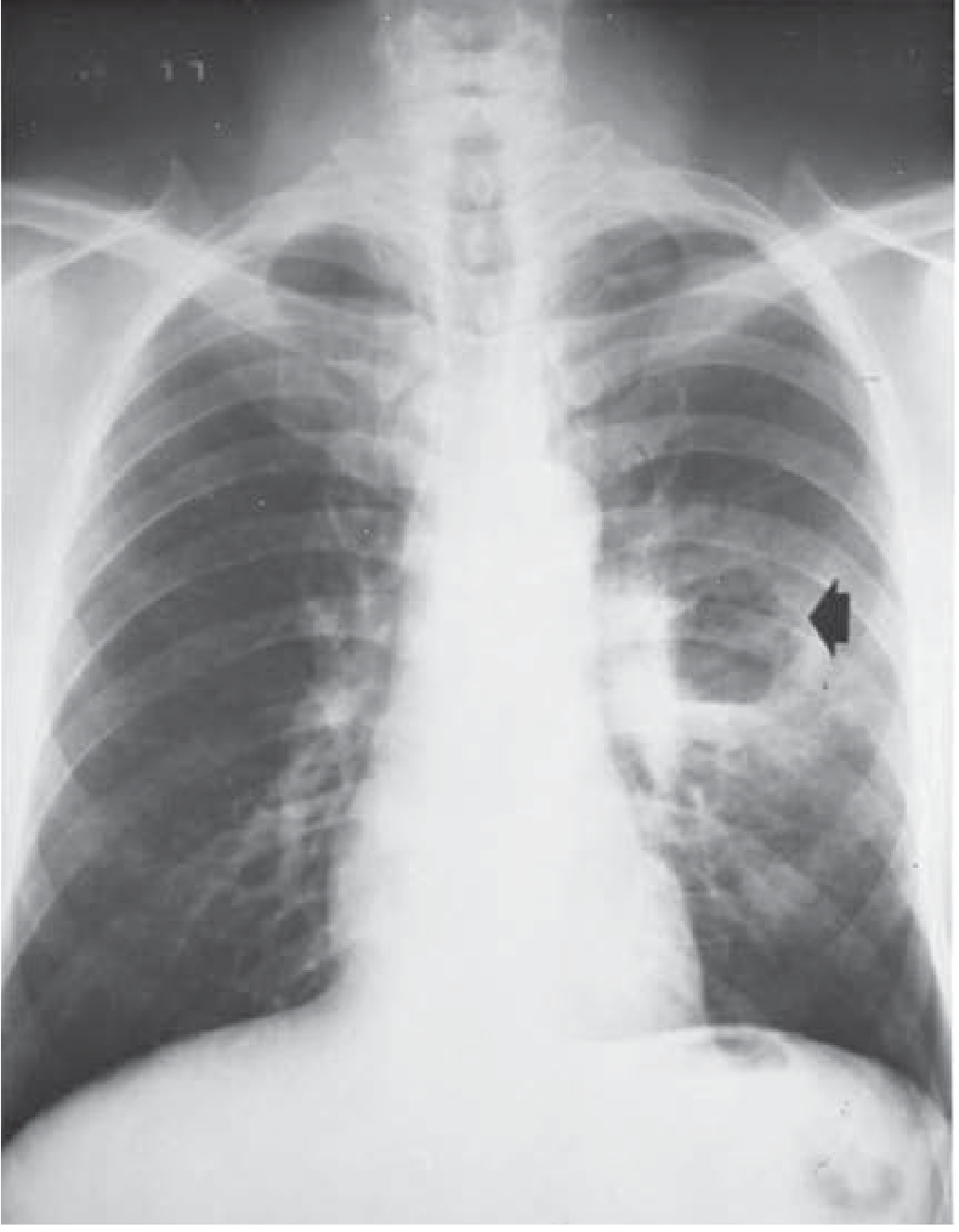
CXR: lung abscess cavity with air-fluid level (arrow) in the left upper lobe — Schwartz's, p. 735
Treatment
Medical (85–95% success):
- IV ampicillin-sulbactam (first-line); alternatives: β-lactam/β-lactamase inhibitor combinations, carbapenems
- Duration: 3–8 weeks (treat until cavity resolves on imaging)
- Postural drainage
- Bronchoscopy for drainage assistance (transbronchial catheterization)
Surgical indications:
- Failure of medical therapy (no improvement after 8 weeks)
- Persistent cavity (≥2 cm, thick-walled)
- Abscess under tension or increasing in size during treatment
- Cavity >4–6 cm in diameter
- Necrotizing infection with multiple abscesses, hemoptysis, abscess rupture, or pyopneumothorax
- Contralateral lung contamination
- Inability to exclude cavitating carcinoma
Surgical procedure: Lobectomy (preferred). Percutaneous drainage (tube thoracostomy or CT-guided catheter) reserved for poor surgical candidates. If abscess ruptures into the pleural space → empyema/pyopneumothorax management. Surgical treatment success rate: 90%; mortality: 1–13%. — Schwartz's Principles of Surgery, 11e
2. Bronchiectasis
Definition
Permanent, pathologic dilation of bronchi with bronchial wall thickening, resulting from recurrent infection and inflammation destroying the bronchial wall. May be localized or diffuse, typically affecting medium-sized airways. — Schwartz's, 11e
Classification
By morphology (Reid classification):
| Type | Description |
|---|---|
| Cylindrical (tubular) | Uniform dilation; mildest form |
| Varicose | Irregular dilation with focal constrictions |
| Saccular (cystic) | Balloon-like outpouchings; most severe |
By etiology:
- Congenital: Cystic fibrosis, primary ciliary dyskinesia (Kartagener syndrome), immunoglobulin deficiencies (IgA, IgG), α1-antitrypsin deficiency
- Acquired — Infectious: Post-necrotizing pneumonia (S. aureus, Klebsiella), pertussis, measles, TB, nontuberculous mycobacteria, allergic bronchopulmonary aspergillosis (ABPA)
- Acquired — Obstructive: Bronchial cancer, foreign body, external compression
- Acquired — Inflammatory/Systemic: Rheumatoid arthritis, inflammatory bowel disease, Sjögren's, GERD with aspiration
- Toxic inhalation: Ammonia, other gases
- Idiopathic: Most common
Clinical Presentation
- Chronic cough with copious purulent sputum (daily; "3-layer sputum" on standing)
- Hemoptysis (can be massive — due to hypertrophied bronchial arteries)
- Dyspnea, pleuritic chest pain
- Fatigue, weight loss
- Recurrent pulmonary infections
- Physical: coarse crackles/wheezes over affected areas; clubbing
- Pulmonary function: obstructive or mixed pattern
Diagnosis
- CT chest (gold standard): shows dilated airways, "signet ring sign" (bronchus diameter > adjacent pulmonary artery), airway wall thickening, mucus plugging, lack of bronchial tapering
- CXR: "tramlines" (parallel bronchial wall markings), cyst clusters, atelectasis
- Sputum cultures: common organisms — H. influenzae, P. aeruginosa, S. aureus, NTM
- PFTs: obstructive or mixed pattern
- Blood tests: autoimmune markers (rheumatoid factor, ANA), immunoglobulin levels, sweat chloride test (if CF suspected)
Treatment
Medical (mainstay):
- Antibiotics: targeted to culture results; inhaled tobramycin or gentamicin; macrolides (azithromycin) — reduce exacerbations
- Airway clearance: chest physiotherapy, postural drainage, oscillating devices, mucolytics
- Treat underlying disease (ABPA → steroids + antifungals; immunodeficiency → IVIG)
- Bronchodilators for airflow obstruction
- Vaccination (influenza, pneumococcal)
Surgical (for localized disease refractory to medical therapy):
- Lobectomy or segmentectomy: for localized, symptomatic disease with maximal medical therapy failure; must exclude multifocal disease and uncorrectable predisposing factors
- Indications: significant hemoptysis (bronchial artery embolization preferred first), localized disease causing recurrent infections
- Bilateral lung transplant: end-stage diffuse disease
3. Spontaneous Pneumothorax
Definition
Air in the pleural space without preceding trauma or iatrogenic cause.
Classification
| Type | Description |
|---|---|
| Primary (PSP) | No clinically apparent underlying lung disease; due to rupture of apical subpleural bleb |
| Secondary (SSP) | Occurs in setting of underlying lung disease |
Primary PSP: Most common in tall, thin, young postadolescent males; smokers. Caused by rupture of apical subpleural blebs (etiology unknown). Recurrence rate: 25–50% after first episode.
Secondary SSP causes: Emphysema (rupture of bleb/bulla — most common), cystic fibrosis, AIDS, asthma, lung abscess, metastatic cancer (especially sarcoma), lung cancer, Langerhans cell histiocytosis. Catamenial pneumothorax (rare) — occurs within 72 hours of menses onset in women aged 20–30 years; related to pleural endometriosis. — Schwartz's, 11e
Clinical Presentation
- Sudden onset ipsilateral pleuritic chest pain and dyspnea
- Physical: decreased breath sounds, hyperresonance on affected side
- Tracheal deviation away from side (tension pneumothorax)
- Hypoxia
Diagnosis
- CXR (upright, expiratory): absent lung markings; visible pleural line
- CT: identifies blebs/bullae, determines extent; guides management
Treatment
Primary PSP, small (<2–3 cm rim), minimal symptoms:
- Observation ± supplemental O₂ (accelerates reabsorption)
- Needle aspiration
Primary PSP with symptoms or complete collapse:
- Chest tube insertion with water-seal drainage
Recurrence or persistent air leak (>3 days):
- VATS (video-assisted thoracoscopic surgery): bleb resection + pleurodesis (talc or pleural abrasion/mechanical scarification) — gold standard
- Indications for surgery on first episode: occupational hazard (pilots, divers), remote location, CT showing multiple blebs, complete lung collapse
Secondary SSP: Management similar (drainage + re-expansion required), plus management of underlying disease; may require thoracoscopic pleurectomy, talc pleurodesis, or lung resection. — Schwartz's, 11e
4. Acute Purulent Pleuritis and Pyopneumothorax
Acute Purulent Pleuritis (Acute Empyema)
Definition & Pathogenesis
Empyema = collection of purulent fluid within the pleural space. Results from invasion by pyogenic bacteria, fungi, or mycobacteria.
Routes of infection:
- Contiguous spread (50–60%): pneumonia (most common — parapneumonic empyema), lung abscess, mediastinal or subphrenic infection
- Direct inoculation (30–40%): penetrating trauma, thoracentesis, postoperative complications, esophageal perforation
- Hematogenous (<1%)
Stages (Light's Classification)
| Stage | Characteristics | Treatment |
|---|---|---|
| Exudative (Stage I) | Thin, free-flowing fluid; low LDH, normal pH; no loculation | Antibiotics + thoracentesis or small-bore drain |
| Fibrinopurulent (Stage II) | Turbid/purulent fluid; fibrin strands; loculations forming; pH <7.2, glucose <60, LDH >1000 | Chest tube drainage + antibiotics ± fibrinolytics (TPA/DNase) |
| Organizing/Chronic (Stage III) | Thick peel; lung entrapment (trapped lung); fibrosis; bronchial tree encasement | VATS decortication (early) or thoracotomy + decortication |
Microbiology
- Gram-positive: Streptococcus pneumoniae, S. aureus (most common)
- Gram-negative: E. coli, Klebsiella, Pseudomonas, Enterobacteriaceae
- Anaerobes (associated with aspiration, dental disease): Streptococcus spp., Bacteroides
- Polymicrobial in up to 50% of cases
Clinical Presentation
- Fever, malaise, loss of appetite, weight loss
- Pleuritic chest pain, dyspnea
- Decreased breath sounds, dullness to percussion
- Tracheal deviation (large collections)
- Signs of sepsis in severe cases
Diagnosis
- CXR: pleural opacity ± loculation; fluid level if associated with bronchopleural fistula
- CT chest: delineates anatomy, identifies loculations, helps plan drainage
- Thoracentesis: pleural fluid analysis — turbid/purulent fluid; pH <7.2, glucose <60 mg/dL, LDH >1000 IU/L → empyema; Gram stain + culture
Treatment
- Antibiotics: broad-spectrum (cover gram-positive, gram-negative, anaerobes); guided by culture
- Drainage: mandatory
- Early stage: chest tube (tube thoracostomy) or pigtail catheter
- Remove chest tube when drainage <100–200 mL/day, imaging shows resolution, clinical improvement
- Intrapleural fibrinolytics (tissue plasminogen activator [tPA] + DNase): aid drainage in Stage II; not routine for complex empyemas (expensive, less effective than thought)
- VATS: early for complex/loculated effusions — reduces hospital stay, achieves good lung expansion
- Thoracotomy + decortication: organized empyema with thick peel; lung entrapment preventing re-expansion — Sabiston Textbook of Surgery
Pyopneumothorax
Definition: Simultaneous presence of pus and air in the pleural space.
Causes:
- Rupture of a lung abscess into the pleural space (most common)
- Bronchopleural fistula complicating empyema
- Penetrating trauma with pleural contamination
- Postoperative (post-pneumonectomy)
Clinical features: Acute presentation with sudden chest pain, dyspnea, high fever, and signs of tension physiology if the bronchial communication is large. Air-fluid level on CXR is diagnostic.
Treatment:
- Emergency chest tube drainage (pleural decompression + pus drainage)
- Broad-spectrum antibiotics
- Bronchoscopy: assess for bronchopleural fistula
- Surgery: lobectomy if pyopneumothorax arises from lung abscess; decortication and fistula closure for established bronchopleural fistula — Schwartz's, 11e
5. Chronic Empyema of the Pleura
Definition
Empyema is considered chronic when it fails to resolve after >3–6 weeks of treatment and enters the organizing phase, characterized by lung entrapment by a thick fibrous cortex (pleural peel).
Pathophysiology
Fibroblast and capillary ingrowth into the fibrinous exudate → formation of an inelastic pleural peel that encases the lung → trapped lung → inability to re-expand → restrictive respiratory impairment. — Sabiston, 11e
Clinical Presentation
- Persistent or low-grade fever
- Continued dyspnea and reduced exercise tolerance
- Persistent pleural opacity on imaging
- Weight loss, malnutrition
- Clubbing, chest wall deformity (scoliosis toward affected side) in long-standing cases
- Chronic sinus tracts (empyema necessitans — tracking to chest wall)
- Amyloidosis (very chronic, rare)
Diagnosis
- CXR: fixed pleural opacity; may show thickened pleural peel; mediastinal shift toward affected side
- CT chest (essential): thickness of pleural peel, underlying lung parenchyma status, residual pleural cavity size, associated bronchial abnormalities
- Pulmonary function tests: restrictive pattern
- Pleural fluid analysis (if accessible): pH, glucose, LDH, culture
Treatment
Goals: Eliminate infection, obliterate the pleural space, allow lung re-expansion, restore function.
| Approach | Indication |
|---|---|
| Antibiotics | All patients; duration guided by clinical response |
| Open drainage (Eloesser flap / open window thoracostomy) | Poor surgical candidates; acts as long-term drainage |
| VATS decortication | Early organizing phase (peel not yet fully calcified); yields good re-expansion if lung parenchyma viable |
| Thoracotomy + decortication | Thick calcified peel; lung entrapment; most effective for full lung re-expansion |
| Muscle flap / omentoplasty | Obliterate residual space after decortication; repair bronchopleural fistula |
| Thoracoplasty | Last resort; collapses chest wall to obliterate space when lung cannot re-expand |
VATS vs. open decortication: VATS is preferred for early organizing empyema — reduced hospital length of stay and morbidity. Thoracotomy is required for mature, calcified peels. — Sabiston Textbook of Surgery
Summary Table
| Disease | Key Diagnosis | Key Treatment |
|---|---|---|
| Lung abscess | CT: thin-walled cavity + air-fluid level | Antibiotics (ampicillin-sulbactam); lobectomy if fails |
| Lung gangrene | CT: massive lobar necrosis | Aggressive antibiotics + surgical resection |
| Bronchiectasis | CT: signet-ring sign, non-tapering airways | Medical (ABx, physio); lobectomy for localized disease |
| Spontaneous pneumothorax | CXR: visible pleural line | Tube thoracostomy; VATS for recurrence |
| Acute purulent pleuritis | Thoracentesis: pH <7.2, pus | Chest tube + antibiotics; VATS/decortication |
| Pyopneumothorax | CXR/CT: air-fluid level in pleural space | Chest tube drainage; lobectomy if abscess rupture |
| Chronic empyema | CT: thick pleural peel, trapped lung | Decortication (VATS or thoracotomy) |
Sources: Schwartz's Principles of Surgery 11e; Sabiston Textbook of Surgery; Mulholland and Greenfield's Surgery 7e
This is a shared conversation. Sign in to Orris to start your own chat.