Liquid oxygen
Liquid oxygen (LOX) is oxygen in its liquid state, achieved by cooling oxygen gas below its boiling point.
Key Properties
| Property | Value |
|---|---|
| Boiling point | −183 °C (−297 °F, 90 K) |
| Melting point | −218.8 °C (−361.8 °F, 54.4 K) |
| Density | 1.141 g/cm³ (at boiling point) |
| Color | Pale blue |
| Magnetic behavior | Paramagnetic (weakly attracted to magnets) |
Production
LOX is produced industrially via the Linde process (fractional distillation of liquefied air):
- Air is compressed and cooled repeatedly until it liquefies
- Liquid air is fractionally distilled — nitrogen (bp −196 °C) boils off first, leaving oxygen-enriched liquid
- Further distillation yields high-purity LOX (99.5%+)
Uses
Rocketry & Aerospace — LOX is the most common oxidizer in liquid-propellant rockets. It's paired with:
- Liquid hydrogen (LH₂) — highest specific impulse (e.g., Space Shuttle main engines, Saturn V upper stage)
- Kerosene/RP-1 — denser and easier to handle (e.g., Falcon 9, Saturn V first stage)
- Liquid methane — emerging choice (e.g., SpaceX Raptor, Rocket Lab Archimedes)
Medical — High-purity oxygen for hospitals, often stored cryogenically and vaporized for use.
Industrial — Steel production (basic oxygen steelmaking), chemical manufacturing, water treatment.
Submarine / Life support — Stored compactly and vaporized as needed.
Handling & Hazards
- Cryogenic burns: Contact with skin causes frostbite instantly
- Oxidizer risk: Vigorously accelerates combustion; materials that burn slowly in air can ignite violently in LOX
- Pressure buildup: If confined, vaporizing LOX generates enormous pressure (expansion ratio ~860:1 liquid to gas)
- Asphyxiation / enrichment: LOX spills can create oxygen-enriched atmospheres, increasing fire/explosion risk
- Materials used with LOX must be LOX-compatible (e.g., stainless steel, aluminum, Teflon) — hydrocarbons and organic materials are incompatible
Storage
Stored in vacuum-jacketed cryogenic dewars or insulated tanks. Even with ideal insulation, some boil-off is inevitable. Large launch facilities manage continuous LOX replenishment right up to launch.
Liquid oxygen and liquid ventilation In anesthesia
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Here is a comprehensive overview of both topics as they relate to anesthesia and critical care, drawn from authoritative textbook sources.
Liquid Oxygen in Anesthesia
Hospital Supply System
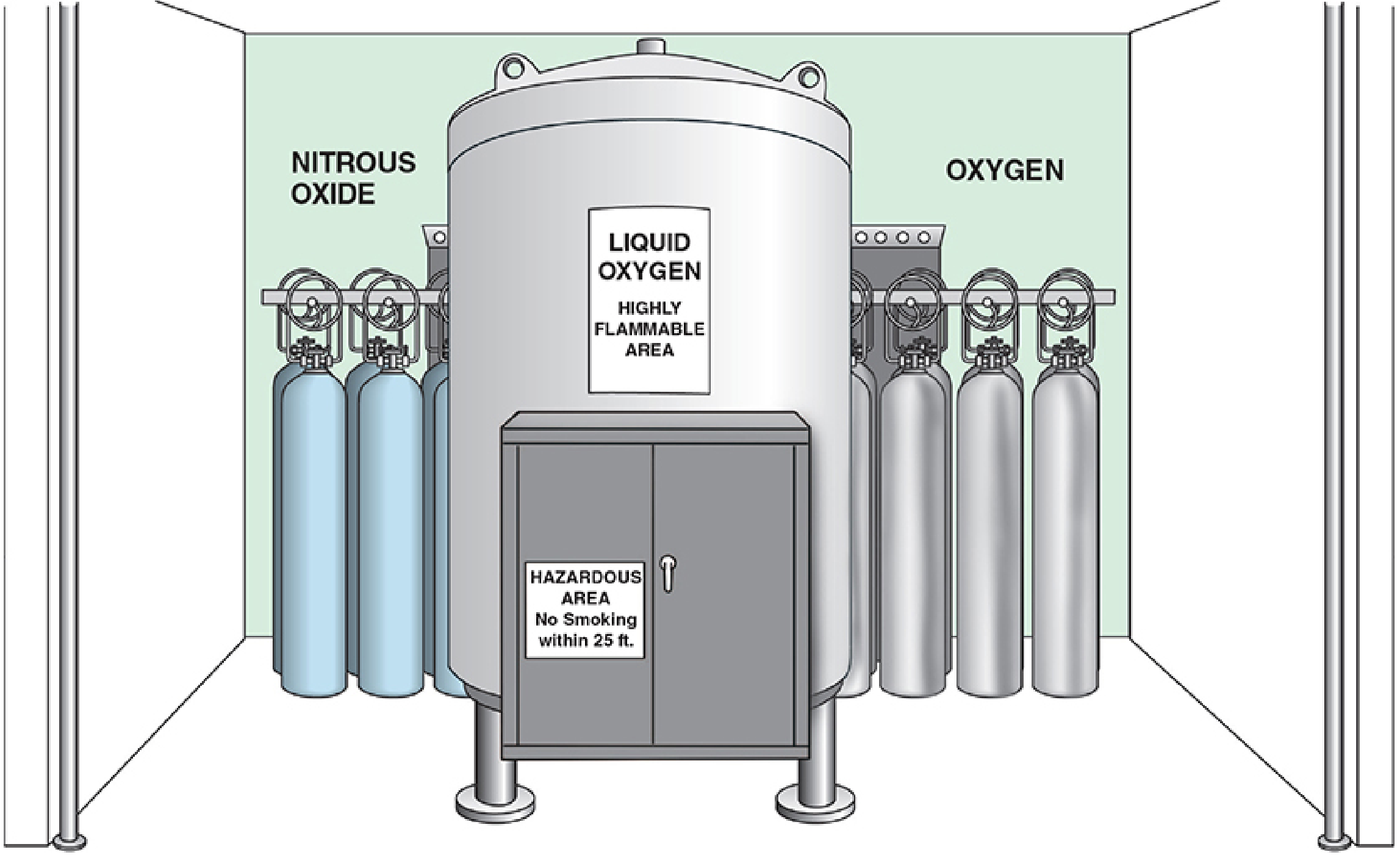
Liquid oxygen (LOX) is the preferred oxygen storage method for large hospitals due to its economy and compactness. Key points from the anesthesia literature:
- LOX must be stored below its critical temperature of −119°C — above this, no amount of pressure can liquefy it
- Large hospitals maintain a bulk liquid oxygen tank with a smaller backup cylinder bank capable of supplying one day's requirement in case of system failure
- Small hospitals typically use two banks of high-pressure H-cylinders connected by a manifold, with automatic switchover when one bank is exhausted
- Before use, the LOX is vaporized and delivered via pipeline at a line pressure of 55 ± 5 psig
- Medical grade oxygen is 99–99.5% pure, manufactured by fractional distillation of liquefied air
Anesthesia Machine Safety Considerations
- Every anesthesia machine must have an E-cylinder (emergency compressed oxygen cylinder) available as a backup against pipeline failure
- The pin index safety system prevents inadvertent connection of wrong gas types
- Oxygen E-cylinders contain a Wood's metal plug (low melting point) for pressure relief in fire
- A pressure of 1000 psig in an E-cylinder = ~330 L of oxygen (approximately half full)
- Oxygen supply failure alarms and protection devices are mandated safety features on modern anesthesia machines
Medical Gas Therapy — Oxygen Indications
Supplemental oxygen is indicated when:
- PaO₂ < 60 mmHg or SpO₂ < 90% on room air (adults and children > 1 month)
- PaO₂ < 50 mmHg or SaO₂ < 88% in neonates
Other therapeutic uses include heliox (upper airway obstruction), hyperbaric oxygen (gas embolism, CO poisoning), and nitric oxide (pulmonary hypertension).
Liquid Ventilation
Liquid ventilation is a distinct concept — using oxygen-dissolving liquids (not liquid oxygen itself) to ventilate the lungs.
Mechanism
- Relies on perfluorocarbons (PFCs) — modified hydrocarbons where hydrogen atoms are replaced with fluorine
- PFCs are inert, nontoxic, and minimally absorbed through the respiratory epithelium
- The most studied agent: perflubron (perfluorooctyl bromide)
- Dissolves ~17× more O₂ than saline
- Dissolves ~4× more CO₂ than saline
Types
| Type | Description |
|---|---|
| Total liquid ventilation | Lungs completely filled with PFC; extracorporeal exchanger oxygenates and removes CO₂ |
| Partial liquid ventilation (PLV) | Lungs partially filled with PFC; conventional ventilator delivers gas tidal volumes on top |
Theoretical Benefits
- Improved lung recruitment — PFCs have lower surface tension than air, opening atelectatic alveoli
- Dependent distribution — liquid flows to dependent lung zones where collapse and injury are greatest
- Secretion clearance — displaced by the liquid
- Anti-inflammatory effects — perflubron has demonstrated anti-inflammatory properties in animal models
Clinical Evidence (ARDS)
Two major randomized controlled trials of partial liquid ventilation in adults with ARDS have been conducted:
- 2002 trial (n=90): PLV with perflubron vs. conventional ventilation — no significant difference in ventilator-free days or any prespecified outcome
- 2006 multicenter trial (n>300): PLV vs. conventional lung-protective ventilation — trend toward higher mortality in the PLV group, plus higher rates of barotrauma, hypoxia, and hypotension
Conclusion: Partial liquid ventilation cannot be recommended for ARDS based on current evidence.
Other Applications Under Investigation
- Therapeutic hypothermia reversal: Warmed oxygenated perfluorocarbon instillation is being studied in animal models as a method of rapidly rewarming the core in severe hypothermia, with shorter rewarming times compared to warm humidified oxygen alone
Summary Table
| Feature | Liquid Oxygen | Liquid Ventilation |
|---|---|---|
| What it is | O₂ gas stored in liquid form at −183°C | Perfluorocarbon liquid used to fill/partial-fill lungs |
| Role in anesthesia | Bulk hospital O₂ supply; vaporized before delivery | Experimental respiratory support in ARDS/hypothermia |
| Clinical status | Standard of care | Not recommended (clinical trials failed) |
| Key agent | Oxygen (LOX) | Perflubron (perfluorooctyl bromide) |
| Key hazard | Cryogenic burns, fire acceleration | Barotrauma, hypoxia, hypotension in trials |
Sources: Morgan and Mikhail's Clinical Anesthesiology, 7e; Murray & Nadel's Textbook of Respiratory Medicine; Harrison's Principles of Internal Medicine 22E
This is a shared conversation. Sign in to Orris to start your own chat.