Explain estrogen to me from speroff
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have enough comprehensive content from Goodman & Gilman's and Berek & Novak's to give a thorough answer. Let me compile this.
Important Note on Sources
Speroff's Clinical Gynecologic Endocrinology and Infertility is not in this library's textbook collection. The most equivalent authoritative sources available are Goodman & Gilman's Pharmacological Basis of Therapeutics (Chapter 48) and Berek & Novak's Gynecology, both of which cover estrogen in comparable depth. What follows draws from those texts.
Estrogens — A Comprehensive Overview
Chemistry & Structure
Estrogens are steroid hormones. The three principal naturally occurring estrogens in humans are 17β-estradiol (most potent), estrone, and estriol — all sharing two key structural features:
- A phenolic A ring with a hydroxyl group at C-3
- A β-OH or ketone at C-17 of ring D
The phenolic A ring is the principal structural determinant for high-affinity binding to both estrogen receptors (ERα and ERβ). Alkyl substitutions on the A ring impair receptor binding, whereas substitutions on rings C or D are tolerated. Ethinyl substitution at C-17 (as in ethinyl estradiol) greatly increases oral potency by inhibiting first-pass hepatic metabolism.
— Goodman & Gilman's, p. 979
Biosynthesis
Steroidal estrogens arise from androstenedione or testosterone via aromatization of the A ring, catalyzed by the enzyme CYP19 (aromatase).
In premenopausal women, the ovaries are the principal source of circulating estrogen, with estradiol as the main secretory product. Ovarian biosynthesis requires two cell types:
- Theca cells: LH acts via adenylyl cyclase–cAMP signaling to drive cholesterol into mitochondria and produce androgen precursors (androstenedione, testosterone)
- Granulosa cells: FSH stimulates CYP19 expression, which aromatizes the androgen precursors to estrogens
In the liver, 17β-hydroxysteroid dehydrogenase (type II) oxidizes circulating estradiol to estrone; both are then converted to estriol. All three are excreted in urine as glucuronide and sulfate conjugates.
In postmenopausal women, the principal source shifts to adipose tissue stroma, where estrone is synthesized from DHEA (adrenal origin) via aromatization.
In men, most circulating estrogens come from extragonadal aromatization of androstenedione and DHEA, not directly from the testes.
— Goodman & Gilman's, p. 979–980
Estrogen Receptors
There are three classes of estrogen receptors:
1. Nuclear Receptors: ERα and ERβ
Both are members of the nuclear receptor superfamily and function as ligand-activated transcription factors.
| Feature | ERα | ERβ |
|---|---|---|
| Gene | ESR1, chromosome 6 | ESR2, chromosome 14 |
| Primary locations | Uterus, vagina, ovaries, mammary gland, hypothalamus, endothelium, vascular smooth muscle | Prostate, ovaries, lung, brain, bone |
| Role in breast cancer | Predominant form driving growth regulation | When co-expressed with ERα, can inhibit ERα-mediated transcription |
| AF-1 domain | Functional | Not functional |
The two ERs share 44% overall amino acid identity but have high homology in their DNA-binding domains, so both recognize similar estrogen response elements (EREs) and regulate many of the same target genes. They exist as multiple mRNA isoforms from differential promoter usage and alternative splicing.
2. G Protein-Coupled Estrogen Receptor (GPER / GPR30)
A membrane-localized receptor that may mediate rapid (non-genomic) effects of estrogen. Its precise physiological role remains unsettled — GPER knockout mice lack reproductive dysfunction but show altered physiological functions consistent with modulatory estrogen signaling.
— Goodman & Gilman's, p. 982–983
Mechanism of Action
After entering the cell by passive diffusion, estrogen binds to ER in the nucleus, where it sits as an inactive monomer bound to heat shock protein 90 (HSP90). Estrogen binding:
- Dissociates HSP90 → causes conformational change in ER
- Promotes receptor dimerization (homodimers ERα/ERα, ERβ/ERβ, or heterodimers ERα/ERβ)
- Dimer binds to estrogen response elements (EREs) in promoter regions of target genes
- Recruits coactivator proteins → assembles general transcription apparatus → initiates transcription
Estrogen also regulates genes lacking a consensus ERE through protein-protein interactions with transcription factors like Sp1 and AP-1 already bound to their own elements. These interactions are ERα/ERβ isoform-specific — for example, 17β-estradiol induces transcription via ERα/AP-1 but inhibits transcription via ERβ/AP-1.
The conformation of the ER's AF-2 domain determines whether coactivators or corepressors are recruited — this is the molecular basis for how SERMs (selective estrogen receptor modulators) work.
— Goodman & Gilman's, p. 983
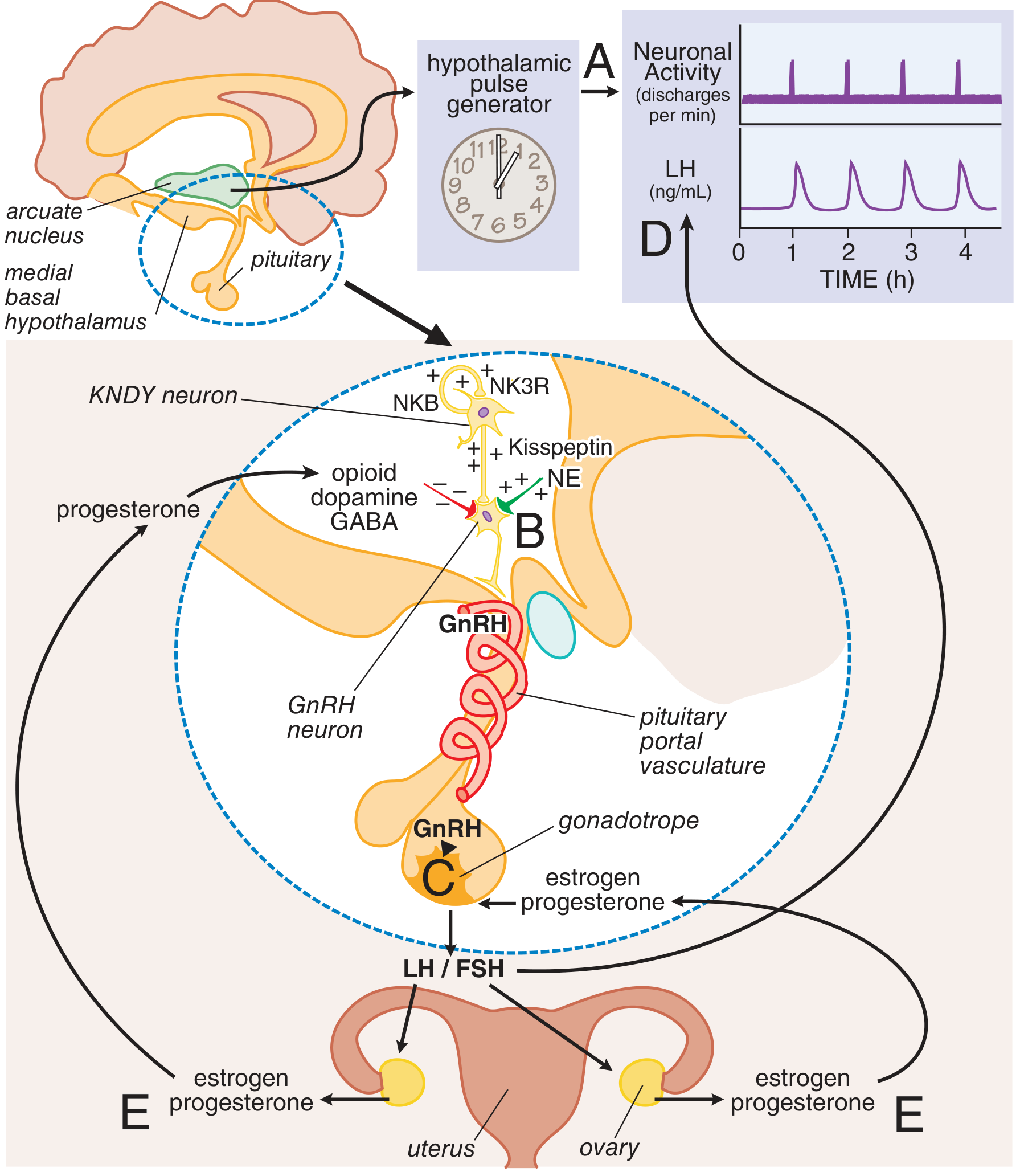
Neuroendocrine Axis: Estrogen's Role in the Menstrual Cycle
A hypothalamus → pituitary → ovary cascade drives the cycle:
- A neuronal oscillator in the arcuate nucleus fires periodically, triggering pulsatile GnRH release into the hypothalamic-pituitary portal circulation
- GnRH acts on pituitary gonadotropes to release LH and FSH (pulsatile)
- LH/FSH drive follicular maturation and ovarian estrogen/progesterone production
- Ovarian steroids exert feedback on the pituitary and hypothalamus
Kisspeptin neurons in the arcuate nucleus are critical regulators:
- Estrogen reduces kisspeptin production in these neurons (negative feedback)
- Neurokinin B stimulates kisspeptin secretion
- Inactivating mutations in GPR54 (kisspeptin receptor) → hypogonadotropic hypogonadism; activating mutations → central precocious puberty
The LH surge (positive feedback by estrogen at the pituitary) triggers ovulation near mid-cycle.
— Goodman & Gilman's, p. 981–982
Physiological Effects
Reproductive Tract
- Drives growth of the uterus, fallopian tubes, vagina, and external genitalia at puberty
- Causes proliferation of the endometrium (proliferative phase)
- Increases vaginal blood flow and maintains vaginal epithelial thickness and lubrication
- Maintains the cervical os open and mucus thin/spinnbarkeit in the follicular phase
Breast
- Stimulates ductal growth and stromal development
- ERα is the predominant driver of breast cancer cell growth
- Estrogen depletion → breast atrophy
Bone
- Critical for bone conservation in both sexes
- Estrogen deficiency → increased osteoclastic bone resorption → osteoporosis
- Postmenopausal estrogen replacement reduces hip fracture risk by ~34% (WHI trial data) — Goodman & Gilman's, p. 982; Berek & Novak's, p. 947
Cardiovascular / Metabolic Effects
- Increases hepatic production of coagulation factors II, VII, IX, X, XII
- Decreases anticoagulants: protein C, protein S, antithrombin III
- Decreases PAI-1 → increases fibrinolysis
- Net effect: both procoagulant and profibrinolytic pathways are upregulated — imbalance can cause adverse thromboembolic events — Goodman & Gilman's, p. 982
Genitourinary Atrophy (Estrogen Deficiency)
- Thinning and loss of elasticity of vaginal epithelium (genitourinary syndrome of menopause / GSM)
- Predisposes to vulvar vaginitis and UTIs
- Reduced vaginal lubrication → dyspareunia
- Postmenopausal provoked vestibulodynia (PVD) may coexist with GSM — Berek & Novak's, p. 895
SERMs — Selective Estrogen Receptor Modulators
SERMs exploit the conformational basis of ER signaling to achieve tissue-selective agonist/antagonist effects:
| SERM | Bone | Uterus | Breast | Vasomotor |
|---|---|---|---|---|
| Raloxifene | Agonist (↓ resorption) | Inactive (no proliferation) | Antagonist (↓ ER+ cancer) | Worsens hot flashes |
| Tamoxifen | Agonist | Agonist (↑ endometrial proliferation) | Antagonist | Minimal effect |
| Fulvestrant | — | Antagonist | Pure antagonist + degrades ERα | — |
| Clomiphene | — | — | Antagonist at pituitary | Induces ovulation |
Raloxifene's agonist activity in bone but not endometrium reflects differential expression of coactivators/corepressors across tissues. Fulvestrant both blocks and degrades ERα, explaining its efficacy in tamoxifen-resistant breast cancers.
— Goodman & Gilman's, p. 983–985
Clinical Applications of Estrogen
- Menopausal hormone therapy (MHT) — short-term relief of vasomotor symptoms; also prevents osteoporosis (FDA-approved indication)
- Contraception — as ethinyl estradiol in combined oral contraceptives
- Induction of sexual maturation in hypogonadal adolescents
- Genitourinary syndrome of menopause — local (vaginal) estrogen preparations restore epithelium and lubrication
- Osteoporosis prevention — even very low-dose estradiol (0.014 mg/day transdermal) increases BMD
WHI trial caveats: HRT (conjugated equine estrogens + MPA) significantly increased risks of coronary heart disease, breast cancer, and stroke; therefore MHT is reserved for symptom control at the lowest effective dose for the shortest duration.
Key takeaway: Estrogen is a phenolic steroid hormone acting primarily through nuclear receptors (ERα, ERβ) as transcription factors, with tissue-selective actions exploited by SERMs. Its roles span reproductive cyclicity, bone maintenance, cardiovascular metabolism, and urogenital integrity — with deficiency producing well-characterized sequelae from menopause.
Sources: Goodman & Gilman's Pharmacological Basis of Therapeutics, Chapter 48; Berek & Novak's Gynecology, Chapters 18 & 34
This is a shared conversation. Sign in to Orris to start your own chat.