Endometriosis
Reading File
Finding Sources
Searching Images
endometriosis pathology laparoscopy
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive overview of endometriosis, synthesized from authoritative textbooks.
Endometriosis
Definition
Endometriosis is defined by the presence of endometrial glands and stroma outside the uterus. It affects up to 10% of women in their reproductive years and nearly half of women with infertility. — Robbins & Kumar Basic Pathology
Classification (Three Types)
| Type | Description |
|---|---|
| Superficial peritoneal | Implants on peritoneal surfaces |
| Ovarian endometriosis | "Chocolate cysts" (endometriomas) |
| Deep infiltrating endometriosis (DIE) | Penetrates >5 mm below peritoneum; highest malignant transformation risk |
Pathogenesis
The exact mechanism remains elusive. Four leading theories exist:
- Regurgitation (Sampson's theory) — Retrograde flow of menstrual endometrium through the fallopian tube implants at ectopic sites (the dominant theory)
- Benign metastasis theory — Endometrial tissue spreads hematogenously or via lymphatics to distant sites (lung, bone, brain)
- Metaplastic theory — Coelomic (peritoneal) epithelium undergoes metaplasia into endometrial tissue, arising from the same müllerian duct precursors
- Extrauterine stem/progenitor cell theory — Bone marrow-derived stem cells differentiate into ectopic endometrial tissue
Once implanted, endometriotic tissue is not merely misplaced endometrium — it actively perpetuates itself through a cycle of inflammation:
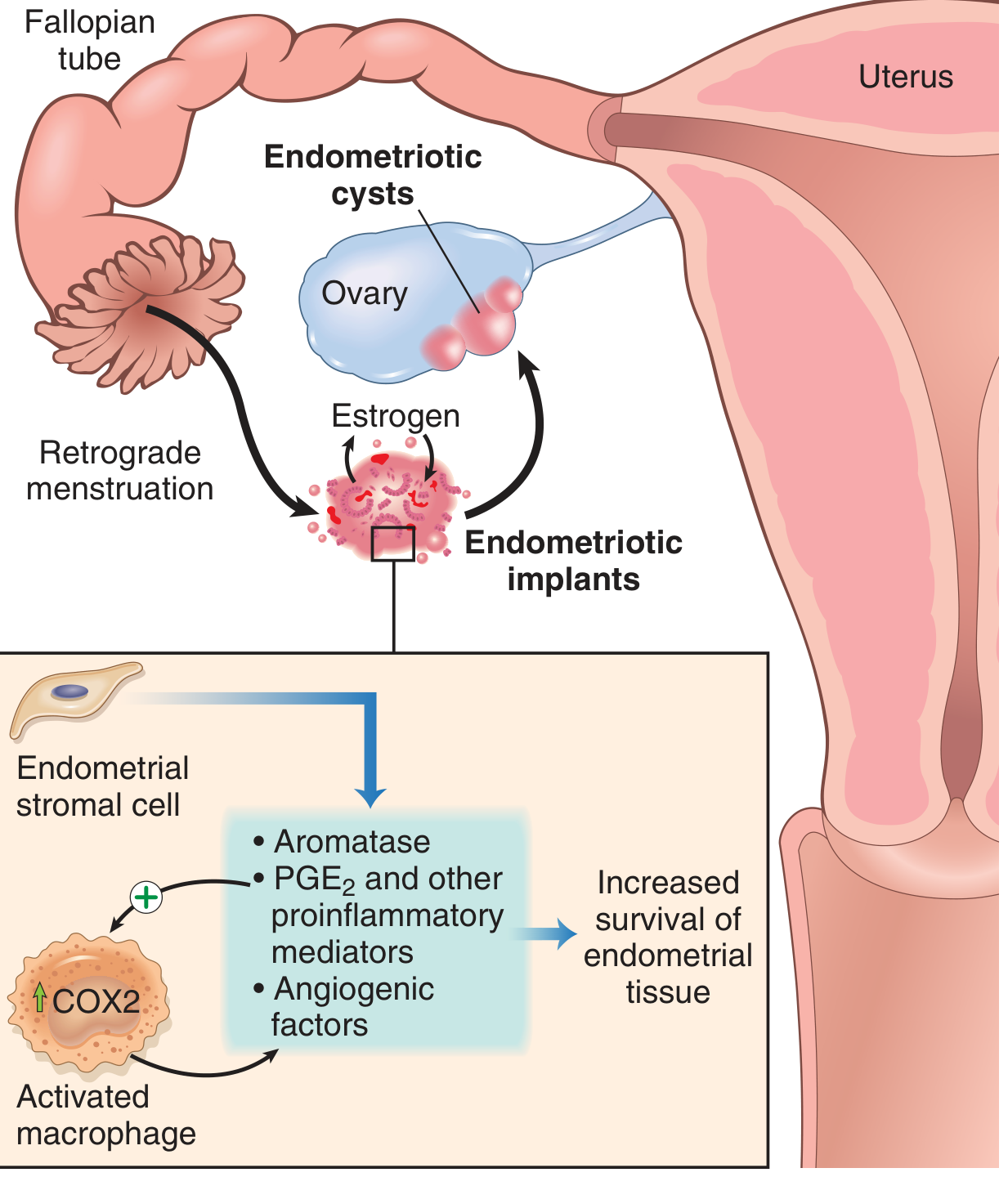
Key molecular drivers:
- Aromatase — elevated in endometriotic stromal cells → increased local estrogen production from androgens
- PGE2 (prostaglandin E2) — proinflammatory, promotes implant survival
- VEGF — drives angiogenesis to feed the implants
- MMPs — facilitate tissue invasion
- Recruited macrophages (COX2↑) amplify the inflammatory cycle
Morphology
Implants appear as red-brown nodules (microscopic to 1–2 cm) on serosal surfaces, caused by cyclic hemorrhage. In widespread disease:
- Organizing hemorrhage leads to fibrous adhesions between fallopian tubes, ovaries, and pelvic structures
- The pouch of Douglas may be obliterated
- Ovarian chocolate cysts (endometriomas) form — cystic masses 3–5 cm filled with old brown blood
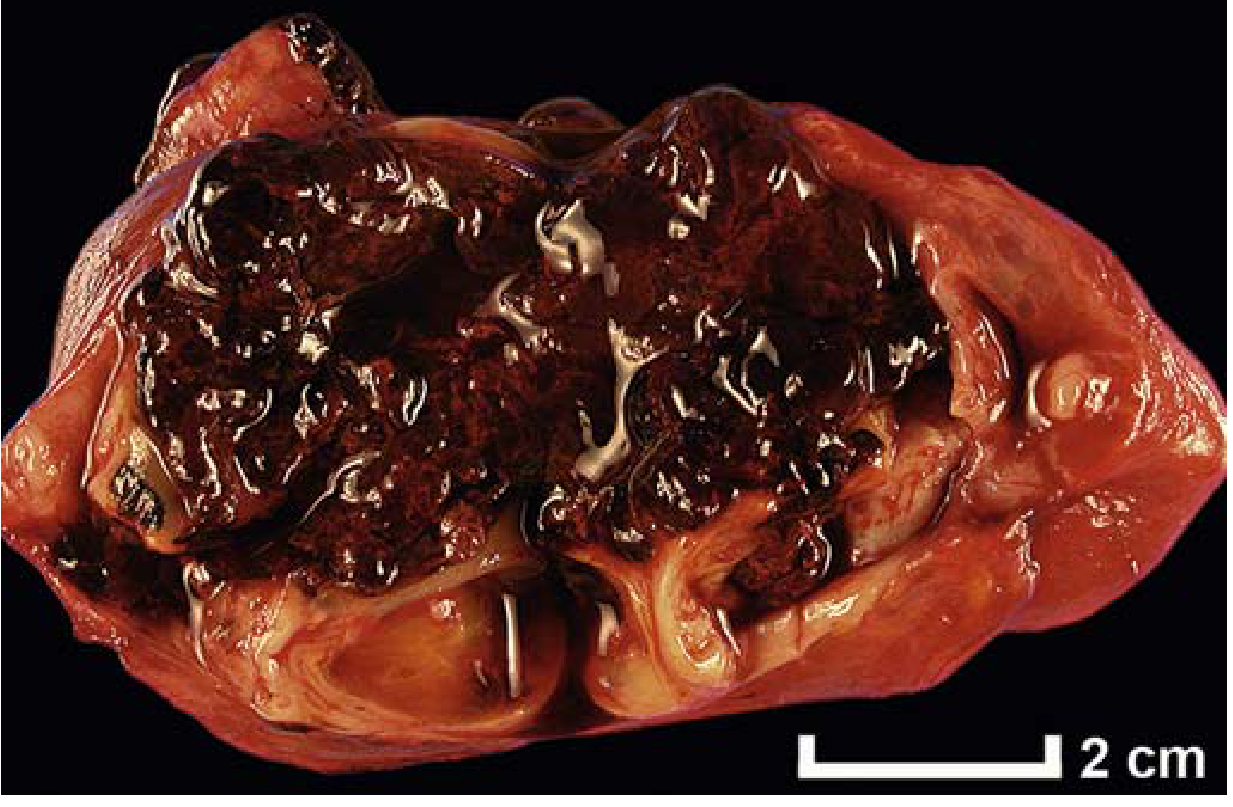
Histologic diagnosis requires identifying both endometrial glands AND stroma at the ectopic site.
Clinical Features
- Dysmenorrhea (most common symptom)
- Dyspareunia (painful intercourse)
- Chronic pelvic pain
- Infertility (~50% of infertile women)
- Rectal bleeding (bowel involvement — sigmoid most common, 12–37% of cases)
- Laparoscopic findings: "powder-burn" lesions, peritoneal implants, dense adhesions, chocolate cysts
Staging (r-ASRM)
Classified I–IV (minimal to severe) based on implant size, adhesions, and involvement of pelvic structures. The Endometriosis Fertility Index (EFI) is a validated tool for predicting post-surgical fertility outcomes.
Treatment
Treatment must be individualized, considering symptom profile, desire for fertility, and disease severity. Endometriosis is a chronic disease with high recurrence after both medical and surgical treatment. — Berek & Novak's Gynecology
1. Analgesics
- NSAIDs (ibuprofen, naproxen, mefenamic acid) — first-line for dysmenorrhea; inhibit prostaglandin synthase
- Started at onset of bleeding, continued up to 3 days
2. Hormonal (Medical) Therapy
Goal: suppress ovulation and reduce estrogen-driven implant growth
| Agent | Mechanism |
|---|---|
| Combined oral contraceptives | Suppress ovulation; continuous use preferred |
| Progestins (norethindrone 5 mg/day) | Decidualization and atrophy of implants |
| GnRH agonists (e.g., leuprolide) | Pituitary downregulation → hypoestrogenic state; often combined with "add-back" HRT to prevent bone loss |
| Danazol | Androgen/anti-gonadotropin; suppresses implants but androgenic side effects limit use |
Medical therapy provides temporary relief — implants recur after treatment cessation.
3. Surgical Treatment
- Laparoscopy is preferred over laparotomy (equivalent efficacy, faster recovery, less morbidity, fewer adhesions)
- Laparotomy only for advanced-stage disease where laparoscopy is impossible
- Goal: excise all visible lesions, lyse adhesions, restore normal anatomy
Peritoneal endometriosis: ablation (coagulation, laser) or excision — both equivalent; excision preferred to allow histologic confirmation
Ovarian endometriomas (≥3 cm): Cystectomy (excision of cyst wall) is preferred over drainage + ablation — lower recurrence of pain, lower endometrioma recurrence, higher spontaneous pregnancy rates (ESHRE guideline)
Deep infiltrating endometriosis: Complete excision in a single procedure at a specialized center with multidisciplinary expertise; asymptomatic patients should NOT undergo surgery unless complete ureteral obstruction with silent renal loss is present
4. Assisted Reproduction
For infertility-associated endometriosis when medical/surgical options are insufficient.
Extrapelvic Manifestations
- GI tract (12–37%): sigmoid colon most common; serosal implants invade muscularis propria → fibrosis, bowel wall contraction, rectal bleeding; MRI demonstrates low-signal fibrotic plaques obliterating the pouch of Douglas
- Diaphragm: upper abdominal extrapelvic endometriosis (requires thorough exploration during surgery)
- Pleura/Lung (rare): catamenial pneumothorax, hemoptysis
- Surgical scars, umbilicus
Prognosis
- Disease progresses in 30–60% of patients within a year of diagnosis
- Recurrence is high after both medical and surgical treatment
- Risk of malignant transformation is mainly confined to deep infiltrating endometriosis (particularly clear cell and endometrioid carcinoma arising in endometriomas)
- Referral to a specialized center is strongly recommended for severe or deep endometriosis
Sources: Robbins & Kumar Basic Pathology; Berek & Novak's Gynecology; Goldman-Cecil Medicine; Grainger & Allison's Diagnostic Radiology
This is a shared conversation. Sign in to Orris to start your own chat.