Here is a comprehensive review of both helminthic zoonoses:
Helminthic Zoonoses: Echinococcosis & Trichinellosis
PART I — ECHINOCOCCOSIS (Hydatid Disease)
Etiology
Echinococcosis is caused by larval-stage cestodes (tapeworms) of the genus Echinococcus. Four species cause human disease:
| Species | Disease Form |
|---|
| E. granulosus | Cystic echinococcosis (CE) — most common |
| E. multilocularis | Alveolar echinococcosis (AE) — most dangerous |
| E. vogeli | Polycystic echinococcosis |
| E. oligarthrus | Polycystic echinococcosis (rare) |
Adult tapeworms live in the small intestine of definitive hosts (carnivores — dogs, wolves, foxes). Intermediate hosts (herbivores — sheep, cattle, deer) ingest eggs from contaminated soil/vegetation and develop larval cysts in visceral organs. Humans are accidental dead-end intermediate hosts infected via fecal-oral route.
Epidemiology
- Over 1 million people are infected worldwide at any given time
- Highly endemic in sheep- and cattle-raising regions: Mediterranean, Middle East, Central Asia, sub-Saharan Africa, South America, parts of Australia
- E. multilocularis is endemic in central Europe, Russia, northern Japan, Alaska, and Canada
- In the U.S., most cases occur in foreign-born individuals; autochthonous cases are rare (Navajo Nation, Alaskan natives)
- Children are more commonly infected due to close contact with dogs
- Socioeconomic burden is enormous — productivity losses and medical costs estimated at $3 billion/year globally
Pathogenesis
- Egg ingestion — humans ingest eggs shed in feces of infected dogs/carnivores
- Oncosphere release — gastric acid dissolves the egg shell; the hexacanth embryo (oncosphere) is released and penetrates the intestinal mucosa
- Hematogenous/lymphatic spread — oncospheres travel via portal blood to the liver (70–80% of cases), then lungs (15%), and occasionally brain, bone, kidney, spleen, or other organs
- Cyst formation:
- E. granulosus → forms a unilocular hydatid cyst with three layers:
- Pericyst: outer fibrous layer formed by host reaction
- Laminated membrane: middle acellular layer (parasite-derived)
- Germinal layer (endocyst): inner scolecogenic layer; produces brood capsules, protoscoleces, and daughter cysts
- The cyst fills with clear fluid ("hydatid fluid") containing hydatid sand (free scoleces, hooklets, brood capsule fragments)
- E. multilocularis → grows as an infiltrative, alveolar mass with no defined outer layer; invades adjacent tissues like a malignancy; can metastasize to lungs, brain
Signs and Symptoms
Cystic Echinococcosis (E. granulosus)
- Often asymptomatic for years to decades (slow growth ~1 cm/year)
- Liver cysts: right upper quadrant (RUQ) pain, hepatomegaly, nausea, palpable mass
- Pulmonary cysts: cough, hemoptysis, dyspnea, chest pain
- Complications:
- Cyst rupture → anaphylaxis (potentially fatal), secondary seeding (peritoneal, pleural)
- Biliary communication → cholangitis, jaundice, biliary obstruction, cholangiohepatitis
- Secondary bacterial infection → hepatic abscess
- Compression of adjacent structures (IVC, bile ducts)
Alveolar Echinococcosis (E. multilocularis)
- Mimics hepatocellular carcinoma or cirrhosis clinically
- Presents with jaundice, hepatomegaly, RUQ pain, portal hypertension
- May metastasize to lungs and brain
- Mortality approaches 100% if untreated within 10–15 years
Diagnosis
Imaging (first-line):
- Ultrasound (90–95% sensitivity): reveals cystic structure; the WHO classification (CE1–CE5) guides management:
- CE1: unilocular simple cyst with hydatid sand
- CE2: multivesicular with daughter cysts
- CE3: detached floating membrane ("water lily sign")
- CE4: heterogeneous; CE5: calcified (inactive)
- CT: better defines cyst location, number, daughter cysts, calcification, biliary involvement
- MRI: superior for biliary tract communication and brain/spinal involvement
- Pathognomonic finding: daughter cysts within the mother cyst
Serology:
- ELISA (sensitivity 60–90%): screening test; higher sensitivity for liver cysts than lung/other sites
- Immunoblot (Western blot): confirmatory test; antigen 5 and antigen B (arc-5) are most specific
- Serology may be negative in up to 40% of cases (especially pulmonary or calcified/inactive cysts)
Cyst fluid/pathology (when available after resection or PAIR):
- Hydatid sand: protoscoleces, hooklets, brood capsule fragments — pathognomonic
- Histology of pericyst shows acellular laminated membrane
Laboratory:
- Peripheral eosinophilia (variable — may be absent)
- Liver function tests may be elevated with biliary involvement
Caution: Percutaneous aspiration of intact cysts is CONTRAINDICATED without concurrent albendazole coverage due to risk of anaphylaxis and secondary seeding.
Differential Diagnosis
| Condition | Distinguishing Features |
|---|
| Simple hepatic cyst | No daughter cysts, seronegative, no calcification pattern |
| Liver abscess (pyogenic/amoebic) | Fever, leukocytosis, tender; amoebic serology |
| Biliary cystadenoma/cystadenocarcinoma | Internal septations, mural nodules, CA 19-9 elevated |
| Polycystic liver disease | Multiple cysts, family history, associated renal cysts |
| Hepatocellular carcinoma | Solid/mixed lesion, AFP elevation, cirrhosis background |
| E. multilocularis vs. liver malignancy | Requires serology + imaging (AE has irregular infiltrative margins) |
| Renal cyst / multilocular renal cyst | Location, no hydatid sand |
Treatment
Medical therapy:
- Albendazole 400 mg twice daily (15 mg/kg/day) in 28-day cycles with 14-day breaks
- Duration: 1–6 months depending on cyst type and response
- Always given before and after any interventional procedure (minimum 4 weeks pre-procedure)
- Mebendazole is an alternative but has poor bioavailability
PAIR (Percutaneous Aspiration, Injection, Re-aspiration):
- Treatment of choice for uncomplicated CE1 and CE3a cysts
- Steps: Puncture under US/CT guidance → Aspirate cyst fluid → Inject scolicidal agent (hypertonic saline 20%, ethanol 95%) → Re-aspirate
- Done under albendazole cover
- Contraindicated: cysts communicating with bile ducts, superficial/inaccessible cysts, CE4/CE5
Surgery:
- Indicated for: large cysts (>10 cm), complicated cysts (biliary fistula, secondary infection, extrahepatic compression), failed PAIR, CE2/CE3b
- Techniques: radical resection (pericystectomy/hepatic resection) preferred; conservative procedures (deroofing, drainage) have higher recurrence
- Intraoperative precautions: isolate field with hypertonic saline-soaked packs; avoid spillage
Alveolar echinococcosis:
- Radical resection (R0) if possible — curative
- Long-term (often lifelong) albendazole if non-resectable
- Liver transplantation in selected cases (requires permanent albendazole post-transplant due to recurrence risk in immunosuppressed state)
Watch and wait: for CE4 and CE5 (inactive, calcified) — observe with serial imaging
Prevention
- Deworm dogs regularly with praziquantel (especially in endemic areas)
- Handwashing after contact with dogs, before eating
- Avoid ingesting raw vegetables or water potentially contaminated with dog feces
- Meat inspection and proper disposal of infected animal organs
- Boiling or cooking suspected food
- Public health education in endemic regions
- Foxes (E. multilocularis): baited praziquantel traps deployed in endemic areas in Europe
PART II — TRICHINELLOSIS (Trichinosis)
Etiology
Caused by nematodes of the genus Trichinella — at least 9 validated species and 3 genotypes:
| Species | Geographic Distribution | Notable Feature |
|---|
| T. spiralis | Worldwide (pigs) | Most common; encapsulated |
| T. nativa | Arctic/subarctic | Freeze-resistant (survives −20°C for months) |
| T. britovi | Europe, West Asia | Sylvatic cycle |
| T. pseudospiralis | Worldwide | Non-encapsulated; birds, mammals |
| T. papuae | Papua New Guinea | Non-encapsulated |
| T. zimbabwensis | Africa | Reptiles |
T. spiralis remains the primary human pathogen globally and is capable of infecting all mammals.
Epidemiology
- 65,818 cases reported globally between 1986 and 2009 (WHO); true incidence much higher
- In the U.S.: declined from 400–500 cases/year in the 1940s to 10–20 cases/year currently — largely due to laws prohibiting feeding raw garbage to pigs
- Sources in developed countries: now primarily wild game (bear, boar, walrus, cougar) rather than commercial pork
- Outbreaks remain common in Eastern Europe, China, Southeast Asia, and South America (often linked to undercooked pork or wild game)
- Arctic outbreaks associated with T. nativa from walrus or polar bear meat
- Transmission occurs when humans eat raw or undercooked meat containing encysted larvae
Pathogenesis
Three overlapping phases:
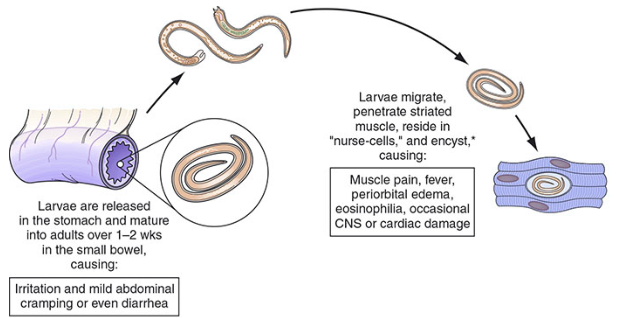
Phase 1 — Intestinal (Enteric) Phase (Days 1–7)
- Ingested encysted larvae are released by gastric acid and intestinal enzymes
- Larvae penetrate small intestinal epithelium and undergo 4 molts over 1–2 weeks → develop into adult worms
- Female adults (2.2 mm) mate and begin releasing live larvae within 5–7 days
- Adult worms cause local intestinal inflammation
Phase 2 — Migratory (Parenteral) Phase (Week 2–3)
- Newborn larvae (~100 μm) enter lymphatics/blood → disseminate hematogenously to all organs
- Larvae penetrate striated muscle fibers (preferentially those with high oxygen supply: diaphragm, masseter, tongue, intercostals, eye muscles, gastrocnemius)
- Extensive eosinophilic inflammation, vasculitis, tissue edema
- Larvae can invade heart, brain, lungs — causing myocarditis, encephalitis, pneumonitis
Phase 3 — Muscle Encystment Phase (Week 3+)
- Larvae modify host striated muscle cells into "nurse cells" (also called Nurse Cell-Parasite Complex)
- Nurse cell: a transformed myocyte that loses myofibrils, becomes granular, and develops an angiogenic network to nourish the larvae
- Larvae become encapsulated in a collagen-rich capsule; remain viable for years to decades
- Calcification occurs over time (years)
- T. pseudospiralis/papuae: do NOT form nurse cells (non-encapsulated)
Signs and Symptoms
Severity depends on larval burden (light infections may be entirely asymptomatic).
Phase 1 — Intestinal (Days 1–7):
- Nausea, vomiting, diarrhea, abdominal cramping
- Often mild and overlooked; may resemble gastroenteritis
Phase 2 — Invasion (Weeks 2–5):
- High fever (38–40°C) — cardinal symptom
- Periorbital/facial edema — classic finding (due to larvae invading extraocular muscles and periorbital tissue)
- Myalgia and muscle tenderness (especially masseter, tongue, eye muscles, limb muscles)
- Subungual splinter hemorrhages, conjunctival hemorrhages
- Maculopapular rash (occasionally)
- Eosinophilia — almost universal (>90% of symptomatic patients); may be dramatic (>50% of WBC)
- Elevated muscle enzymes: creatine phosphokinase (CPK), LDH, aldolase
- Respiratory symptoms: dyspnea, cough (larval migration through lungs)
Severe/Potentially Fatal Complications:
- Myocarditis: tachycardia, arrhythmias, heart failure (ECG: ST-T changes, AV block); leading cause of death
- Encephalitis/meningitis: headache, confusion, seizures, focal deficits; CSF pleocytosis (lymphocytic + eosinophilic)
- Pneumonitis: cough, hemoptysis
Phase 3 — Convalescent (Weeks 5–8+):
- Gradual resolution of fever and edema
- Persistent myalgia and fatigue may last months
- Complete recovery in most mild-moderate cases
Diagnosis
Clinical triad (highly suggestive):
- Periorbital edema
- Myalgia/muscle tenderness
- Eosinophilia
Plus: epidemiological exposure (undercooked meat, especially pork or wild game), often in an outbreak setting.
Laboratory:
- Eosinophilia >1,500/μL (often very high); absent in severe disease with immunosuppression
- Elevated CPK, LDH, aldolase (reflect muscle invasion)
- Normal or mildly elevated liver enzymes
- Leukocytosis common during acute phase
Serology (confirmatory):
- ELISA (antigen from T. spiralis excretory-secretory products): becomes positive at weeks 3–4 after infection
- Western blot: confirmation
- Limitation: may be negative early in infection; false-positives can occur
Muscle biopsy (definitive but invasive):
- At least 1 gram of deltoid or gastrocnemius muscle
- Squash preparation (pressing muscle between glass slides) or histology
- Reveals encysted larvae within nurse cells
- Best yield at weeks 4–6
- Trichinoscopy is used for meat inspection
Epidemiological investigation:
- Suspected meat source should be sent for trichinoscopy or digestion testing
Cardiac evaluation:
- ECG, troponin, echocardiography for cardiac involvement
Imaging:
- CT brain for neurological symptoms; may show small enhancing lesions
- Chest X-ray: pulmonary infiltrates (rare)
Differential Diagnosis
| Condition | Distinguishing Features |
|---|
| Polymyositis/dermatomyositis | No fever, no eosinophilia, autoantibodies (anti-Jo-1), skin changes |
| Polyarteritis nodosa | Eosinophilia absent/mild, ANCA+, renal/GI involvement predominates |
| Viral myositis (influenza, Coxsackie) | No periorbital edema, less dramatic eosinophilia, serology |
| Toxocariasis (visceral larva migrans) | Eosinophilia present, but primarily hepatosplenic; different muscle pattern |
| Systemic lupus erythematosus | ANA+, multi-organ involvement, no epidemiological exposure |
| Infectious mononucleosis | Atypical lymphocytosis, +monospot, periorbital edema (early), no eosinophilia |
| Angioedema (allergic) | Rapid onset, no fever, no eosinophilia |
| Trichinellosis vs. myocarditis (other causes) | Eosinophilia + epidemiological link to undercooked meat |
Treatment
Anthelmintic therapy:
- Albendazole 400 mg twice daily × 10–14 days — drug of choice (better CNS penetration than mebendazole)
- Mebendazole 200–400 mg three times daily × 3 days, then 400 mg three times daily × 10 days — alternative
- Both are effective against intestinal adult worms and migrating larvae but have limited efficacy against encysted muscle larvae
- Treatment should be initiated as early as possible (during the intestinal phase)
Corticosteroids (for severe disease):
- Prednisone 40–60 mg/day (or 1 mg/kg/day) × 5–14 days
- Indicated for: myocarditis, encephalitis, severe allergic manifestations
- Reduces larval-associated inflammation and allergic reactions
- Use simultaneously with anthelmintics (not alone — steroids alone may prolong intestinal phase)
Mild disease: supportive care only (NSAIDs for myalgia, hydration, rest)
Cardiac involvement: antiarrhythmic agents as needed; monitoring in ICU for severe myocarditis
Prevention
Cooking:
- Cook pork and wild game thoroughly until internal temperature reaches ≥71°C (160°F) — until meat is no longer pink in the center
- Microwave cooking is unreliable (uneven heating)
- Well-done is the standard recommendation
Freezing:
- Freezing pork at −15°C for ≥3 weeks (or −23°C for ≥10 days) kills T. spiralis
- Ineffective against T. nativa (Arctic species) — freezing does NOT reliably kill it
- Smoking, drying, and curing are NOT reliable methods of killing larvae
Animal husbandry:
- Ban feeding raw garbage/scraps to pigs (the most impactful public health measure in the U.S.)
- Control rodents on farms (rodents are reservoir for T. spiralis)
- Meat inspection: digestion method (pooled sample from diaphragm pillar) or trichinoscopy — mandatory in many countries
Outbreak response:
- Identify and trace the contaminated meat source
- Notify public health authorities
- Treat exposed individuals during the intestinal phase if confirmed exposure
Summary Comparison Table
| Feature | Echinococcosis | Trichinellosis |
|---|
| Causative agent | Echinococcus spp. (tapeworm larva) | Trichinella spp. (roundworm) |
| Transmission | Fecal-oral (dog feces → eggs) | Ingestion of undercooked infected meat |
| Definitive host | Dogs, wolves, foxes | None (direct life cycle in single host) |
| Primary organ affected | Liver (70–80%), lungs (15%) | Skeletal muscle (all striated muscle) |
| Hallmark finding | Hydatid cyst with daughter cysts | Periorbital edema + eosinophilia + myalgia |
| Key lab finding | ELISA (arc-5 antigen) | Eosinophilia, elevated CPK |
| Imaging | US/CT (cyst with daughter cysts) | Not diagnostic; muscle biopsy definitive |
| Treatment | Albendazole + PAIR/surgery | Albendazole ± prednisone |
| Prevention | Deworm dogs, handwashing, meat inspection | Cook meat thoroughly; ban raw scraps to pigs |
Sources: Goldman-Cecil Medicine; Tietz Textbook of Laboratory Medicine (7th ed.); Mulholland & Greenfield's Surgery (7th ed.); Harrison's Principles of Internal Medicine (22nd ed.); Adams & Victor's Principles of Neurology (12th ed.)