Helminthic zoonoses (Echinococcosis. Trichinellosis). Etiology, epidemiology, pathogenesis, sign and symptoms, diagnosis, differential diagnosis, treatment, prevention.
Read multiple medical textbook sections about Echinococcosis and Trichinellosis and return a comprehensive synthesis. Read the following files and line ranges: 1. Goldman-Cecil Medicine - Echinococcosis section: File: /textbooks/9780323930345/9780323930345_block49.md Read from line 1 to line 300 (covers echinococcosis section) 2. Harrison's Principles - Trichinellosis section: File: /textbooks/9781265977061/9781265977061_block26.md Read lines 1813 to 1900 3. Tietz Textbook - Echinococcus Species section: File: /textbooks/9780323775724/9780323775724_block38.md Read lines 2200 to 2320 4. Tietz Textbook - Trichinella Species section: File: /textbooks/9780323775724/9780323775724_block38.md Read lines 2050 to 2110 5. Goldman-Cecil Medicine - Trichinellosis section: File: /textbooks/9780323930345/9780323930345_block49.md Read lines 1255 to 1340 6. Sherris Medical Microbiology - Echinococcus section: File: /textbooks/9781260464283/9781260464283_block7.md Read lines 1650 to 1760 Please read ALL of these sections using read_file tool with appropriate offset/limit. Then provide a detailed structured synthesis covering for BOTH diseases (Echinococcosis and Trichinellosis): - Etiology (causative organisms, taxonomy) - Epidemiology (geographic distribution, prevalence, transmission, life cycle) - Pathogenesis (mechanism of disease, host response) - Signs and Symptoms (clinical manifestations, stages) - Diagnosis (lab tests, imaging, serology) - Differential Diagnosis - Treatment (drugs, doses, surgery) - Prevention Return the FULL synthesized text with all details from the sources. Include any image URLs you encounter for relevant diagrams/illustrations. Cite book names when providing information.
Read the following medical textbook sections and return the full raw text content from each. Do NOT summarize — return the actual paragraph text as written in the files. 1. Goldman-Cecil Medicine - Echinococcosis full chapter: File: /textbooks/9780323930345/9780323930345_block49.md Read offset=0, limit=300 first, then offset=300, limit=300 if needed 2. Harrison's Principles - Trichinellosis section: File: /textbooks/9781265977061/9781265977061_block26.md Read offset=1813, limit=100 (adjust to 0-based if needed: try offset=1810, limit=90) Actually use read_file with offset=1810 and limit=90 3. Goldman-Cecil Medicine - Trichinellosis section: File: /textbooks/9780323930345/9780323930345_block49.md Read offset=1255, limit=100 4. Tietz Textbook - Trichinella section: File: /textbooks/9780323775724/9780323775724_block38.md Read offset=2050, limit=60 5. Tietz Textbook - Echinococcus section: File: /textbooks/9780323775724/9780323775724_block38.md Read offset=2200, limit=120 6. Sherris Medical Microbiology - Echinococcus section: File: /textbooks/9781260464283/9781260464283_block7.md Read offset=1650, limit=120 Return ALL the raw text you find, organized by source. Include all headings, bullet points, dosages, and clinical details exactly as written. Do not omit anything.
Echinococcus granulosus life cycle hydatid cyst
Trichinella spiralis life cycle muscle larvae
Helminthic Zoonoses: Echinococcosis & Trichinellosis
ECHINOCOCCOSIS (Hydatid Disease)
Etiology
| Species | Disease Form |
|---|---|
| E. granulosus complex (including E. canadensis; 10 genotypes) | Cystic echinococcosis (CE) — most common |
| E. multilocularis | Alveolar echinococcosis (AE) — more aggressive |
| E. vogeli / E. oligarthrus | Polycystic echinococcosis (South/Central America) |
Epidemiology
- Global distribution: highest prevalence in Eurasia, Africa, Australia, and South America, particularly in rural sheep-raising regions
- Community-based surveys show infection rates up to 6.6% in endemic areas
- In the US, ~200 cases/year, mostly imported; indigenous cases among Basque sheep farmers in western states and Native Americans in the southwest
- WHO estimates >1 million individuals infected worldwide; classified among 20 neglected tropical diseases (NTDs)
- Endemic in arctic and alpine zones of the Northern Hemisphere
- Highly endemic in western China, Tibet, central Asia, and alpine Europe
- Emerging in western Canada
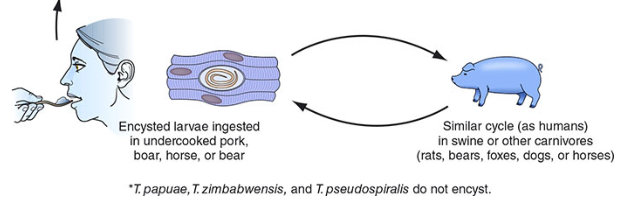
- Definitive hosts: Dogs, wolves, foxes (harbor adult tapeworms in small bowel)
- Intermediate hosts: Sheep, cattle, camels, moose, caribou, rodents (harbor cysts in viscera)
- Human infection: Accidental — by ingesting Echinococcus eggs from dog/fox feces via contaminated hands, food, or soil
- After ingestion, eggs hatch → oncospheres penetrate intestinal wall → travel via portal blood → lodge in liver (first filter), then lungs, brain, bone, heart, kidney
- In the liver/lungs, larvae form cysts; in canines that eat infected viscera, scolices mature into adult tapeworms (3–5 mm, only 3 proglottids in E. granulosus)
- Pastoral: Most common; dogs fed raw sheep/cattle viscera; shepherds infected by handling dogs (eggs on fur → hands → ingestion)
- Sylvatic: Alaska and western Canada; wolves + moose/caribou
Pathogenesis
- Cyst has three layers:
- Pericyst (adventitia): Host-derived fibrous outer layer
- Laminated layer: Acellular, outermost parasite layer
- Germinal (inner) layer: Produces brood capsules and protoscolices
- Cyst fills with clear fluid, brood capsules, protoscolices ("hydatid sand") and may develop daughter cysts
- Cyst grows slowly: ~1 cm/5–6 months; can exceed 10 cm over years
- Damage is primarily mechanical from cyst enlargement; rupture causes hypersensitivity reactions (urticaria, anaphylaxis) and dissemination
- Grows as a budding, infiltrative tumor-like mass rather than a contained cyst — hence the name "alveolar"
- Destroys liver tissue progressively; may disseminate hematogenously to lung and brain
- Fatal if untreated
Signs and Symptoms
- Liver (70% of cases): Upper abdominal pain/discomfort, hepatomegaly, palpable mass, jaundice, biliary colic, portal hypertension, ascites, inferior vena cava compression
- Lung (2nd most common): Cough (salty taste, vomiting of hydatid membranes if rupture into bronchi), chest pain, hemoptysis, pleural effusion, pneumothorax, eosinophilic pneumonitis
- Brain: Seizures, paralysis
- Spine: Neurologic symptoms
- Bone: Pathologic fractures
- Heart: Cardiac mass, pericardial effusion, arrhythmias, ventricular rupture
- Rupture: Fever, pruritus, urticaria, anaphylactic shock, dissemination
- Biliary involvement: Mimics cholecystitis; obstructive jaundice
- Epigastric pain, hepatomegaly, obstructive jaundice
- Resembles liver cancer — progressive, tumor-like mass expanding over decades
- May metastasize to lung and brain
- Weight loss, malaise, eventual liver failure
Diagnosis
- Ultrasound — procedure of choice; WHO classification defines 6 cyst stages in 3 groups:
- Active (viable): CE1 (unilocular), CE2 (multivesicular with daughter cysts)
- Transitional: CE3a ("water lily sign" — floating membranes), CE3b (solid with daughter cysts)
- Inactive (nonviable): CE4, CE5 (solidification + calcification)
- CT — detects smaller cysts outside liver, precise localization; >50% of hepatic cysts show calcific rim on X-ray
- MRI — best for postsurgical residual lesions, cardiac, and extrahepatic sites; T2-weighted series preferred when ultrasound not feasible
- Indirect hemagglutination, latex agglutination, EIA (screen) → immunoblot (confirm)
- Sensitivity: ~90% for hepatic, ~60% for pulmonary cysts; lower for brain/splenic cysts
- Serology typically negative in inactive cysts (CE4/CE5); titers rise after treatment (cyst disruption releases antigen)
- Cross-reactivity with T. solium (cysticercosis) — immunoblot needed for confirmation
- Protoscolices (100–150 μm, with hooklets) and free hooklets ("hydatid sand") in aspirated/excised cyst material
- Histopathology: Laminated membrane + brood capsules = CE; infiltrative back-to-back brood capsules without thick laminated wall = AE
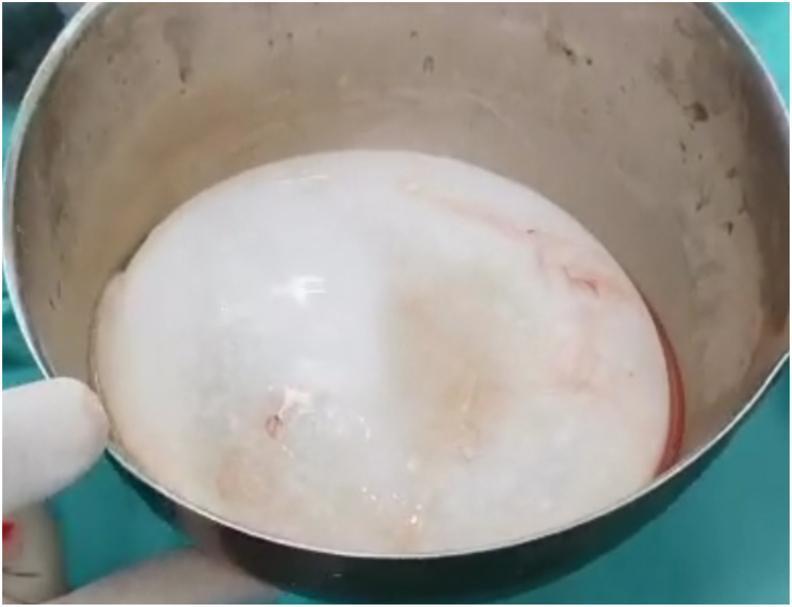
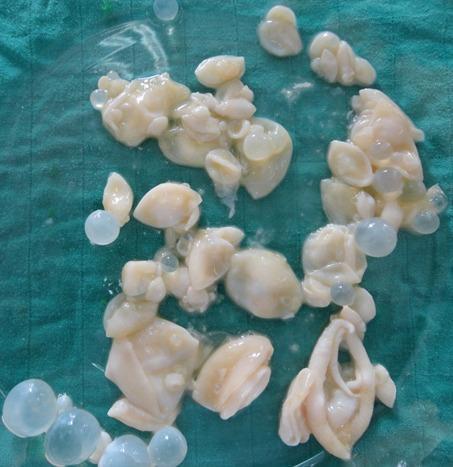
Differential Diagnosis
- Liver: Hepatocellular carcinoma, liver abscess (pyogenic or amoebic), nonparasitic simple cyst, polycystic liver disease, cholangiocarcinoma
- Lung: Primary lung tumor, lung abscess, tuberculoma, aspergilloma
- Brain: Primary or metastatic brain tumor, neurocysticercosis, brain abscess
- Bone: Primary bone tumor, metastatic disease
- AE specifically: Hepatocellular carcinoma (very closely mimics on imaging)
Treatment
- Inactive CE4 and CE5 cysts without complications can be monitored without intervention
- Drug of choice; oral 10–15 mg/kg/day continuously (without interruption) for ≥30 days
- Effective for small/medium CE1 and CE3a cysts; limited efficacy on cysts >10 cm or CE2/CE3b
- AE: Albendazole continued indefinitely (suppresses growth but rarely curative)
- After apparently curative AE surgery: albendazole for ≥2 years
- Adverse effects: hepatotoxicity, leukopenia, thrombocytopenia, alopecia; avoid in pregnancy
- For uncomplicated CE1 and CE3a cysts
- Scolicidal agent: hypertonic saline (preferred); anaphylaxis risk 0.03%, reversible allergic reactions 1.7%
- Must be combined with albendazole (≥30 days after puncture)
- CE2 and CE3b tend to relapse after PAIR
- Indications: complicated cysts (rupture, biliary fistula, compression of vital structures, superinfection, hemorrhage), CE2/CE3b, cysts at high risk of rupture, AE
- Perioperative albendazole (10–15 mg/kg/day): start 1 week before, continue 4 weeks after surgery
- Some add praziquantel (50 mg/kg/day) perioperatively
- AE may require liver transplantation if resection is incomplete
- Open, conservative, or laparoscopic techniques
Prevention
- Deworm dogs regularly and dispose of infected carcasses/offal by burning or burial
- Prohibit feeding of raw viscera to dogs
- Handwashing after contact with dogs (especially in endemic areas)
- Control dog populations near sheep flocks
- Education of shepherds and hunters in endemic areas
- Vaccination of intermediate hosts (sheep) with EG95 vaccine has shown promise in some programs
TRICHINELLOSIS (Trichinosis)
Etiology
| Species | Distribution | Primary Hosts |
|---|---|---|
| T. spiralis | Worldwide | Domestic pigs, rats, carnivores |
| T. pseudospiralis | Worldwide | Mammals and birds (non-encapsulating) |
| T. nativa | Arctic/subarctic | Bears, foxes, walruses |
| T. britovi | Europe, W. Africa, W. Asia | Wild carnivores (not domestic swine) |
| T. nelsoni | Equatorial E. Africa | Felids, hyenas, bush pigs |
| T. murrelli | N. America, Japan | Wild animals |
| T. papuae | Papua New Guinea, SE Asia | Pigs, crocodiles, turtles (non-encapsulating) |
| T. zimbabwensis | Tanzania | Crocodiles (non-encapsulating) |
| T. patagoniensis | S. America | Cougars |
Epidemiology
- T. spiralis is enzootic worldwide in omnivorous/carnivorous animals (bears, boars, rats, pigs)
- US trend: Declined from 400–500 cases/year in the 1940s to ~10–20 cases/year currently, due to education, pig-raising regulations, and pork freezing/processing requirements
- Global burden: A review of 1986–2009 outbreaks documented 65,818 cases from 41 countries, with 42 deaths; 86% from Europe (nearly 50% from Romania pre-2000)
- Transmission routes:
- Domestic cycle: Undercooked pork (most common worldwide) — pigs infected by eating raw meat scraps/rats
- Sylvatic cycle: Wild game — bear, wild boar, walrus, horse (now the majority of US cases)
- Dogs (Asia/Africa), horses (Italy/France)
- Herbivores (cattle, horses) not natural hosts but implicated when contaminated with pork
- Curing and smoking do not reliably kill larvae
- T. nativa (Arctic): present in walrus and bear meat; resistant to freezing
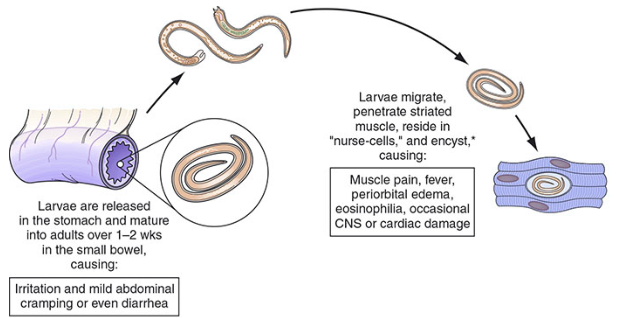
- Humans ingest encysted larvae in undercooked meat
- Gastric acids/proteases release larvae → invade small bowel mucosa → mature to adults in 2 days
- Adults embed in columnar epithelium; females (3 mm) produce newborn larvae within 5 days of mating; adults expelled by immune response within 3–5 additional weeks
- Newborn larvae (with a swordlike stylet) penetrate lamina propria → lymphatics/blood → migrate throughout body
- Larvae survive only in striated skeletal and cardiac muscle cells → transform muscle cells into "nurse cells" (unique to Trichinella) where they encyst and can survive for decades
- Calcification occurs eventually
- Humans are dead-end hosts — larvae cannot re-develop into adults in the same host
Pathogenesis
- Severity correlates with worm burden: <10 larvae/g muscle → asymptomatic; >50 larvae/g → potentially life-threatening
- The number of encysted larvae ingested is the key determinant of disease severity
- Phase 1 (Intestinal): Larval release + adult worm invasion of intestinal mucosa → local inflammation → diarrhea, abdominal pain
- Phase 2 (Migration/Systemic): Newborn larvae circulate and invade striated muscle → marked local and systemic hypersensitivity: fever, eosinophilia, periorbital edema
- Phase 3 (Muscle encystment): Nurse cell formation → myositis, myalgias, weakness
- Cardiac involvement: larvae invade myocardium → myocarditis, arrhythmias, heart failure
- CNS: encephalitis (rare)
- Acquired immunity expels adult intestinal worms but has little effect on muscle-dwelling larvae
Signs and Symptoms
- Diarrhea, abdominal pain, nausea, vomiting, constipation
- Self-limited; resolves within ~10 days
- Fever
- Periorbital and facial edema (classic sign)
- Eosinophilia (>90% of symptomatic patients; can exceed 50% at 2–4 weeks)
- Subconjunctival, retinal, and subungual ("splinter") hemorrhages
- Maculopapular rash
- Headache, cough, dyspnea, dysphagia
- Myocarditis (tachyarrhythmias, heart failure) — major cause of death
- Encephalitis, pneumonitis (uncommon but potentially fatal)
- Myalgias, muscle tenderness, weakness — inflammatory myositis
- Most commonly involved muscles: extraocular muscles, biceps, jaw, neck, lower back, diaphragm
- Elevated CPK and LDH
- Symptoms peak ~3 weeks post-infection; gradual recovery over months
- T. pseudospiralis infection (non-encapsulating): prolonged polymyositis-like illness
Diagnosis
- Eosinophilia: >90% of symptomatic patients; peaks >50% at 2–4 weeks
- Elevated muscle enzymes: CPK, LDH (most symptomatic patients)
- Serology (anti-Trichinella antibodies): EIA (most common), latex agglutination, indirect immunofluorescence
- IgG antibodies most sensitive
- Usually detectable 2–5 weeks post-infection (may be negative in first 2 weeks)
- Repeat testing may be needed
- Cross-reactivity with other nematodes; immunoblot for confirmation
- Muscle biopsy (definitive): ≥1 g of deltoid or gastrocnemius (near tendon insertions); coiled larvae seen in squash preparations (fresh tissue compressed between glass slides); pepsin digestion improves visualization; larvae may be missed on routine histology sections alone
- PCR: Detects Trichinella DNA; allows species identification from biopsy
Differential Diagnosis
- Periorbital edema: Angioedema, allergic reaction, nephrotic syndrome, orbital cellulitis
- Myositis/myalgias + eosinophilia: Polymyositis, dermatomyositis, eosinophilic myositis, fasciitis
- Eosinophilia + fever: Other helminthic infections (toxocariasis, fascioliasis, strongyloidiasis), drug hypersensitivity, hypereosinophilic syndrome
- Myocarditis: Viral myocarditis, other causes
- CNS involvement: Viral encephalitis, other parasitic CNS infections (neurocysticercosis)
- Intestinal phase: Acute gastroenteritis (bacterial, viral), food poisoning
Treatment
| Severity | Treatment |
|---|---|
| Mild | Supportive care: bed rest, antipyretics, analgesics |
| Moderate | Albendazole 400 mg twice daily × 10–14 days, OR Mebendazole 200–400 mg tid × 3 days, then 500 mg tid × 10 days |
| Severe (myocarditis, encephalitis) | Add glucocorticoids (prednisone 1 mg/kg/day × 5 days, or 40–60 mg/day × 8–14 days) WITH albendazole |
- Albendazole/mebendazole are active against intestinal adult worms and prevent further larval release; efficacy against fully encysted muscle larvae is limited
- Giving benzimidazoles during systemic phase may worsen symptoms (dying larvae → exacerbated inflammatory response); combine with corticosteroids in severe cases
- Corticosteroids alone used for severe myositis and myocarditis
Prevention
- Thorough cooking of pork and wild game until meat is no longer pink (internal temperature ≥63°C/145°F)
- Freezing at −15°C for 3 weeks or −20°C for at least 3 days kills T. spiralis in pork
- Exception: T. nativa (Arctic species in walrus/bear) is resistant to freezing — cooking is the only reliable method
- Curing, salting, smoking, and drying are NOT reliable
- Regulations: Ban on feeding unprocessed meat scraps/garbage to pigs (enforced in US, Canada, EU) has dramatically reduced domestic pork transmission
- Meat inspection programs (including the ECDC-endorsed approach)
- Consumer education regarding wild game preparation
Summary Comparison Table
| Feature | Echinococcosis (CE) | Echinococcosis (AE) | Trichinellosis |
|---|---|---|---|
| Agent | E. granulosus | E. multilocularis | Trichinella spp. (mainly T. spiralis) |
| Type | Cestode (larval) | Cestode (larval) | Nematode |
| Transmission | Dog feces → eggs ingested | Fox feces → eggs ingested | Undercooked infected meat |
| Main location | Liver (70%), lung | Liver (invasive) | Small bowel (adults) → striated muscle (larvae) |
| Latency | 5–20 years | Years–decades | Days–weeks |
| Key sign | Cystic mass | Tumor-like liver mass | Periorbital edema, myositis, eosinophilia |
| Key diagnosis | Ultrasound + serology | CT/MRI + serology | Eosinophilia + muscle biopsy + serology |
| Drug of choice | Albendazole ± PAIR/surgery | Albendazole + surgery | Albendazole (+ steroids if severe) |
| Prevention | Deworm dogs, hygiene | Deworm dogs/cats, avoid fox-contaminated food | Cook meat thoroughly |