synthesis of ketamine hydrochloride and how it acts on the body, how it works in the body, its side effects and uses.
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive information from authoritative textbooks. Let me compile a complete response.
Ketamine Hydrochloride
1. Chemical Synthesis
Ketamine (2-(2-chlorophenyl)-2-(methylamino)cyclohexan-1-one) is synthesized through a well-established multi-step route:
Starting material: 2-Chlorobenzonitrile (or 2-chlorophenyl Grignard reagent)
Key synthetic steps:
- Cyclopentanone ring formation / Grignard reaction: A 2-chlorophenyl Grignard reagent (from 2-chlorobromobenzene and magnesium) is reacted with cyclopentanone or, alternatively, cyclohexanone is used with o-chlorobenzonitrile to build the arylcyclohexanone skeleton.
- Amino ketone intermediate: The aryl ketone undergoes α-bromination, then nucleophilic substitution with methylamine to introduce the N-methyl amino group.
- Rearrangement / oxidation: The cyclopentylamine intermediate is converted through a Baeyer-Villiger-type oxidation or ring-expansion step to give the 2-aminocyclohexanone scaffold.
- Salt formation: The free base is dissolved in anhydrous ethanol or isopropanol and treated with hydrochloric acid to precipitate ketamine hydrochloride as a crystalline white salt (pKa ~7.5, molecular weight 274.19 g/mol).
The commercial product is a racemic mixture of the (S)- and (R)-enantiomers. The (S)-(+) enantiomer (esketamine) is ~4× more potent and available separately as the nasal spray formulation (Spravato).
2. Pharmacokinetics — How It Works in the Body
Physicochemical properties: Ketamine has a low molecular weight, high lipid solubility, and a pKa near physiologic pH (~7.5), which allows it to cross the blood–brain barrier rapidly.
| Property | Value |
|---|---|
| Onset (IV) | 30–60 seconds; maximal effect ~1 minute |
| Duration (IV, 2 mg/kg) | 10–20 minutes of anesthesia; full orientation in 15–30 min |
| Plasma level for anesthesia | 0.6–2 mcg/mL (adults); 0.8–4 mcg/mL (children) |
| Plasma level for analgesia | ≥0.1 mcg/mL |
| Distribution | High lipophilicity → rapid brain uptake, then redistribution to peripheral tissues accounts for its short duration |
| Metabolism | N-demethylation by liver CYP450 (primarily CYP3A) → norketamine (active metabolite, ~1/3 the potency) → dehydronorketamine (inactive) |
| Elimination | Renal excretion of metabolites |
Peak ketamine levels occur ~20 minutes after IM injection; intranasal onset approaches IV. The (S)+ enantiomer undergoes ~10% faster hepatic biotransformation, enabling quicker recovery than the racemic mixture.
— Miller's Anesthesia, 10e; Barash Clinical Anesthesia, 9e; Kaplan & Sadock's Synopsis of Psychiatry
3. Mechanism of Action
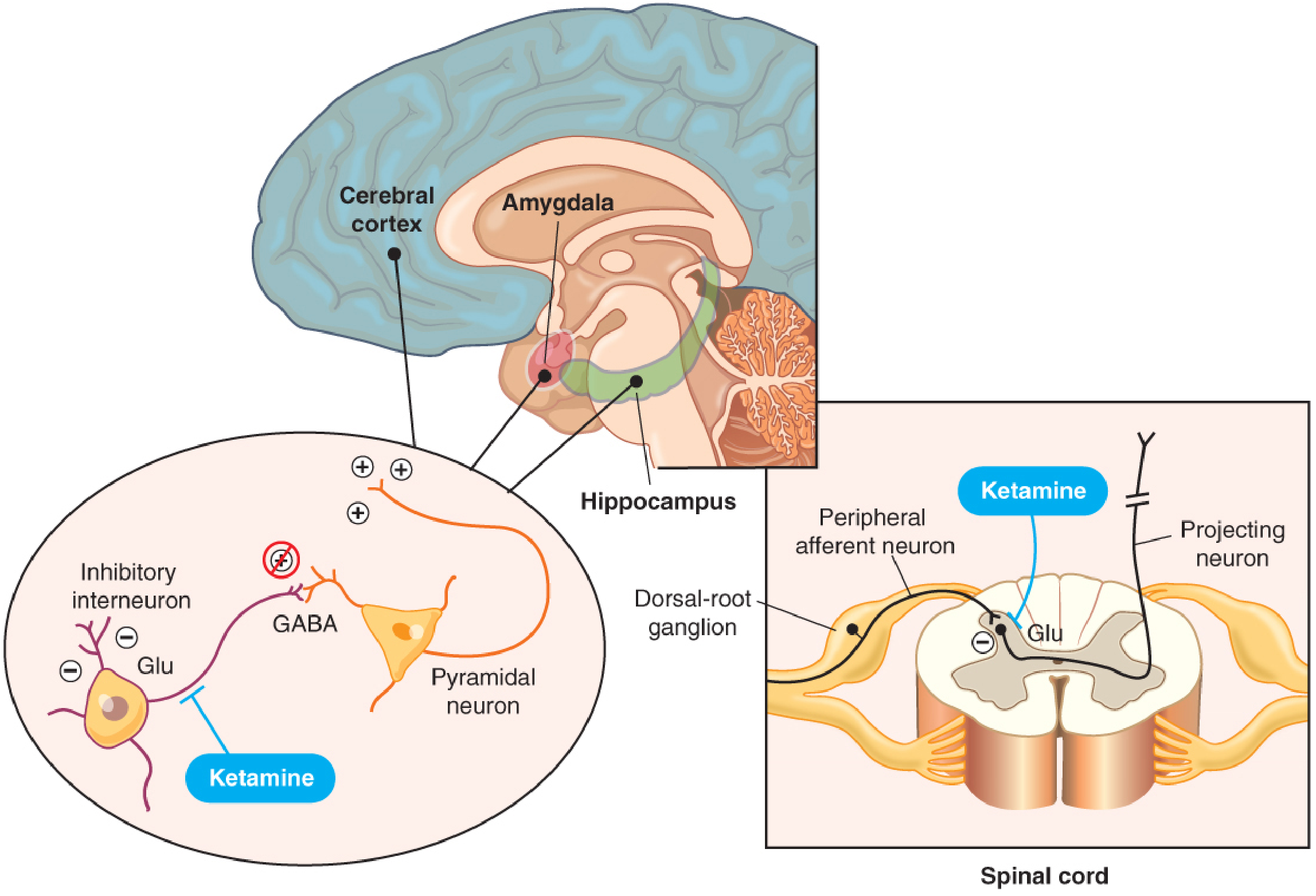
Ketamine acts at multiple receptor systems:
Primary: NMDA Receptor Antagonism
- Ketamine is a non-competitive, open-channel blocker of the N-methyl-D-aspartate (NMDA) glutamate receptor — it only enters and blocks the channel when it is open (use-dependent blockade).
- It preferentially binds NMDA receptors on inhibitory GABAergic interneurons in the cortex, amygdala, and hippocampus, removing their inhibitory tone on pyramidal neurons → net disinhibition and dissociated/excitatory cortical activity.
- This inhibition of NMDA-mediated glutamatergic input to the GABAergic system produces dissociative anesthesia: patients appear cataleptic with eyes open, maintain some protective reflexes, have profound analgesia, but have no recall of surgery.
- EEG shows depression of thalamocortical pathways with simultaneous activation of the limbic system — the basis of its dissociative character.
- At the spinal cord level, ketamine blocks NMDA receptor–mediated nociceptive signals in the dorsal horn → supraspinal and spinal analgesia, prevention of central sensitization and wind-up.
Secondary Receptors
- Opioid receptors (μ, δ, κ) — contributes to analgesia
- Monoaminergic receptors — dopaminergic, serotonergic, and adrenergic activity
- At high concentrations: sigma opioid receptors, muscarinic receptor blockade (→ bronchodilation, salivation), facilitation of GABA-ergic transmission
- Inhibition of acetylcholine release at the spinal cord level
- Sympathomimetic effect: ketamine stimulates the CNS to cause sympathoneuronal release of norepinephrine and inhibits neuronal reuptake of catecholamines (cocaine-like effect) → ↑HR, ↑BP, ↑cardiac output
— Miller's Anesthesia, 10e, p. 2552–2553
4. Clinical Uses
Anesthesia (Dissociative Anesthesia)
- Induction dose: 1–2 mg/kg IV (onset 1 min, duration 10–20 min); 4–6 mg/kg IM
- Causes a cataleptic state: unconscious, eyes open, profound analgesia, spontaneous respiration maintained, airway reflexes partially preserved
- Excellent for hemodynamically unstable patients: hemorrhagic shock, sepsis, trauma, cardiac tamponade — its sympathomimetic effect supports blood pressure unlike other induction agents
- Drug of choice in reactive airway disease (asthma) — produces significant bronchodilation
- Used in pediatric procedural sedation: fracture reduction, wound care, burn dressing changes
- Safe in uncooperative or pre-hospital patients (IM route)
Analgesia
- Analgesic blood concentrations are sub-anesthetic (≥0.1 mcg/mL)
- Reduces postoperative pain scores and opioid consumption; most effective when given as a bolus before incision + infusion postoperatively
- Valuable in opioid-tolerant patients and those with chronic opiate therapy
- Intranasal ketamine reduces breakthrough pain in chronic and neuropathic pain patients
Chronic Pain
- Most extensively studied in complex regional pain syndrome (CRPS) — decreases pain scores and opioid consumption (though duration of benefit is limited)
- Used in severe cancer pain (reduces but does not eliminate opioid requirements)
Psychiatric — Antidepressant
- Rapid-onset antidepressant effect: reduced depression symptoms and suicidal ideation within 1 hour of administration
- Standard antidepressant regimen: IV infusion of 0.5 mg/kg over 40 minutes
- Duration of effect: days to ~2 weeks after a single injection
- Esketamine (Spravato) — FDA-approved intranasal formulation for treatment-resistant depression and MDD with acute suicidal ideation
- Mechanism of antidepressant effect is unclear; may not be purely NMDA antagonism (memantine, another NMDA antagonist, does not improve depression), suggesting dopaminergic/adrenergic contributions
Status Epilepticus
- Ketamine is of substantial benefit in refractory status epilepticus (despite paradoxically increasing limbic activity, it does not cause clinical seizures)
— Barash Clinical Anesthesia, 9e; Miller's Anesthesia, 10e; Kaplan & Sadock's Comprehensive Textbook of Psychiatry
5. Side Effects
Central Nervous System
- Psychomimetic/emergence reactions (most significant): hallucinations, vivid dreams, out-of-body experiences, delirium — occurs in 10–20% of patients; can be frightening. Attenuated by co-administration of benzodiazepines (most effective) or droperidol
- Nystagmus (lateral gaze nystagmus is characteristic)
- Increased salivation and lacrimation
- Purposeless limb/trunk movements (increased skeletal muscle tone)
- Increased cerebral metabolic rate (CMR) and cerebral blood flow (CBF) — by ~25–36%; use with caution in patients with impaired intracranial compliance unless combined with sedatives
- Increased intraocular pressure — contraindicated in open-eye injuries
Cardiovascular
- ↑ Heart rate, ↑ blood pressure, ↑ cardiac output (sympathomimetic): increases myocardial oxygen consumption — potentially harmful in ischemic heart disease or vascular aneurysms
- Paradoxically, direct myocardial depression (masked by sympathetic stimulation); if catecholamine stores are depleted (prolonged shock, severe sepsis), cardiovascular collapse can occur
- ↑ Pulmonary vascular resistance — use cautiously in severe right heart dysfunction or elevated pulmonary artery pressure
Respiratory
- Generally preserves spontaneous respiration and airway reflexes
- Can cause transient apnea with rapid IV injection or high doses
- Bronchodilation (beneficial in asthma)
Contraindications
| Condition | Reason |
|---|---|
| Uncontrolled ↑ ICP (spontaneously breathing) | Further ICP elevation possible |
| Open globe / ocular injury | ↑ intraocular pressure |
| Ischemic heart disease (sole anesthetic) | ↑ myocardial O₂ demand |
| Vascular aneurysms | Sudden hypertension risk |
| Schizophrenia / psychotic disorders | Worsens psychosis |
| Neonates (caution) | NMDA antagonists may accentuate neuroapoptosis |
| Intrathecal/epidural (chlorobutanol-preserved formulation) | Chlorobutanol is neurotoxic |
Key references:
- Miller's Anesthesia, 2-Volume Set, 10e — pp. 2552–2567
- Barash, Cullen, and Stoelting's Clinical Anesthesia, 9e — pp. 1487–1492
- Kaplan & Sadock's Comprehensive Textbook of Psychiatry
- Kaplan and Sadock's Synopsis of Psychiatry
This is a shared conversation. Sign in to Orris to start your own chat.