Hemolytic uremic syndrome
Reading File
Finding Sources
Finding Sources
Searching Images
hemolytic uremic syndrome pathology blood smear
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
I now have comprehensive information from multiple authoritative sources. Let me compile the full answer.
Hemolytic Uremic Syndrome (HUS)
Definition
HUS is a thrombotic microangiopathy (TMA) characterized by the clinical triad of:
- Microangiopathic hemolytic anemia (MAHA)
- Thrombocytopenia
- Acute kidney injury (AKI)
It is the most common cause of AKI requiring dialysis/transplantation in children aged 1–5 years, with an annual incidence of ~6.1 cases per 100,000 population.
— Andrews' Diseases of the Skin, p. 3327; Tintinalli's Emergency Medicine, p. 2112
Classification
Typical HUS (Epidemic, Diarrhea-Positive, STEC-HUS)
- ~90% of pediatric cases
- Caused by Shiga toxin-producing E. coli (STEC), most commonly O157:H7
- Preceded by a prodrome of bloody diarrhea
- Source: undercooked meat, unpasteurized milk, contaminated produce
- 10–15% of children with STEC infection develop HUS
Atypical HUS (aHUS, Diarrhea-Negative, Nonepidemic)
- Complement dysregulation is the dominant mechanism
- Triggers include: S. pneumoniae infection (~5%), pregnancy/postpartum, OCP use, antiphospholipid syndrome, systemic sclerosis, genetic mutations
- Worse prognosis than typical HUS
— Tintinalli's Emergency Medicine, p. 2113; Robbins, Cotran & Kumar, p. 853–854
Pathophysiology
Typical HUS (Shiga Toxin-Mediated)
Shiga-like toxins (resembling Shigella dysenteriae toxins) are absorbed from the gut into the circulation and:
- Activate endothelial cells → increased leukocyte adhesion molecules and endothelin, decreased nitric oxide
- Cause endothelial apoptosis (facilitated by cytokines such as TNF)
- Inhibit Factor H (a complement regulatory protein) → possible complement hyperactivation
- Net result: platelet activation, vasoconstriction, microthrombus formation in renal vessels → glomerular ischemia, oliguria, AKI
— Robbins, Cotran & Kumar, p. 853–854
Atypical HUS (Complement-Mediated)
Mutations in alternative complement pathway regulatory proteins allow uncontrolled C3b activity and complement activation on endothelial surfaces:
| Mutation | Protein Function | Prognosis |
|---|---|---|
| Factor H (CFH) — most common | Degrades C3b; major downregulator of alternative pathway | Death/ESRD >50%; recurrence >75% |
| Factor I (CFI) | Cleaves C3b and C4b | Death/ESRD >50%; recurrence >75% |
| Membrane Cofactor Protein (CD46/MCP) | Cofactor for CFI | Better prognosis |
| Complement Factor B | Component of C3 convertase | Severe |
| Thrombomodulin | Activates complement regulators | Death/ESRD >50% |
| C3 itself | Direct gain-of-function | Severe |
| Anti-CFH autoantibodies (~6%) | Block CFH activity | Autoimmune HUS |
Heterozygous CFH mutations are most common; abnormal CFH complexes with normal CFH, inactivating it. Compound heterozygotes (mutations in two genes) present earlier.
— Andrews' Diseases of the Skin, p. 3333–3334
S. pneumoniae-Associated HUS
Pneumococcal neuraminidase cleaves N-acetylneuraminic acid from RBC and endothelial cell surfaces, exposing the Thomsen-Friedenreich (T) antigen → immune response → TMA. Higher mortality than STEC-HUS.
— Tintinalli's Emergency Medicine, p. 2117
Renal Pathology (Morphology)
In acute disease:
- Patchy or diffuse cortical necrosis and subcapsular hemorrhages (grossly)
- Glomeruli: thickened capillary walls, mesangiolysis, microthrombi (fibrin/platelet) in capillary loops
- Arterioles and small arteries: endothelial swelling and detachment, subendothelial accumulation of fluffy material, RBC fragmentation ("onion-skin" lesions in chronic/recurrent disease)
- Renal vessels are especially vulnerable because the exposed subendothelial membrane is susceptible to complement-mediated damage
The morphologic findings of typical HUS, atypical HUS, and TTP are indistinguishable from each other on biopsy alone.
— Robbins, Cotran & Kumar, p. 853–855; Andrews' Diseases of the Skin, p. 3331
Clinical Features
| Feature | Typical HUS | Atypical HUS |
|---|---|---|
| Age | Children <10 y (most common) | Any age; 67% childhood |
| Prodrome | Bloody diarrhea (1 week prior) | Variable; no diarrhea |
| Fever | Usually absent | Variable |
| Renal involvement | Universal; hallmark | Universal; often severe |
| Neurologic symptoms | <50% | Possible |
| Skin | Unusual; petechiae/retiform purpura | Similar |
| Recurrence | Low | High (CFH/CFI >75%) |
Symptoms after GI prodrome resolves (~1 week): pallor, edema, oliguria, shortness of breath, seizures/encephalopathy. Other complications: hypertension, heart failure, intussusception, DM, metabolic acidosis, colitis.
— Tintinalli's Emergency Medicine, p. 2119; Andrews' Diseases of the Skin, p. 3329
Laboratory Evaluation
| Test | Finding |
|---|---|
| Hemoglobin | 5–9 g/dL (severe MAHA) |
| Peripheral smear | Schistocytes, helmet cells, burr cells (fragmented RBCs) |
| Platelet count | <150,000/mm³ |
| WBC | May be elevated (poor prognosis) |
| Coombs test | Negative (mechanical/non-immune hemolysis) |
| Electrolytes | Hyponatremia, hyperkalemia, metabolic acidosis |
| Bilirubin | Elevated (hemolysis) |
| Renal function | Elevated creatinine, BUN |
| Urinalysis | Hematuria (gross or micro), granular/hyaline casts, proteinuria |
| Stool culture | For STEC; test for Shiga toxin |
| ADAMTS13 | Normal (distinguishes from TTP) |
Peripheral smear showing schistocytes — a hallmark of microangiopathic hemolytic anemia:
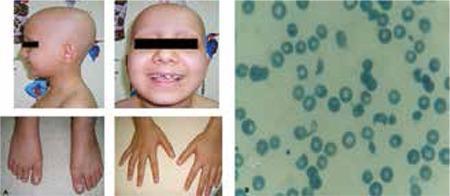
— Tintinalli's Emergency Medicine, p. 2121–2123
HUS vs. TTP
| Feature | HUS | TTP |
|---|---|---|
| Dominant organ | Kidney (severe AKI) | Brain (neurologic symptoms) |
| ADAMTS13 | Normal | Very low (<10%) |
| Mechanism | Shiga toxin / complement | ADAMTS13 deficiency → vWF multimers |
| Fever | Usually absent | Present (part of pentad) |
| Renal failure | Common, severe | Uncommon |
| Treatment response to plasma exchange | Moderate | Excellent (>80% response) |
— Robbins, Cotran & Kumar, p. 854
Treatment
Typical HUS (STEC-HUS)
- Supportive care is the primary approach
- IV fluids (early aggressive volume expansion, 10–20 mL/kg NS) may improve outcomes in the absence of fluid overload
- Correct electrolyte disturbances (hyperkalemia, hyponatremia)
- Blood transfusion for severe anemia
- Platelet transfusion: contraindicated — worsens thrombotic process
- Antibiotics: contraindicated in diarrheal illness — increase risk of HUS (possibly by releasing more toxin)
- Antiperistaltic agents: contraindicated
- Renal replacement therapy required in ~50% of acute cases
- Eculizumab or plasma exchange may be helpful (evidence remains limited for STEC-HUS)
- ~95% of children survive the acute phase; most regain normal renal function
Atypical HUS (aHUS)
- Eculizumab (humanized anti-C5 monoclonal antibody): FDA-approved for complement-mediated aHUS; proven effective for CFH and CFI mutations — removes terminal complement activation
- Cost: ~$400,000/year (a practical consideration)
- Plasma exchange: removes autoantibodies and provides functional complement factors, but less effective than in TTP
- Immunosuppressants (corticosteroids, azathioprine, MMF, rituximab, vincristine): used in atypical/autoimmune HUS
- Kidney transplantation: role is unclear — high recurrence risk; success depends on mutation type (MCP mutations have best outcomes post-transplant)
- OCP use should be avoided — can trigger aHUS in patients with CFH (8%) and CFI (20%) mutations
Disposition
- All HUS patients require hospitalization
- Neurologic symptoms + oliguric renal failure → ICU admission
— Tintinalli's Emergency Medicine, p. 2129–2132; Andrews' Diseases of the Skin, p. 3337–3339
Prognosis
| Type | Acute mortality | Long-term |
|---|---|---|
| Typical (STEC-HUS) | ~5% in children | Only 40% normal renal function at 15–25 years |
| S. pneumoniae-HUS | Higher than STEC | Worse renal outcomes |
| aHUS (CFH/CFI mutations) | High | >50% death or ESRD; recurrence >75% |
| aHUS (MCP mutations) | Lower | Better prognosis |
Despite surviving the acute phase, long-term renal outcomes in typical HUS are more guarded than often assumed — in one study, only 10 of 25 patients had normal renal function at 15–25 year follow-up.
— Robbins, Cotran & Kumar, p. 853–854; Andrews' Diseases of the Skin, p. 3333–3335
This is a shared conversation. Sign in to Orris to start your own chat.