Gastric ulcer vs duodenal ulcer in detail with reference to standard textbooks
Reading File
Finding Sources
Finding Sources
Searching Images
gastric ulcer vs duodenal ulcer comparison diagram
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive data from multiple authoritative sources. Let me compile the detailed comparison.
Gastric Ulcer vs Duodenal Ulcer — A Detailed Comparison
1. Definition
A peptic ulcer is a chronic mucosal defect that penetrates through the muscularis mucosae, occurring in areas exposed to acid-pepsin. The final common pathway is an imbalance between aggressive factors (HCl, pepsin, H. pylori, NSAIDs) and defensive factors (mucus, bicarbonate, prostaglandins, mucosal blood flow, epithelial renewal).
"Duodenal ulcer was traditionally viewed as a disease of increased acid-peptic action on the duodenal mucosa, whereas gastric ulcer was viewed as a disease of weakened mucosal defenses." — Schwartz's Principles of Surgery, 11th ed.
2. Epidemiology
| Feature | Gastric Ulcer (GU) | Duodenal Ulcer (DU) |
|---|---|---|
| Relative frequency | Less common | More common (~4:1 ratio) |
| Sex | Equal in men and women | ~2× more common in men |
| Age of onset | Older patients (mean ~55–65 yrs) | Younger patients (mean ~45–55 yrs) |
| Lifetime risk | ~4% (females), ~10% (males) overall PUD | DU predominates in younger cohorts |
| Trends | Increasing in elderly (NSAID-driven) | Declining as H. pylori prevalence falls |
| Malignant potential | Yes — must biopsy to exclude cancer | Virtually never malignant |
"On average, gastric ulcer patients are older than duodenal ulcer patients, and the incidence is increasing in the elderly, perhaps because of increasing NSAID and aspirin use." — Schwartz's Principles of Surgery, 11th ed.
3. Location
Gastric Ulcer
- Most commonly on the lesser curvature of the stomach, at the junction of the body and antrum (angularis incisura / incisura angularis)
- The Johnson classification of gastric ulcers by site:
- Type I — Lesser curvature at the incisura (most common, ~60%); normal or ↓ acid
- Type II — Body of stomach + concurrent duodenal ulcer; associated with ↑ acid
- Type III — Prepyloric; associated with ↑ acid (behaves like duodenal ulcer)
- Type IV — High on lesser curvature, near gastroesophageal junction; normal or ↓ acid
- Type V — Anywhere in stomach; NSAID-induced
Duodenal Ulcer
- First part of the duodenum (duodenal bulb), within a few centimetres of the pyloric valve
- The anterior wall of the duodenal bulb is the most common site
"Peptic ulcers are most common in the gastric antrum and first portion of the duodenum." — Robbins & Kumar Basic Pathology, 11th ed.
4. Pathophysiology
Gastric Ulcer
The dominant mechanism is defective mucosal defense:
- H. pylori colonizes the antrum, releases CagA toxin and urease → breaks down the mucus-bicarbonate barrier and causes direct epithelial injury
- NSAIDs inhibit COX-1 → ↓ prostaglandin E₂ → ↓ mucus and bicarbonate secretion → ↓ mucosal blood flow
- Acid secretion is normal or reduced in Type I GU (parietal cell mass is not increased)
- Bile reflux from the duodenum contributes to mucosal damage in some patients
- Smoking ↓ mucosal blood flow and ↓ prostaglandin synthesis
Duodenal Ulcer
The dominant mechanism is acid hypersecretion delivered to the duodenum:
- H. pylori preferentially infects the antrum → antral gastritis → ↑ gastrin release → ↑ parietal cell stimulation → elevated BAO and MAO
- Parietal cell mass may be increased (~2× normal)
- H. pylori also suppresses somatostatin-secreting D cells → loss of inhibitory brake on gastrin
- Increased rate of gastric emptying → larger acid load per unit time reaches duodenum
- Decreased duodenal bicarbonate secretion (due to H. pylori) impairs neutralization
- Gastric metaplasia in the duodenum → H. pylori can colonize duodenum → local mucosal injury
"Gastric ulcers form primarily because the mucosal barrier is defective... In duodenal ulcers, patients produce more acid than normal controls in response to any known acid secretory stimulus." — Costanzo Physiology, 7th ed. / Schwartz's Surgery, 11th ed.
5. Acid Secretion Profile
| Parameter | Gastric Ulcer | Duodenal Ulcer |
|---|---|---|
| Basal acid output (BAO) | Normal or ↓ (Type I) | ↑ (mean higher than controls) |
| Peak/maximal acid output (MAO) | Normal or ↓ | ↑ |
| Fasting serum gastrin | Normal or ↑ | Normal (but inappropriately high) |
| Parietal cell mass | Normal or ↓ | Often ↑ (up to 2×) |
| Gastric emptying rate | Normal or delayed | Often accelerated |
| Duodenal bicarbonate | Normal | ↓ |
"Patients with type I gastric ulcer usually have normal or decreased acid secretion." — Schwartz's Principles of Surgery, 11th ed.
6. Gross Morphology
Both types share key features (from Robbins & Cotran Pathologic Basis of Disease):
- Solitary in >80% of patients
- Round to oval, sharply punched-out defect with clean, smooth base
- Mucosal margin is flush with surrounding mucosa (NOT heaped up — raised margins suggest carcinoma)
- Mucosal folds radiate from the ulcer crater ("radiating folds" on barium study)
- Scarring may involve full thickness, drawing surrounding mucosa inward
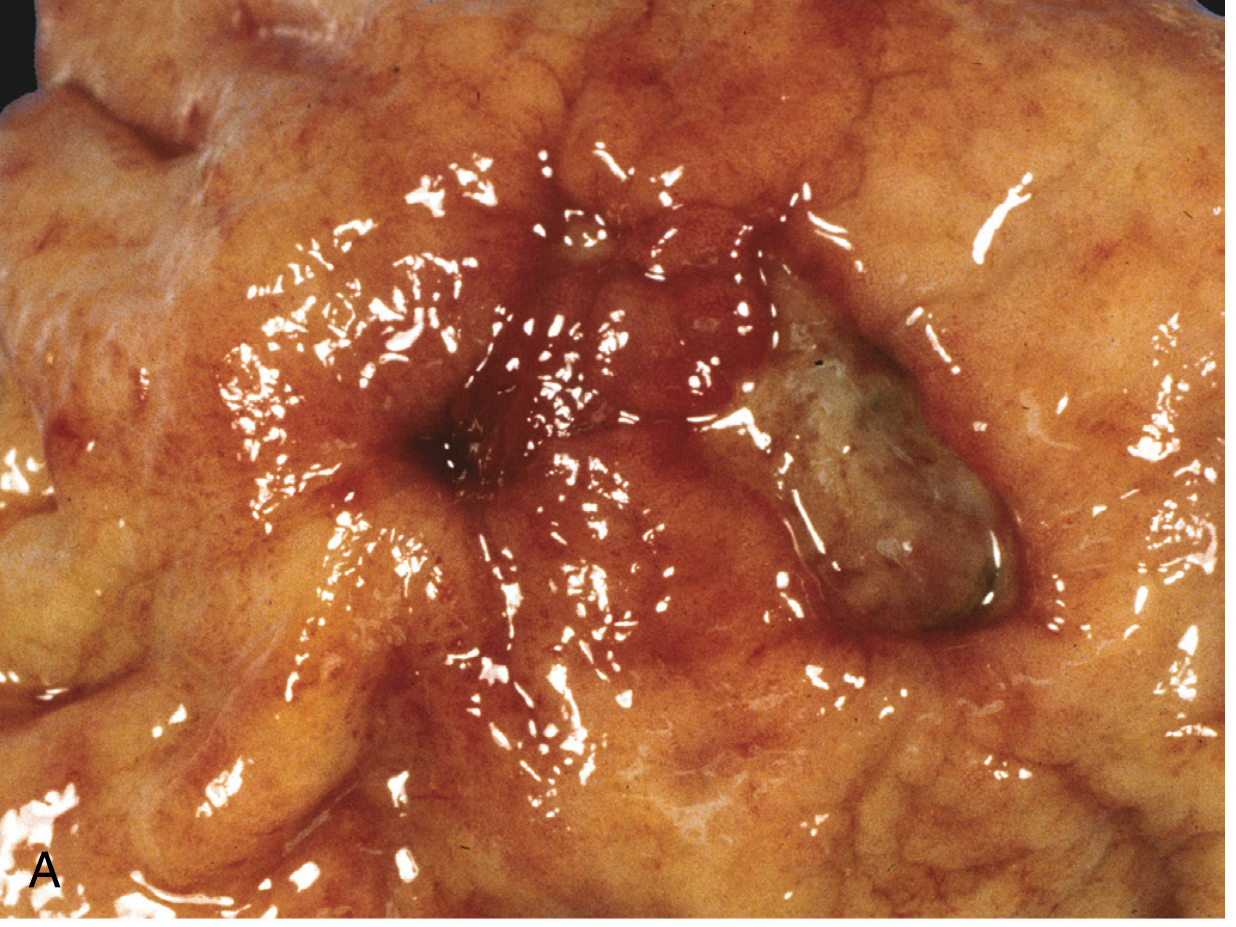
7. Histology
Four zones from surface to base (Robbins & Cotran):
- Thin fibrinoid necrosis — on the luminal surface
- Neutrophilic inflammatory infiltrate — active inflammation
- Granulation tissue — immature vessels, mononuclear leukocytes
- Collagenous/fibrous scar at the base — thickened, occasionally thrombosed vessels (source of life-threatening haemorrhage)
8. Clinical Features
| Feature | Gastric Ulcer | Duodenal Ulcer |
|---|---|---|
| Pain timing | With meals (food worsens pain) | 2–3 hours after meals; nocturnal pain (hunger pain) |
| Pain relief with food | No — food may worsen pain | Yes — food initially relieves pain |
| Nocturnal pain | Less common | Common — wakes patient from sleep (~2/3 of patients) |
| Weight | Weight loss (pain with eating → food avoidance) | May gain or maintain weight (eating relieves pain) |
| Nausea/vomiting | More common | Less common unless obstructed |
| Pain quality | Epigastric, burning, gnawing | Epigastric, burning, gnawing |
| Periodicity | Variable | Characteristic periodic flares (weeks on/weeks off) |
"Patients with duodenal ulcer often experience pain 2 to 3 hours after a meal and at night. Two-thirds of patients with duodenal ulcers will complain of pain that awakens them from sleep. The pain of gastric ulcer more commonly occurs with eating and is less likely to awaken the patient at night." — Schwartz's Principles of Surgery, 11th ed.
9. Complications
| Complication | Gastric Ulcer Notes | Duodenal Ulcer Notes |
|---|---|---|
| Haemorrhage | From left gastric artery branches | From gastroduodenal artery (posterior DU) — life-threatening |
| Perforation | Less common; anterior GU can perforate into lesser sac | Anterior DU perforates into peritoneal cavity; free air under diaphragm |
| Penetration | Into lesser omentum, liver, colon | Into pancreas (posterior DU) → pancreatitis, epigastric pain radiating to back |
| Gastric outlet obstruction | Prepyloric/pyloric GU | Pyloric/duodenal scarring; more common in DU |
| Malignant transformation | Possible (must biopsy!) | Virtually zero risk |
"The significant difference is the possibility of malignancy in a gastric ulcer. This critical difference necessitates... biopsies." — Sabiston Textbook of Surgery, 21st ed.
10. H. pylori and Aetiology
| Gastric Ulcer | Duodenal Ulcer | |
|---|---|---|
| H. pylori association | ~60–70% | ~90–95% |
| NSAID association | High (especially Type V) | Moderate |
| Mechanism with H. pylori | Mucosal barrier disruption | Acid hypersecretion + ↓ bicarbonate |
| Zollinger-Ellison syndrome | Can occur (multiple, refractory) | Can occur (multiple, refractory) |
"More than 70% of PUD cases are associated with H. pylori infection." — Robbins & Kumar Basic Pathology, 11th ed.
H. pylori uses urease to generate NH₃, alkalinising its local environment and allowing survival in the acidic stomach. The ¹³C-urea breath test exploits this: the patient ingests ¹³C-urea → H. pylori urease converts it to ¹³CO₂ → detected in expired breath.
11. Diagnosis
Upper GI endoscopy (EGD) is the gold standard.
- All gastric ulcers must be biopsied (multiple biopsies from margin and base) to exclude malignancy
- Duodenal ulcers do not require routine biopsy unless atypical
- Biopsy antrum/body for H. pylori (rapid urease test, histology)
Non-invasive H. pylori tests: ¹³C-urea breath test, stool antigen test, serology
Barium meal (upper GI series):
- GU: niche/crater on lesser curvature, radiating folds, Hampton's line (thin lucent line across ulcer neck)
- DU: deformity of duodenal cap ("cloverleaf" deformity), spasm, constant filling defect
Alarm features requiring urgent endoscopy: age >55, dysphagia, weight loss, haematemesis/melaena, anaemia, palpable mass.
12. Medical Treatment
Both types:
- PPI therapy (omeprazole, lansoprazole, pantoprazole) — mainstay; heals both GU and DU
- DU: 4–6 weeks; GU: 8–12 weeks (needs endoscopic confirmation of healing)
- H. pylori eradication — Triple or quadruple therapy; dramatically reduces recurrence
- Discontinue NSAIDs/aspirin where possible; if continued, add PPI co-prescription
- Lifestyle: stop smoking, avoid alcohol and ulcerogenic drugs
"Helicobacter treatment dramatically decreases the recurrence rate of both duodenal and gastric ulcer." — Schwartz's Principles of Surgery, 11th ed.
13. Surgical Treatment
Surgery is now reserved for complications (perforation, uncontrolled haemorrhage, obstruction, intractable disease).
| Procedure | Gastric Ulcer | Duodenal Ulcer |
|---|---|---|
| Perforation | Omental patch + biopsy; gastrectomy if suspicious | Graham patch (omental patch) |
| Haemorrhage | Oversewing / wedge excision | Duodenotomy + ligation of GDA |
| Elective resection | Gastrectomy (Billroth I preferred) for Type I; include ulcer in specimen | Highly selective vagotomy (HSV); vagotomy + drainage |
| Type II/III GU | Acid-reducing procedure required (as for DU) | — |
14. Summary Comparison Table
| Feature | Gastric Ulcer | Duodenal Ulcer |
|---|---|---|
| Site | Lesser curvature, incisura angularis | First part of duodenum (bulb) |
| Frequency | Less common | More common |
| Age | Older | Younger |
| Sex | M = F | M > F (2:1) |
| Acid secretion | Normal or ↓ | ↑ (↑ BAO, ↑ MAO) |
| Primary pathogenesis | Defective mucosal defense | Acid hypersecretion |
| H. pylori | ~70% | ~95% |
| Pain relation to food | Precipitated/worsened by food | Relieved by food; returns 2–3 hr later |
| Nocturnal pain | Uncommon | Common (wakes from sleep) |
| Weight | Loss | May maintain/gain |
| Malignancy risk | Present — always biopsy | Absent |
| Haemorrhage source | Left gastric artery | Gastroduodenal artery |
| Perforation site | Anterior → peritoneum | Anterior → peritoneum; posterior → pancreas |
| Healing duration (PPI) | 8–12 weeks | 4–6 weeks |
| Endoscopic biopsy | Mandatory | Not routinely required |
Key References:
- Schwartz's Principles of Surgery, 11th Edition — Chapter 26
- Robbins & Cotran Pathologic Basis of Disease, 10th Edition — Chapter 17
- Robbins & Kumar Basic Pathology, 11th Edition — Chapter 13
- Costanzo Physiology, 7th Edition — Chapter 8
- Guyton and Hall Textbook of Medical Physiology — Chapter 67
- Sabiston Textbook of Surgery, 21st Edition — Chapter on Gastric and Duodenal Ulcers
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease — Chapter 20/53
Triple therapy
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Excellent — I now have comprehensive data from Harrison's (22nd ed.), Yamada's Gastroenterology (7th ed.), and Schwartz's Surgery. Here is the full answer.
Triple Therapy for H. pylori Eradication
Background
The concept of triple therapy arose because monotherapy and dual therapy were inadequate for reliable H. pylori eradication. Triple therapy became the dominant worldwide strategy for treating H. pylori infection, which underlies the vast majority of peptic ulcer disease.
"Failure of monotherapy and dual therapy for treatment of H. pylori infection prompted the development of triple therapy and other multidrug regimens." — Yamada's Textbook of Gastroenterology, 7th ed.
Classic Triple Therapy — Components
Triple therapy = 1 PPI + 2 antibiotics, given twice daily for 7–14 days (14 days now preferred in most guidelines, especially North America).
The Three Standard Regimens
| Regimen | Drugs | Dose | Frequency |
|---|---|---|---|
| PAC (Clarithromycin-based) | PPI + Amoxicillin + Clarithromycin | PPI std dose; Amox 1 g; Clarithr 500 mg | Twice daily |
| PCM (Penicillin-allergy) | PPI + Clarithromycin + Metronidazole | PPI std dose; Clarithr 250–500 mg; Metro 400 mg | Twice daily |
| PAM (Non-clarithromycin) | PPI + Amoxicillin + Metronidazole | PPI std dose; Amox 1 g; Metro 400 mg | Twice daily |
PAC and PCM are clinically equivalent. PCM is used in penicillin-allergic patients. PAM (without clarithromycin) is generally not recommended as first-line — less effective in most studies.
(Yamada's Gastroenterology, 7th ed., Table 49.5)
PPI Options
Any standard PPI can be used twice daily:
| PPI | Standard Dose |
|---|---|
| Omeprazole | 20 mg BD |
| Lansoprazole | 30 mg BD |
| Pantoprazole | 40 mg BD |
| Rabeprazole | 20 mg BD |
| Esomeprazole | 40 mg BD |
| Dexlansoprazole | 60 mg once daily |
Role of PPI in triple therapy:
- Raises intragastric pH → creates environment less hostile to antibiotics
- Increases half-life and stability of clarithromycin and amoxicillin in gastric mucus
- H. pylori is in a replicative state at higher pH → more susceptible to bactericidal antibiotics
- Has synergistic bactericidal effect
"Acid suppression with PPI has synergistic bactericidal effects and stabilizes antibiotics, so increasing their half-life... H. pylori is more likely in a nonreplicative state in low gastric pH but is more replicative and therefore susceptible to amoxicillin and clarithromycin when the pH rises." — Yamada's Gastroenterology, 7th ed.
A pre-packaged formulation Prevpac (lansoprazole 30 mg + clarithromycin 500 mg + amoxicillin 1 g, all BD × 14 days) is FDA-approved. — Harrison's Principles of Internal Medicine, 22nd ed.
Duration
| Duration | Comment |
|---|---|
| 7 days | Original regimen; lower eradication rates; now generally insufficient |
| 10 days | Intermediate; used in some European guidelines |
| 14 days | Currently recommended (North America, Maastricht V/Florence); higher cure rates |
"Simpler (dual therapy) and shorter regimens (7 and 10 days) are not as effective as triple therapy for 14 days." — Harrison's Principles of Internal Medicine, 22nd ed.
Mechanism of Each Drug
| Drug | Mechanism | Relevance |
|---|---|---|
| PPI | Irreversible H⁺/K⁺-ATPase blockade → ↑ pH | Creates permissive environment for antibiotic activity |
| Amoxicillin | Inhibits bacterial cell wall synthesis (PBP binding) | Bactericidal; low resistance rates globally |
| Clarithromycin | Macrolide; inhibits 50S ribosomal subunit → blocks translocation | Key bactericidal agent; rising resistance is the main problem |
| Metronidazole | Reduced to toxic free radicals in anaerobic organisms → DNA strand breaks | Used when penicillin allergy or as clarithromycin alternative |
Efficacy and the Resistance Problem
Historically, triple therapy achieved eradication rates of >90%. Currently, rates have fallen significantly in many countries due to clarithromycin resistance:
- Cure rates of <80% (and in some regions <50%) are now widely reported — in Greece, India, Italy, Japan, Mexico, and Spain
- Clarithromycin resistance rates of >15% are now common across most of Europe, Asia, and the Americas
- Metronidazole resistance is also significant but has less clinical impact (metronidazole resistance can be partially overcome by higher doses)
"Using triple therapy as first-line treatment, cure rates of less than 80% (and even below 50%) are now widely reported across countries." — Yamada's Textbook of Gastroenterology, 7th ed.
Maastricht V/Florence Consensus (and ACG guidelines) stratify regions by clarithromycin resistance:
| Resistance Rate | Recommendation |
|---|---|
| <15% (low resistance) | Clarithromycin-based triple therapy acceptable first-line (14 days) |
| >15% (high resistance) | Triple therapy should NOT be used first-line; prefer bismuth quadruple therapy |
| Prior macrolide exposure | Always avoid clarithromycin-containing triple therapy regardless of regional rates |
Causes of Triple Therapy Failure
- Clarithromycin resistance — most important cause
- Poor patient adherence — high pill burden / side effects
- Inadequate acid suppression (especially in CYP2C19 extensive metabolisers who rapidly metabolise PPIs)
- Metronidazole resistance (when PAM used)
- Reinfection (rare in developed countries)
"Failure of H. pylori eradication with triple therapy in a compliant patient is usually due to infection with a resistant organism. Additional important factors in treatment failure include inadequate acid suppression and inadequate adherence." — Harrison's Principles of Internal Medicine, 22nd ed.
Alternative and Rescue Regimens
When triple therapy fails or is contraindicated (high clarithromycin resistance, prior macrolide use):
1. Bismuth Quadruple Therapy (now often first-line)
PPI + Bismuth subcitrate/subsalicylate + Tetracycline + Metronidazole × 10–14 days
- Eradication rates ≥90% first-line; 79–88% as second-line
- Major drawback: 17–34 pills/day (Pylera combination capsule reduces this to 14/day)
- Metronidazole resistance has much less impact here than clarithromycin resistance does in triple therapy
2. Sequential Therapy
- Days 1–5: PPI + Amoxicillin
- Days 6–10: PPI + Clarithromycin + Tinidazole (or metronidazole)
- Eradication ~85–90% in original studies
3. Concomitant (Non-bismuth Quadruple) Therapy
- PPI + Amoxicillin + Clarithromycin + Nitroimidazole — all four drugs simultaneously × 10–14 days
- Overcomes clarithromycin resistance better than sequential therapy
4. Levofloxacin Triple Therapy (second-line)
- PPI + Amoxicillin + Levofloxacin 500 mg OD × 5–7 days
- Used when both clarithromycin and metronidazole resistance suspected
5. Rifabutin Triple Therapy (salvage/third-line)
- PPI + Amoxicillin + Rifabutin
- Reserved for multiply treatment-resistant cases; used in areas of high fluoroquinolone resistance
(Harrison's, 22nd ed., Table 335-4; Yamada's, 7th ed.)
Confirming Eradication
Eradication must always be confirmed ≥4 weeks after completing therapy (and ≥2 weeks after stopping PPI):
- ¹³C-urea breath test — preferred non-invasive test
- Stool antigen test — acceptable alternative
- Repeat endoscopy with biopsy — used when endoscopy is required anyway (e.g., gastric ulcer follow-up)
Indications for H. pylori Eradication (and Thus Triple Therapy)
| Indication | Status |
|---|---|
| Active peptic ulcer disease (gastric or duodenal) | Definite |
| History of PUD (even if healed or not actively treating) | Definite |
| Gastric MALT lymphoma (low-grade) | Definite |
| After resection for early gastric cancer | Definite |
| Dyspepsia (test-and-treat strategy) | Recommended |
| NSAID use in H. pylori–positive patients | Recommended |
| Unexplained iron deficiency anaemia | Controversial |
| Immune thrombocytopenic purpura (ITP) | Controversial |
Summary
| Feature | Detail |
|---|---|
| Regimen | PPI + Amoxicillin + Clarithromycin (PAC) — first-line in low-resistance areas |
| Duration | 14 days (current standard) |
| Target eradication | ≥85–90% |
| Main failure cause | Clarithromycin resistance |
| When to avoid | Clarithromycin resistance >15%, prior macrolide use → use bismuth quadruple instead |
| Confirm eradication | ¹³C-urea breath test ≥4 weeks post-treatment |
Key References:
- Harrison's Principles of Internal Medicine, 22nd Edition — Chapter 335 (Table 335-4)
- Yamada's Textbook of Gastroenterology, 7th Edition — Chapter 49 (Table 49.5)
- Schwartz's Principles of Surgery, 11th Edition — Chapter 26
- Maastricht V/Florence Consensus Guidelines (Malfertheiner et al., Gut 2017/2022)
This is a shared conversation. Sign in to Orris to start your own chat.