Helminthic zoonoses (Echinococcosis. Trichinellosis).
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
Loading Image
I now have all the information needed to write a comprehensive response. Here is the full overview:
Helminthic Zoonoses: Echinococcosis & Trichinellosis
ECHINOCOCCOSIS (Hydatid Disease)
Etiology
Echinococcosis is a zoonosis caused by larval cestodes (tapeworms) of the genus Echinococcus:
| Species | Disease | Cycle |
|---|---|---|
| E. granulosus complex | Cystic echinococcosis (most common) | Pastoral: dogs ↔ sheep/cattle |
| E. multilocularis | Alveolar echinococcosis (more aggressive) | Sylvatic: foxes ↔ rodents |
| E. vogeli, E. oligarthrus | Polycystic echinococcosis (rare) | South/Central America |
Life Cycle
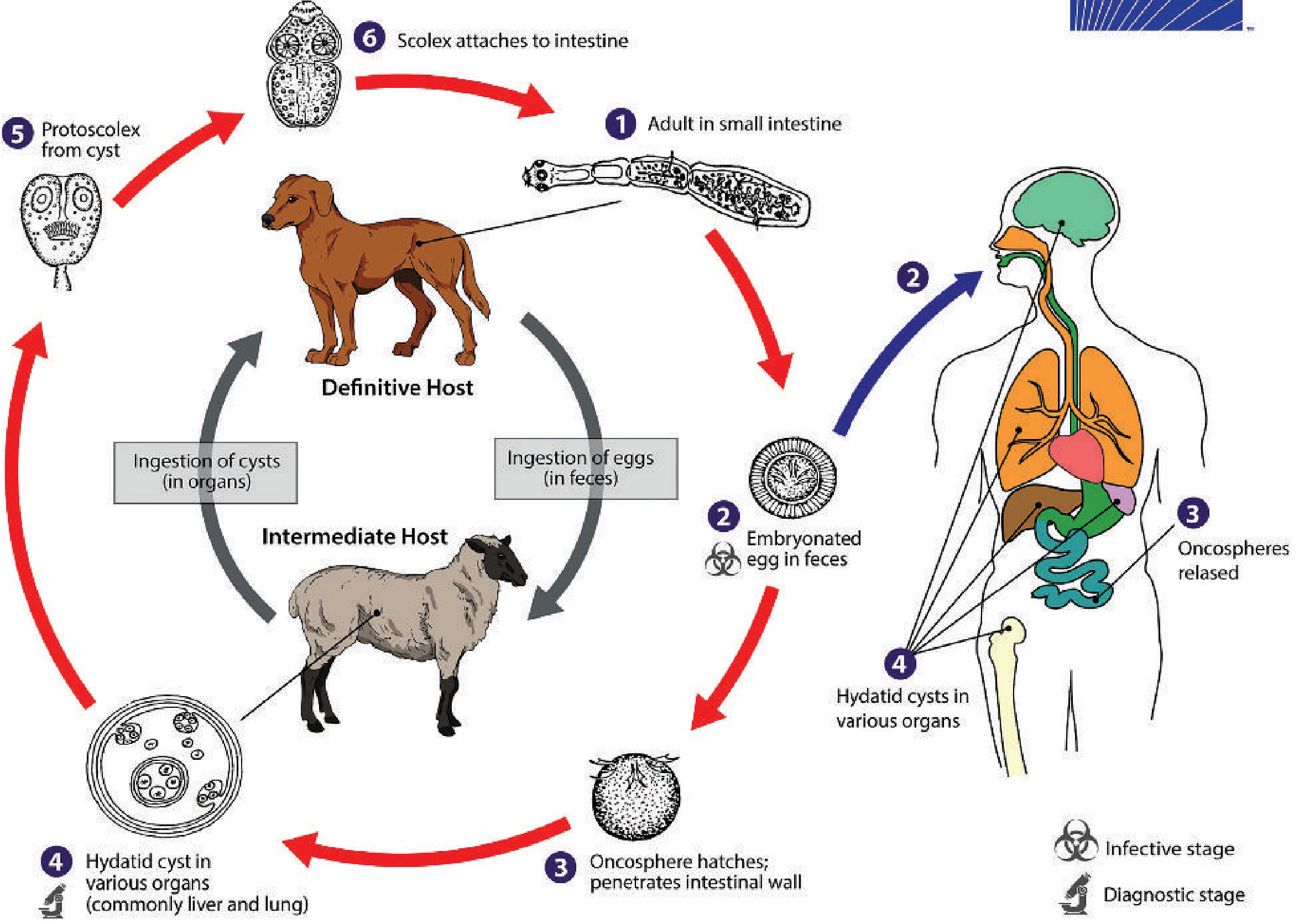
Definitive host = canids (dogs, foxes). Adult worms (3–6 mm) reside in the small intestine and shed eggs in feces.
Intermediate host = sheep, goats, pigs, horses, camels. Following ingestion of eggs, an oncosphere hatches, penetrates the intestinal wall, and travels via portal blood to form a slowly growing hydatid cyst — most commonly in the liver, less often the lungs.
Humans are accidental "dead-end" intermediate hosts who inadvertently ingest eggs from contaminated dog feces. The cyst develops identically — liver (most common), lungs, and rarely brain, bone, or spleen.
- In E. granulosus, cysts are unilocular — a single large cyst containing brood capsules with protoscoleces (immature tapeworms), sometimes with smaller daughter cysts inside.
- In E. multilocularis, the lesion is tumor-like, with numerous small cysts that invade and infiltrate adjacent structures (always nearly in the liver), and may disseminate hematogenously.
Epidemiology
- Highly endemic in eastern/southern Europe, northern Africa, Central Asia, and southern South America — especially rural sheep-raising regions where dogs are fed raw viscera.
- E. multilocularis: northern hemisphere (Europe, Asia, North America).
- WHO classifies echinococcosis among the 20 neglected tropical diseases (NTDs); >1 million people are infected worldwide.
Clinical Features
Both forms have a long asymptomatic incubation while the cyst slowly grows over years.
- Liver involvement: abdominal pain, nausea, vomiting; palpable mass.
- Lung involvement: cough, dyspnea, chest pain.
- Cyst rupture: can cause anaphylaxis and secondary seeding — potentially fatal.
- Alveolar echinococcosis: progressive weight loss, malaise, biliary obstruction, and ultimately liver failure; fatal if untreated.
Diagnosis
- Imaging (CT/ultrasound): cyst with daughter cysts or calcification is characteristic; WHO ultrasound classification (CE1–CE5) guides management.
- Serology: EIA, IFA, or indirect hemagglutination for screening; immunoblot for confirmation (cross-reactivity with T. solium cysticercosis possible). Sensitivity 60–90%; liver cysts most likely to be seropositive; brain/spleen cysts may be seronegative.
- Microscopy of aspirated/excised material: identification of protoscoleces and free hooklets ("hydatid sand" — protoscoleces + hooklets + calcareous corpuscles), confirming the diagnosis definitively.
- Protoscoleces measure 100–110 µm × 120–150 µm.
- Aspiration requires precautions to prevent spillage (secondary seeding, anaphylaxis).
Treatment
| Type | Approach |
|---|---|
| Cystic (E. granulosus) simple cysts | PAIR (Percutaneous Aspiration, Injection of scolicidal agent, Re-aspiration) + albendazole |
| Surgical | Resection for complex or ruptured cysts |
| Alveolar (E. multilocularis) | Surgery (curative resection if possible) + long-term albendazole |
| Medical alone | Albendazole; indicated when surgery not feasible |
TRICHINELLOSIS (Trichinosis)
Etiology
Caused by nematodes of the genus Trichinella. Multiple species cause human disease:
| Species | Geographic distribution |
|---|---|
| T. spiralis | Worldwide (classical agent) |
| T. pseudospiralis | Worldwide (does not encapsulate) |
| T. nativa | Arctic / north temperate |
| T. nelsoni | Africa |
| T. britovi | Europe and Western Asia |
| T. papuae | Papua New Guinea & Thailand (does not encapsulate) |
All species infect muscle cells; most encapsulate within myocytes, forming the characteristic "nurse cell," except T. pseudospiralis and T. papuae.
Life Cycle
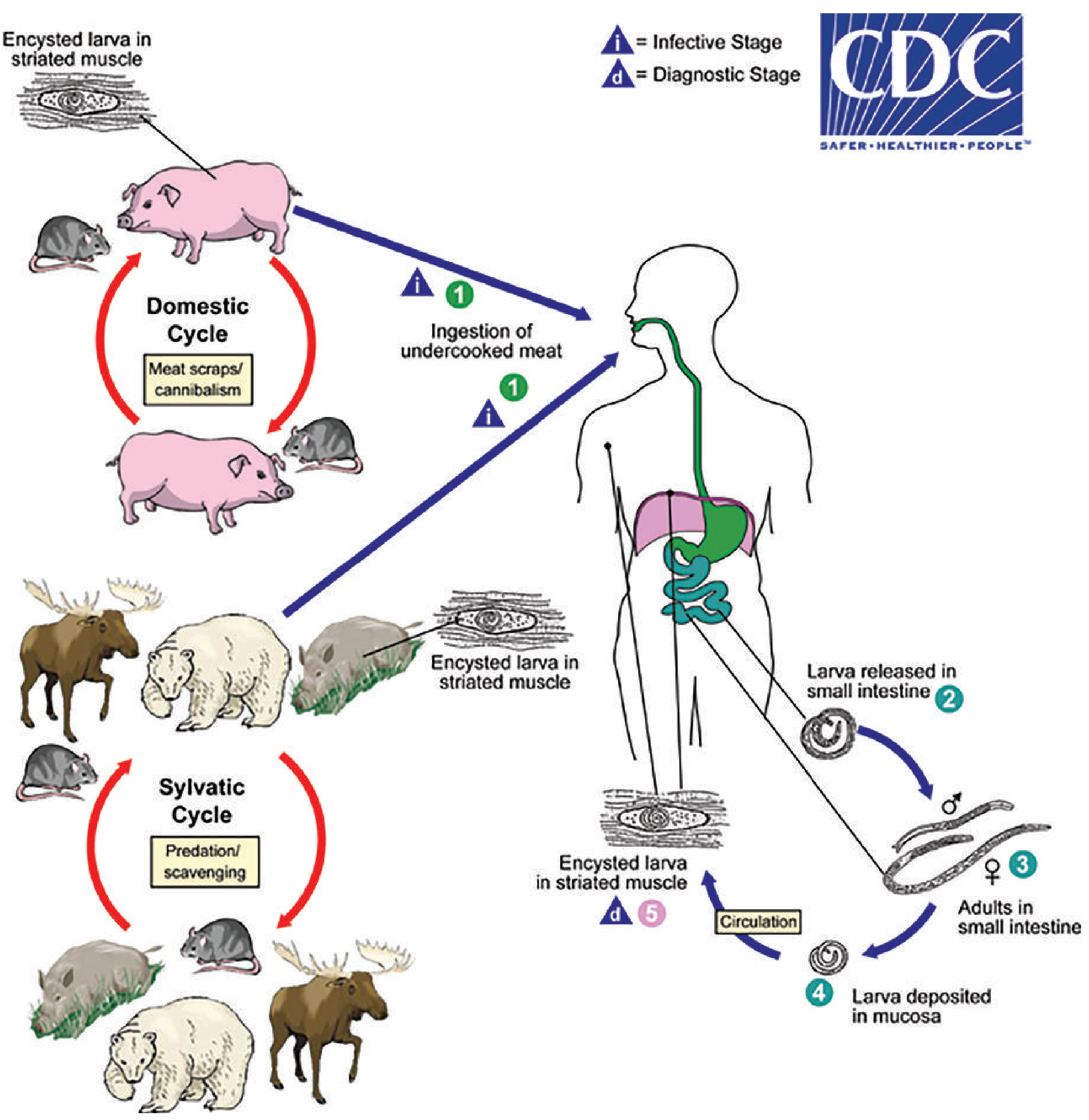
Uniquely, a single host can simultaneously be the definitive host, intermediate host, and dead-end host.
- Ingestion of undercooked meat containing viable encysted larvae (infective stage).
- Larvae released in the small intestine → mature into adult male/female worms.
- Adults mate; the female deposits larvae into the intestinal mucosa (~1 week post-infection).
- Larvae enter the bloodstream → migrate to striated muscle throughout the body.
- Larva penetrates the muscle cell → induces permanent phenotypic changes → forms the "nurse cell" (encapsulating species). The larva can survive within this cell for the life of the host; eventual calcification and death of larvae can occur.
Domestic cycle: pigs ↔ rodents → humans via undercooked pork (most common worldwide).
Sylvatic cycle: carnivores/scavengers (bears, wild boar, walrus) → humans via consumption of undercooked wild game.
Epidemiology
- Found in 66 countries. A global review (1986–2009) documented 65,818 cases from 41 countries with 42 deaths.
- 86% of cases from the European region; historically, up to 50% from Romania prior to 2000.
- Burden reduced significantly by meat inspection and veterinary health measures; remaining risk comes from free-range pigs and wild game consumption.
Clinical Features
Severity depends on the worm burden. Light infections are often asymptomatic or mildly symptomatic.
Phase 1 — Intestinal stage (days 1–7):
- Diarrhea, abdominal pain, nausea (short-lived).
Phase 2 — Larval migration/muscle invasion (from ~1 week post-infection):
- Fever, myalgia, headache
- Periorbital/facial edema (classic)
- Rash, conjunctivitis, splinter hemorrhages
- Eosinophilia (prominent)
- Elevated muscle enzymes (CPK, LDH)
Rare severe manifestations: myocarditis, encephalitis — potentially fatal.
Diagnosis
- Clinical + epidemiological: history of undercooked meat (pork, bear, walrus); outbreak association; fever + myalgia + periorbital edema + eosinophilia is the classic tetrad.
- Serology: EIA (most common) for IgM, IgG, IgE; latex agglutination. Antibodies may not be detectable until 2–5 weeks after infection — repeat testing may be needed.
- Muscle biopsy (confirmatory): deltoid or gastrocnemius; larvae seen on squash preparations or H&E sections.
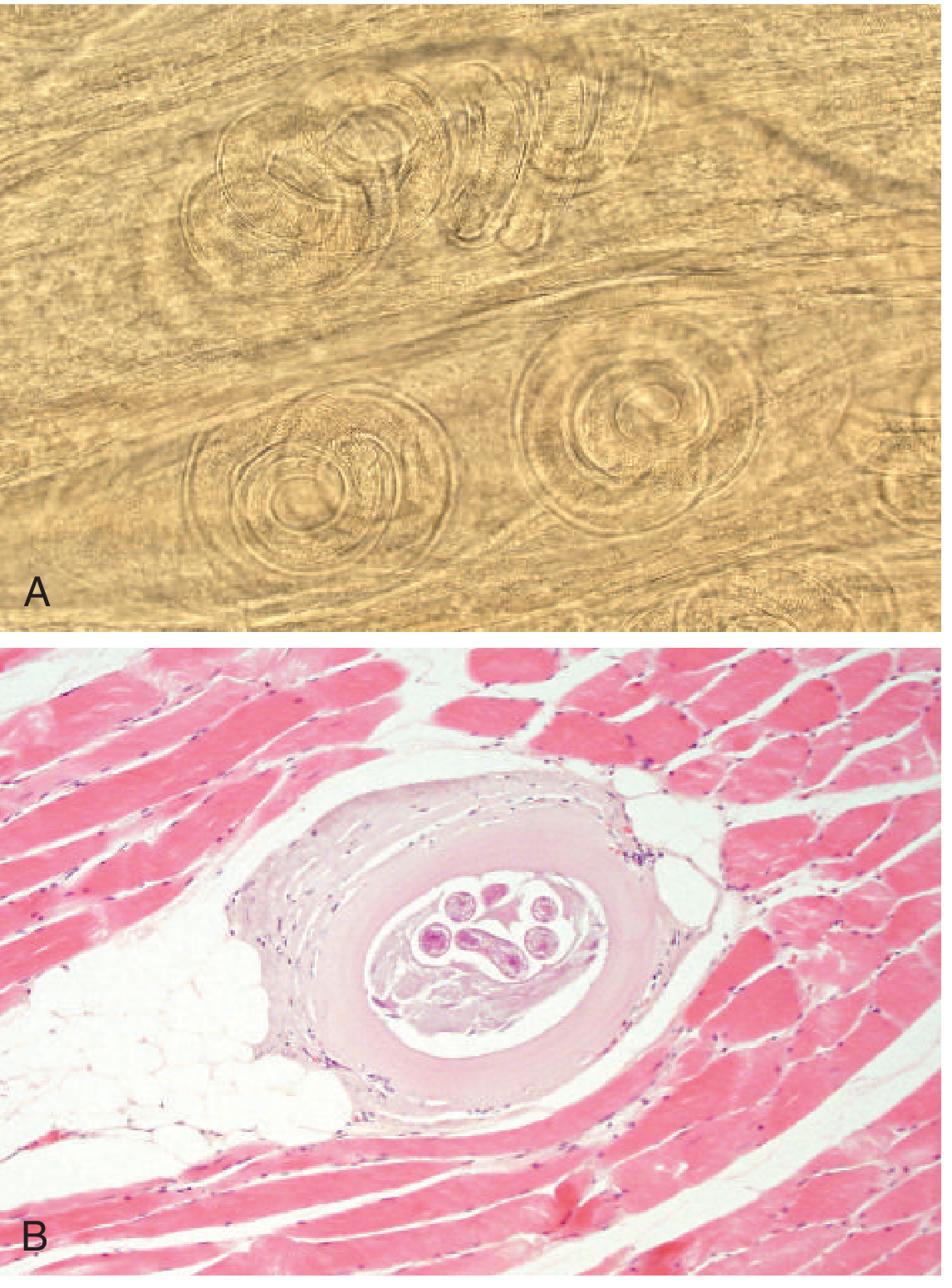
Microscopy: coiled larvae visible in squash preparations (unstained); pepsin digestion improves visualization. On H&E sections, larvae appear as cross-sections or longitudinal sections within the nurse cell.
- Molecular testing of biopsy material can identify the infecting Trichinella species.
Treatment
| Indication | Treatment |
|---|---|
| Intestinal/early stage | Albendazole or mebendazole (kills adult worms, halts larval release) |
| Established muscle larvae | Drugs have limited efficacy once larvae are encapsulated in myocytes |
| Severe inflammation | Add corticosteroids to reduce swelling |
Comparison Summary
| Feature | Echinococcosis | Trichinellosis |
|---|---|---|
| Pathogen type | Cestode (tapeworm larva) | Nematode (roundworm) |
| Key species | E. granulosus, E. multilocularis | T. spiralis (and others) |
| Definitive host | Dogs/canids | Carnivorous mammals (pigs, bears, etc.) |
| Human infection route | Ingestion of eggs in dog feces | Ingestion of encysted larvae in undercooked meat |
| Target organ(s) | Liver, lungs (cysts) | Striated muscle (larvae encyst) |
| Key pathology | Unilocular or alveolar hydatid cysts | Nurse cell formation in myocytes |
| Hallmark lab finding | "Hydatid sand" on microscopy | Eosinophilia + elevated muscle enzymes |
| Main treatment | PAIR / surgery + albendazole | Albendazole/mebendazole (± corticosteroids) |
— Tietz Textbook of Laboratory Medicine, 7th Edition (pp. 3607–3615)
Classification of helminthic zoonoses Life cycles and transmission mechanisms Pathogenesis of organ damage Clinical manifestations Diagnostic imaging and serology Differential diagnosis with tumors and abscesses Antiparasitic treatment principles Surgical indications Preventive strategies
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Here is the comprehensive structured overview across all requested topics:
Helminthic Zoonoses: Echinococcosis & Trichinellosis
A Systematic Review
1. Classification of Helminthic Zoonoses
Helminths are classified into three major phyla. The two diseases in focus occupy distinct positions:
| Phylum | Class | Order | Representative Zoonoses |
|---|---|---|---|
| Platyhelminthes (flatworms) | Cestoda (tapeworms) | Cyclophyllidea | Echinococcosis, Cysticercosis, Coenurosis |
| Nematoda (roundworms) | Adenophorea | Trichurida | Trichinellosis |
| Platyhelminthes | Trematoda (flukes) | Strigeatida | Schistosomiasis |
Echinococcosis is a larval cestode infection — humans harbor the larval (metacestode) stage, not the adult worm. Four recognized species affect humans:
| Species | Disease Form | Geographic Niche |
|---|---|---|
| E. granulosus complex | Cystic echinococcosis (CE) — most common | Global; pastoral sheep-raising zones |
| E. multilocularis | Alveolar echinococcosis (AE) — most dangerous | Northern hemisphere; arctic/alpine |
| E. vogeli | Polycystic echinococcosis | South/Central America |
| E. oligarthrus | Polycystic echinococcosis | South/Central America |
Trichinellosis is caused by tissue nematodes of the genus Trichinella (≥9 species, 13 genotypes):
| Species | Key Feature | Distribution |
|---|---|---|
| T. spiralis | Encapsulates; classical agent | Worldwide |
| T. pseudospiralis | Does not encapsulate; birds & mammals | Worldwide |
| T. nativa | Freeze-resistant | Arctic/subarctic |
| T. nelsoni | Felid predators; bush pigs | Equatorial E. Africa |
| T. britovi | Wild carnivores; not domestic swine | Europe, W. Africa, W. Asia |
| T. murrelli | Wild animals | North America, Japan |
| T. papuae | Does not encapsulate; crocodiles, pigs | Papua New Guinea, SE Asia |
| T. zimbabwensis | Crocodiles | Tanzania |
| T. patagoniensis | Cougars | South America |
2. Life Cycles and Transmission Mechanisms
Echinococcosis

Cystic Echinococcosis (E. granulosus):
- Definitive host (dogs, canids): Adult worms (3–6 mm) reside in the small intestine → shed embryonated eggs in feces.
- Intermediate host (sheep, goats, cattle, horses, camels): Eggs ingested → oncosphere hatches in duodenum → penetrates intestinal wall → transported via portal circulation → lodges in hepatic capillaries → develops into hydatid cyst; less commonly lungs.
- Canid re-infection: Dog ingests cyst-containing viscera → protoscoleces released → mature into adult worms in intestine → cycle repeats.
- Human infection (dead-end intermediate host): Accidental ingestion of eggs via contaminated food, water, soil, or direct contact with dogs → identical oncosphere–cyst development.
Alveolar Echinococcosis (E. multilocularis):
- Definitive hosts: foxes, wolves, dogs.
- Intermediate hosts: rodents (voles, lemmings).
- Humans infected by contact with soil contaminated by fox/dog feces containing ova.
- The larval mass grows by exogenous budding (not as a contained cyst), invading and infiltrating liver parenchyma like a slow-growing tumor.
Risk factors: dog/sheep ownership, rural pastoral residence, increasing age.
Trichinellosis

A single host simultaneously serves as definitive host (harbors reproducing adults), intermediate host (harbors encysted larvae), and potentially a dead-end host (if not consumed).
- Ingestion of undercooked meat containing viable encysted larvae (infective stage).
- Gastric acid and proteases liberate larvae in the stomach/duodenum.
- Larvae invade the small bowel mucosa → mature into adult males and females (~1 week).
- Mated females deposit newborn larvae into the intestinal mucosa → larvae enter the circulation.
- Larvae migrate to striated muscle throughout the body (most commonly: extraocular muscles, biceps, jaw, neck, lower back, diaphragm).
- Each larva induces permanent phenotypic transformation of the muscle cell → "nurse cell" (nurse cell supports larva for the host's lifetime; eventual calcification occurs).
- Non-encapsulating species (T. pseudospiralis, T. papuae, T. zimbabwensis) use alternative intracellular survival strategies.
- Cycle continues only if infected muscle is consumed by a carnivore/omnivore.
Transmission routes:
- Domestic cycle: undercooked pork (pigs ↔ rats) — historically most common worldwide.
- Sylvatic cycle: bear, wild boar, walrus, horse meat (via contamination or adulteration).
- Dogs (parts of Asia/Africa), horses (Italy, France), crocodiles — less common sources.
3. Pathogenesis of Organ Damage
Echinococcosis
Cystic Echinococcosis:
| Component | Pathogenic Mechanism |
|---|---|
| Hepatic cyst growth (1–3 cm/yr) | Mechanical mass effect → hepatomegaly, bile duct compression → cholestasis, jaundice, cholangitis |
| Portal vein compression | Portal hypertension |
| Cyst rupture | Release of protoscoleces → anaphylaxis (sensitized host); peritoneal seeding → secondary echinococcosis |
| Biliary communication | Intrabiliary rupture → biliary colic, obstructive jaundice, cholangitis, biliary fistula |
| Pulmonary cyst | Compression atelectasis; rupture → endobronchial spillage, aspiration pneumonia, pneumothorax, hydropneumothorax |
| Osseous cyst | Bone destruction, pathological fracture |
| CNS cyst | Mass effect → raised ICP, seizures, focal deficits |
The hydatid cyst has a distinct architecture:
- Outer (pericyst): host-derived fibrous layer — the reactive zone.
- Middle (ectocyst/laminated layer): acellular, parasite-derived, white-laminated membrane.
- Inner (endocyst/germinal layer): living parasite tissue → produces protoscoleces, brood capsules, and daughter cysts ("hydatid sand" = protoscoleces + free hooklets + calcareous corpuscles).
Alveolar Echinococcosis:
- Exogenous budding creates a tumor-like infiltrating mass of small, irregular cavities — no containing fibrous wall.
- Invades bile ducts, hepatic veins, portal vein → biliary obstruction, Budd-Chiari syndrome, liver failure.
- Hematogenous/lymphatic dissemination to lungs, brain, spleen.
- Fatal if untreated due to progressive hepatic destruction.
Trichinellosis
Damage occurs in two sequential phases:
Phase 1 — Intestinal invasion (days 1–7):
- Adult worms damage small bowel mucosa → acute enteritis with nausea, vomiting, diarrhea.
- Host immune responses attempt to expel adults (partial efficacy).
Phase 2 — Larval migration and muscle invasion (week 2–3+):
- Migrating larvae provoke marked local and systemic hypersensitivity → fever, eosinophilia, urticaria.
- Periorbital/facial edema: a classic sign caused by larval migration and immune-mediated capillary leak around orbital vessels.
- Splinter hemorrhages of the subconjunctivae, retina, and nail beds: vasculitis from migrating larvae.
- Myocarditis: larvae in cardiac muscle → tachyarrhythmias or heart failure — the principal cause of death.
- Encephalitis: larvae in CNS → focal deficits, confusion, coma.
- Pneumonitis: larval migration through lung tissue.
- Nurse cell formation: the muscle cell undergoes radical architectural transformation — sarcomeric structures are replaced, and the nurse cell becomes metabolically specialized to support the larva. With time, calcification occurs.
T. pseudospiralis (non-encapsulating) causes a prolonged polymyositis-like illness because persistent larval motility sustains inflammation.
4. Clinical Manifestations
Echinococcosis
Cystic Echinococcosis:
| Stage | Symptoms |
|---|---|
| Silent/incubation (years–decades) | Asymptomatic; cyst detected incidentally on imaging |
| Hepatic (symptomatic) | RUQ pain, hepatomegaly, nausea, vomiting; symptoms when cyst >10 cm |
| Biliary obstruction | Jaundice, cholangitis, pruritus |
| Portal hypertension | Ascites, varices |
| Cyst rupture | Sudden severe abdominal pain, urticaria, anaphylaxis, peritoneal seeding |
| Pulmonary | Cough, hemoptysis, dyspnea; rupture → expectoration of "salty water," membranes |
| CNS (~4% of cases) | Seizures, headache, raised ICP, focal neurological deficits |
| Osseous | Pathological fracture, chronic pain |
Alveolar Echinococcosis:
- Insidious onset: right upper quadrant discomfort, hepatomegaly, weight loss, malaise.
- Progressive biliary obstruction (jaundice, cholangitis).
- Eventually: hepatic failure, portal hypertension.
- Fatal without treatment; resembles hepatocellular carcinoma clinically.
Trichinellosis
Classic tetrad: fever + periorbital/facial edema + myalgia + eosinophilia.
| Phase | Timing | Manifestations |
|---|---|---|
| Intestinal | Days 1–7 | Diarrhea, cramping abdominal pain, nausea, vomiting |
| Larval migration | Week 2 | Fever, urticaria, periorbital edema, conjunctival/subretinal hemorrhages, splinter hemorrhages, headache, cough |
| Muscle invasion | Weeks 2–4 | Myalgia, muscle stiffness/edema, weakness — especially extraocular muscles, jaw, diaphragm, biceps |
| Convalescence | Weeks 4–8+ | Gradually subsiding symptoms; calcification of larvae over months |
Severe/rare: myocarditis (arrhythmia, heart failure), encephalitis, pneumonitis — accounts for most fatalities.
Light infections are commonly asymptomatic.
5. Diagnostic Imaging and Serology
Echinococcosis — Imaging
Ultrasound (sensitivity 90–95% for hepatic cysts): First-line. The WHO/IWGE classification guides management:
| WHO Stage | Description | Clinical Status |
|---|---|---|
| CE1 | Unilocular, anechoic + "hydatid sand" on movement | Active |
| CE2 | Multivesicular, "honeycomb" / daughter cysts | Active |
| CE3a | Detached membranes — "water lily sign" | Transitional |
| CE3b | Daughter cysts in solid matrix | Transitional |
| CE4 | Heterogeneous, no daughter cysts | Inactive |
| CE5 | Calcified wall | Inactive |
CT: Demonstrates daughter cysts within the mother cyst, calcification, intraperitoneal rupture, biliary communication. Superior to US for extra-hepatic sites.
MRI: Best delineation of cyst wall layers (pericyst, laminated membrane). T1: cyst fluid isointense to CSF. In active cysts: thin rim of enhancement + surrounding edema. Late: calcification (indicates dead cyst).
Brain hydatid (Grainger & Allison): Large, isolated, unilocular, well-defined thin-walled cysts on CT/MRI. Multiple peripheral daughter cysts within a large mother cyst = pathognomonic. Cyst fluid appears similar to CSF. Active cysts show thin rim of enhancement and surrounding edema on MRI.
Pulmonary hydatid imaging signs:
- Unruptured: homogeneous, spherical/oval, sharply demarcated lesion with mass effect.
- "Water lily sign" (Camalote sign): ruptured membranes floating on cyst fluid — detached inner membrane.
- "Rising sun sign" / "Serpent sign": crumpled membranes collapsed at cyst bottom.
- "Empty cyst sign": all contents expectorated.
- Rupture into pleural space → hydropneumothorax.
- Cyst rupture resembles the air crescent of a mycetoma.
Alveolar echinococcosis imaging: Heterogeneous, nodular, cauliflower-like mass with numerous irregular small cysts. Central necrosis with calcification. MRI/CT may mimic hepatocellular carcinoma, cholangiocarcinoma, or liver metastases.
Echinococcosis — Serology
| Method | Role |
|---|---|
| ELISA | First-line screening; most sensitive (60–90%) |
| Indirect hemagglutination (IHA), IFA | Initial screening alternatives |
| Immunoblot (Western blot) | Confirmation; resolves cross-reactivity with T. solium cysticercosis (false positives in up to 25% of NCC) |
- Sensitivity varies by location: liver cysts → most likely to be seropositive; spleen, lung, brain cysts → often seronegative.
- Bone cysts: higher seropositivity.
- False-positives: neurocysticercosis, other cestode infections.
- Definitive diagnosis: identification of protoscoleces and free hooklets in aspirated/excised material.
Trichinellosis — Diagnosis
Laboratory:
- Eosinophilia >90% of symptomatic patients; peaks at >50% at 2–4 weeks.
- Elevated muscle enzymes (CPK, LDH) — present in most symptomatic patients.
- Specific antibody rise (IgG, IgM, IgE by EIA/immunoblot): usually detectable only after week 3. Repeat testing if initially negative. Cross-reactivity with other nematodes — confirm with immunoblot.
Muscle biopsy (definitive):
- At least 1 g from a symptomatic muscle, near the tendon insertion (deltoid, gastrocnemius, biceps).
- Squash preparation between glass slides: coiled larvae visible — superior to routine histologic sections (larvae may be missed on sections alone).
- H&E sections: larvae seen as cross- and longitudinal sections within the nurse cell capsule.

Presumptive clinical diagnosis: fever + eosinophilia + periorbital edema + myalgia + consumption of suspect undercooked meat (especially if others from the same meal are also ill).
6. Differential Diagnosis with Tumors and Abscesses
Hepatic Hydatid Cyst
| Differential | Key distinguishing features |
|---|---|
| Pyogenic liver abscess | Fever, leukocytosis (not eosinophilia), no daughter cysts; thick irregular wall with debris; blood cultures often positive |
| Amoebic liver abscess | RUQ pain, fever, exposure history, raised serum amoebal serology; "anchovy paste" pus; responds to metronidazole |
| Simple hepatic cyst | Thin wall, no septae, no daughter cysts, no hydatid sand, seronegative |
| Polycystic liver disease | Multiple cysts in liver and kidneys; no serologic evidence of Echinococcus |
| Hepatocellular carcinoma | Solid/heterogeneous mass, arterial enhancement on CT, AFP elevated, cirrhosis background; no daughter cysts |
| Cholangiocarcinoma | Biliary dilatation, ductal origin, CA 19-9 elevated, no cyst architecture |
| Cystadenoma/cystadenocarcinoma | Internal septae + solid papillary nodules; no protoscoleces on aspiration |
| Alveolar echinococcosis | Mimics primary hepatic malignancy most closely (irregular infiltrating mass, central necrosis, nodular calcification); distinguished by serology (E. multilocularis-specific Ag), PET-CT for viability |
Pulmonary Hydatid Cyst
| Differential | Key distinguishing features |
|---|---|
| Pulmonary malignancy | Irregular spiculated margins, no clear daughter cysts, no characteristic signs on rupture |
| Lung abscess | Thick-walled cavity, air-fluid level, fever/leukocytosis, no "water lily sign" |
| Aspergilloma (mycetoma) | Air crescent sign in upper lobe, immunocompromised host, Aspergillus serology/culture, no daughter cysts |
| Bronchogenic cyst | Mediastinal or parenchymal; no daughter cysts; no hydatid serology |
| Tuberculoma | Calcified nodule, upper lobe predominance, TB contact history, Mantoux positive |
Trichinellosis
| Differential | Key features of trichinellosis |
|---|---|
| Polymyositis / dermatomyositis | Persistent; no eosinophilia; no dietary exposure; no periorbital edema in the same context; T. pseudospiralis can closely mimic this |
| Viral myositis | No eosinophilia; acute, self-limiting; no nurse cells on biopsy |
| Other parasitic myositis (cysticercosis, toxocariasis) | Different exposure history; cysts on imaging in cysticercosis |
| Systemic vasculitis | Splinter hemorrhages, eosinophilia may overlap; no dietary exposure; muscle biopsy negative for larvae |
| Acute GI infection | Intestinal phase mimics gastroenteritis; eosinophilia and myalgia appearing later confirm trichinellosis |
7. Antiparasitic Treatment Principles
Echinococcosis
Benzimidazoles (the pharmacological mainstay):
| Drug | Mechanism | Notes |
|---|---|---|
| Albendazole | Binds parasite β-tubulin → inhibits microtubule polymerization → impairs glucose uptake and larval cell division | Preferred; better absorbed; dose 10–15 mg/kg/day in cycles |
| Mebendazole | Same mechanism | Less well absorbed; requires longer treatment (≥3 months post-surgery); alternative |
Key principles:
- For PAIR or surgery: albendazole started 4 days pre-procedure and continued ≥4 weeks post-operatively.
- For E. multilocularis (alveolar): albendazole 10–15 mg/kg/day indefinitely if resection is incomplete; apparently curative surgery should be followed by 2-year course to reduce relapse risk.
- Drugs alone (medical treatment) are indicated when surgery is not feasible.
- Benzimidazoles are cytostatic, not reliably cysticidal — they suppress growth but do not reliably kill established cysts in humans. Combination with PAIR increases efficacy.
Trichinellosis
| Severity | Treatment |
|---|---|
| Mild | Supportive (bed rest, antipyretics, analgesics) |
| Moderate | Albendazole 400 mg BID × 10–14 days or Mebendazole 200–400 mg TID × 3 days, then 400–500 mg TID × 10 days |
| Severe (myocarditis, encephalitis, severe myositis) | Add glucocorticoids (e.g., prednisone 40–60 mg/day) |
Key principles:
- Benzimidazoles are active against intestinal adult worms → prevent further larval dissemination. Most effective if given early (before full encystment).
- Efficacy against encysted/encysting larvae in muscle is not conclusively demonstrated — glucocorticoids reduce the inflammatory component but do not kill larvae.
- Most lightly infected patients recover uneventfully without antiparasitic drugs.
8. Surgical Indications
Echinococcosis — Surgery
Cystic Echinococcosis (E. granulosus):
| Indication | Preferred Approach |
|---|---|
| Large cysts (>10 cm) or complicated cysts (rupture, biliary fistula, compression of vital structures) | Surgical resection (open or laparoscopic) |
| Infected cysts / secondary bacterial superinfection | Surgical drainage |
| Inaccessible to PAIR (deep bone, brain, heart) | Open surgery |
| Simple, accessible cysts without biliary communication | PAIR (Percutaneous Aspiration, Injection of scolicidal agent, Re-aspiration) as first-line with concurrent albendazole |
Surgical principles:
- Aim: removal of cyst intact without spillage of contents (prevents anaphylaxis and peritoneal seeding).
- Aspiration of cyst contents prior to resection reduces spillage risk.
- After aspiration: injection of ethyl alcohol or 20% hypertonic saline to kill residual protoscoleces (scolicidal agent).
- Exception: if aspirate is bilious (confirms biliary communication), do not inject alcohol → risk of sclerosing cholangitis → resect without scolicidal injection.
- Approaches: pericystectomy (removal of pericyst intact), partial hepatectomy, or total hepatectomy with transplant (rare, in advanced AE).
Alveolar Echinococcosis (E. multilocularis):
- Surgery is the mainstay of treatment.
- Curative goal: radical resection of all infected tissue (analogous to oncologic resection).
- When radical resection requires it: liver transplantation is an option.
- Inoperable disease: prolonged albendazole indefinitely suppresses lesion growth; mortality approaches that of the age-matched general population with benzimidazole treatment.
Trichinellosis — Surgery
There is no surgical role in trichinellosis per se. Treatment is entirely medical (anthelminthics + corticosteroids). However:
- Muscle biopsy is a diagnostic procedure (≥1 g of involved muscle, near tendon insertion).
- Surgical removal might be relevant only if a rare abscess or localized complication develops (uncommon).
9. Preventive Strategies
Echinococcosis — Prevention
Human-focused:
- Thorough handwashing after contact with dogs or soil in endemic areas.
- Avoid close contact with stray dogs; do not allow dogs to lick the face.
- Wash all fruit and vegetables thoroughly, especially if grown in areas accessed by dogs.
- Safe drinking water.
- Public health education in endemic communities.
Veterinary/animal reservoir control (the most impactful interventions):
- Regular deworming of dogs with praziquantel (kills adult Echinococcus in the canine intestine) — cornerstone of control programs.
- Meat inspection: condemn and safely dispose of infected livestock viscera; do not feed raw offal to dogs.
- Vaccination of sheep with the EG95 recombinant vaccine (against E. granulosus) — reduces cyst development in intermediate hosts, breaks the cycle.
- Controlled slaughter practices.
Environmental:
- Fox population management in E. multilocularis endemic areas.
- Oral baiting of foxes with praziquantel-laced baits (used in Europe).
Trichinellosis — Prevention
Consumer level:
- Cook pork and wild game thoroughly until no longer pink (internal temperature ≥71°C / 160°F) — kills larvae reliably.
- Freezing: -15°C for ≥3 weeks kills most Trichinella species in pork.
- Critical exception: Arctic species (T. nativa) in walrus or bear meat are freeze-resistant and may survive conventional freezing — thorough cooking is mandatory.
Food industry/regulatory:
- Meat inspection and testing of pork products.
- Education of pork producers about pig-raising hygiene (preventing access to rodents, condemned carcasses).
- Prohibition of feeding raw garbage/offal to pigs.
- Irradiation of pork as a supplementary measure (highly effective but not universally applied).
Surveillance:
- Outbreak investigation and tracing of contaminated meat lots.
- Approximately 10–20 cases/year in the USA now vs. 400–500/year in the 1940s — reflecting the success of control programs in domestic pork.
- Wild game consumption (bear, walrus) now accounts for the majority of remaining North American cases.
Summary Comparison
| Feature | Echinococcosis | Trichinellosis |
|---|---|---|
| Pathogen | Cestode larva (Echinococcus spp.) | Nematode (Trichinella spp.) |
| Route | Ingestion of eggs (feco-oral from dogs) | Ingestion of encysted larvae in meat |
| Key reservoir | Dogs (definitive) / sheep (intermediate) | Pigs, wild carnivores |
| Primary target organ | Liver (>60%), lungs, brain | Striated muscle (all groups) |
| Hallmark pathology | Unilocular hydatid cyst / alveolar infiltration | Nurse cell in myocyte |
| Key symptom | Asymptomatic mass → mass effects, anaphylaxis on rupture | Periorbital edema + myalgia + fever + eosinophilia |
| Imaging hallmark | Daughter cysts within mother cyst; water lily sign | CT/MRI: muscle calcifications (chronic) |
| Serology | ELISA (60–90%); immunoblot confirmation | EIA (IgG most sensitive); detectable from week 3 |
| Definitive lab Dx | Protoscoleces + hooklets ("hydatid sand") | Coiled larvae on muscle biopsy (squash prep) |
| Drug treatment | Albendazole (± PAIR / surgery) | Albendazole or mebendazole (+ steroids if severe) |
| Surgery | Yes — for large/complicated cysts; radical resection for AE | No — diagnostic biopsy only |
| Key prevention | Dog deworming, meat inspection, EG95 vaccine for sheep | Thorough cooking of pork/game; pig-raising hygiene |
Sources: Tietz Textbook of Laboratory Medicine, 7th ed.; Harrison's Principles of Internal Medicine, 22nd ed. (2025); Mulholland and Greenfield's Surgery, 7th ed.; Grainger & Allison's Diagnostic Radiology; Goldman-Cecil Medicine, International Edition.
This is a shared conversation. Sign in to Orris to start your own chat.