Extubation criteria
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Here is a comprehensive summary of extubation criteria from authoritative sources:
Extubation Criteria
1. Prerequisites: Readiness to Wean
Before attempting extubation, the underlying disease process should be resolving and ventilator support should be minimal. (The Washington Manual of Medical Therapeutics)
| Parameter | Target |
|---|---|
| FiO₂ | ≤ 40% |
| PEEP | 5 cm H₂O to maintain SpO₂ > 90% |
| pH / PaCO₂ | At patient's baseline |
| Minute ventilation | < 10 L/min |
| Respiratory rate | < 30 breaths/min |
| Spontaneous tidal volume | > 5 mL/kg IBW |
| RSBI (f/VT) | ≤ 105 |
RSBI > 105 reliably predicts weaning failure; RSBI ≤ 105 is a necessary but not sufficient predictor of success.
2. Spontaneous Breathing Trial (SBT)
A 30–120 minute SBT (on PSV, CPAP, or T-piece) should be performed daily. Failure criteria during an SBT:
- Significant rise in RR
- Oxygen desaturation
- Diaphoresis
- Hemodynamic compromise
- Increased work of breathing
3. Clinical Extubation Readiness Criteria
Mental Status
- Awake, alert, and cooperative
- Able to follow commands (in adults)
- Resolution of intoxication; pain adequately controlled
Airway / Reflexes
- Intact cough and gag reflexes
- Able to protect airway from aspiration
- No excessive airway edema or instability
- Negative Inspiratory Force (NIF) ≥ −30 cmH₂O
Secretions
- Thin and scant
- Suctioning required no more frequently than every 4 hours
Neuromuscular Strength
- Strong cough
- Able to lift head off the bed and hold for > 5 seconds
Hemodynamics
- Stable, adequately resuscitated
4. Cuff Leak Test
Indicated when laryngeal edema is suspected (angioedema, traumatic intubation, prolonged intubation):
- Absent cuff leak → preclude extubation
- Treat with IV corticosteroids for 12–24 hours before attempting extubation
- Dexamethasone given >12 hours before extubation is more effective at preventing post-extubation upper airway obstruction
5. Pediatric-Specific Criteria (Barash Clinical Anesthesia / Miller's Anesthesia)
For children < 7 years old, following commands is often not attainable. Five predictors of successful extubation:
- Conjugate gaze
- Facial grimace
- Eye opening
- Purposeful movement
- Tidal volume > 5 mL/kg
Success rate increases from 88.3% (1 predictor) → 100% (all 5 predictors).
Predictors of failure in pediatrics:
- URI within 7 days of procedure
- Midazolam premedication (age > 1 year)
- ETCO₂ > 55 mmHg
6. Post-Extubation Management
- Close monitoring is mandatory after extubation
- In COPD patients extubated from acute respiratory failure: extubation to NPPV reduces mortality
- Extubation failure is defined as reintubation within 24 hours; reported rates: 6–23.5%
- Upper airway obstruction accounts for 37–41% of extubation failures
Sources: Miller's Anesthesia 10e | The Washington Manual of Medical Therapeutics | Barash Clinical Anesthesia 9e
Apache score 2
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
APACHE II Score
Acute Physiology and Chronic Health Evaluation II — the most widely used severity-of-illness (SOI) scoring system in North American ICUs.
Total score = Acute Physiology Score (APS) + Age Points + Chronic Health Points
Maximum possible = 71 points. Uses the worst values in the first 24 hours of ICU admission.
Part 1 — Acute Physiology Score (12 Variables)
| Variable | +4 | +3 | +2 | +1 | 0 | +1 | +2 | +3 | +4 |
|---|---|---|---|---|---|---|---|---|---|
| Rectal temp (°C) | ≥41 | 39–40.9 | — | 38.5–38.9 | 36–38.4 | 34–35.9 | 32–33.9 | 30–31.9 | ≤29.9 |
| Mean BP (mmHg) | ≥160 | 130–159 | 110–129 | — | 70–109 | — | 50–69 | — | ≤49 |
| Heart rate (bpm) | ≥180 | 140–179 | 110–139 | — | 70–109 | — | 55–69 | 40–54 | ≤39 |
| Respiratory rate (breaths/min) | ≥50 | 35–49 | — | 25–34 | 12–24 | 10–11 | 6–9 | — | ≤5 |
| Oxygenation | |||||||||
| — If FiO₂ ≥ 0.5: A-aDO₂ | ≥500 | 350–499 | 200–349 | — | <200 | ||||
| — If FiO₂ < 0.5: PaO₂ (mmHg) | >70 | 61–70 | — | 55–60 | <55 | ||||
| Arterial pH | ≥7.7 | 7.6–7.69 | — | 7.5–7.59 | 7.33–7.49 | — | 7.25–7.32 | 7.15–7.24 | <7.15 |
| Serum Na⁺ (mEq/L) | ≥180 | 160–179 | 155–159 | 150–154 | 130–149 | — | 120–129 | 111–119 | ≤110 |
| Serum K⁺ (mEq/L) | ≥7 | 6–6.9 | — | 5.5–5.9 | 3.5–5.4 | 3–3.4 | 2.5–2.9 | — | <2.5 |
| Serum creatinine (mg/dL) | ≥3.5 | 2–3.4 | 1.5–1.9 | — | 0.6–1.4 | — | <0.6 | — | — |
| (Double score if acute renal failure) | |||||||||
| Hematocrit (%) | ≥60 | — | 50–59.9 | 46–49.9 | 30–45.9 | — | 20–29.9 | — | <20 |
| WBC (×10³/μL) | ≥40 | — | 20–39.9 | 15–19.9 | 3–14.9 | — | 1–2.9 | — | <1 |
| GCS contribution | 15 − actual GCS score = points assigned |
The GCS contribution is: 15 minus the patient's actual GCS (so GCS 15 = 0 points; GCS 3 = 12 points)
Part 2 — Age Points
| Age (years) | Points |
|---|---|
| < 44 | 0 |
| 45–54 | 2 |
| 55–64 | 3 |
| 65–74 | 5 |
| ≥ 75 | 6 |
Part 3 — Chronic Health Points
Add 2 points for elective post-op patients, or 5 points for non-operative / emergency post-op patients, if the patient has a history of severe chronic organ insufficiency or is immunocompromised:
- Hepatic: cirrhosis with portal hypertension or encephalopathy
- Cardiovascular: NYHA Class IV (symptoms at rest or minimal activity)
- Pulmonary: chronic hypoxemia, hypercapnia, polycythemia, or ventilator dependence
- Renal: chronic peritoneal or hemodialysis
- Immune: immunocompromised host
Score Interpretation & Predicted Mortality
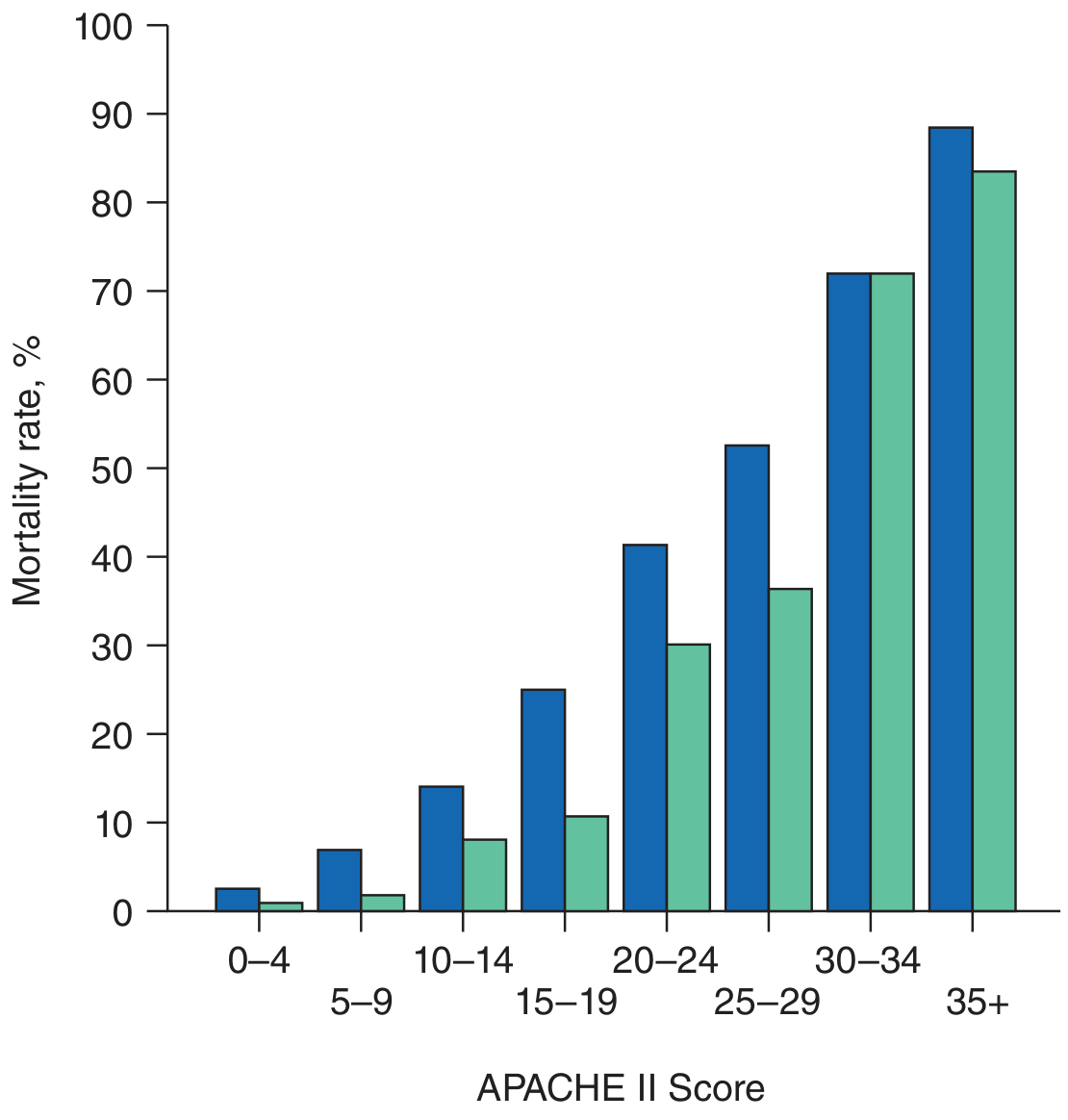
| APACHE II Score | Approximate Mortality (Non-op / Post-op) |
|---|---|
| 0–4 | ~3% / ~1% |
| 5–9 | ~7% / ~2% |
| 10–14 | ~15% / ~8% |
| 15–19 | ~25% / ~11% |
| 20–24 | ~40% / ~30% |
| 25–29 | ~53% / ~37% |
| 30–34 | ~72% / ~72% |
| ≥ 35 | ~89% / ~84% |
Postoperative patients have consistently lower mortality for the same score compared to non-operative patients.
Key Notes
- The predicted hospital mortality is derived from a formula incorporating: APACHE II score + whether emergency surgery was performed + a disease-specific diagnostic category weighting
- For acute renal failure, double the creatinine point score
- Updated versions (APACHE III, IV) exist but APACHE II remains the most widely used in clinical practice
- In acute pancreatitis, a score ≥ 8 is used to define severe disease
- In acute liver failure (e.g., acetaminophen toxicity), APACHE II > 20 is a criterion for liver transplant listing
— Harrison's Principles of Internal Medicine, 22e
CRYSTALLOIDS AND COLLOIDS. DESCRIBE PENTASTARCH
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have enough content from the textbooks to give a comprehensive answer. Let me compile it.
Crystalloids and Colloids
CRYSTALLOIDS
Crystalloid solutions contain small ionic or molecular solutes that freely cross capillary membranes, distributing throughout the entire extracellular fluid (ECF) compartment. Only ~20% of infused normal saline remains intravascular.
Commonly Used Crystalloids
| Solution | Na⁺ (mEq/L) | Cl⁻ (mEq/L) | Other | Tonicity |
|---|---|---|---|---|
| Normal saline (0.9% NaCl) | 154 | 154 | — | Isotonic |
| Lactated Ringer's (LR) | 130 | 109 | Lactate, K⁺, Ca²⁺ | Isotonic |
| Plasmalyte | 140 | 98 | Acetate, gluconate | Isotonic |
| 0.45% NaCl (½ NS) | 77 | 77 | — | Hypotonic |
| 3% NaCl | 513 | 513 | — | Hypertonic |
| 5% Dextrose in water (D5W) | — | — | Glucose | Isotonic in bag, hypotonic in vivo |
Key Points
- Lactated Ringer's is the preferred resuscitation crystalloid — isotonic, repletes the interstitial compartment, does not aggravate electrolyte abnormalities. Lactate is metabolized to bicarbonate in the liver; mild metabolic alkalosis may occur after large-volume infusion.
- Normal saline risks hyperchloremic metabolic acidosis with massive volumes (due to excess Cl⁻).
- Crystalloids are the first-line fluid in hemorrhagic shock — balanced salt solutions decrease transfusion requirements in mild-to-moderate hemorrhage.
- Crystalloid overresuscitation is associated with increased risk of ARDS, MODS, elevated ICP, and abdominal compartment syndrome.
COLLOIDS
Colloids contain large molecules (proteins, starches, dextrans) with low capillary permeability. They generate oncotic pressure, preferentially expanding plasma volume (PV). Approximately 70% of infused 5% albumin stays intravascular vs. only ~20% of normal saline.
Despite theoretical advantages, exhaustive research has failed to demonstrate the superiority of colloids over crystalloids in terms of patient outcomes. — Barash Clinical Anesthesia 9e
Types of Colloids
1. Albumin
- Human-derived protein; available as 4–5% (isooncotic) or 20–25% (hyperoncotic)
- Reflection coefficient (σ) for albumin: 0.6–0.9
- Theoretical benefit: protects lung from interstitial edema
- Drawback: rapid flux across pulmonary capillaries; prolongs resuscitation phase; delays post-resuscitation diuresis; may suppress albumin synthesis and depress immunoglobulin levels
2. Dextrans (Polysaccharides)
| Agent | Concentration | MW | Notes |
|---|---|---|---|
| Dextran 40 | 10% | 40 kDa | Hyperoncotic; exerts large initial volume effect; rapidly excreted; used in peripheral vascular disease, hyperviscosity syndromes |
| Dextran 70 | 6% | 70 kDa | Not hyperoncotic; volume expansion > volume infused; lasts up to 48 hours |
- Side effects: decreased platelet adhesiveness, reduced factor VIII activity
- Allergic reactions: up to 5%; anaphylaxis: 0.6%
3. Hydroxyethyl Starch (HES) — Hetastarch / Pentastarch
Hetastarch (HES 450/0.7)
- Amylopectin derivative
- Volume expansion lasts ~36 hours
- Anaphylaxis rate: 0.006% (much lower than dextrans)
- Side effects similar to dextrans but less frequent
PENTASTARCH — Detailed Description
Pentastarch (HES 200/0.5 or HES 264/0.45) is a second-generation, low-molecular-weight hydroxyethyl starch:
| Feature | Pentastarch | Hetastarch |
|---|---|---|
| Molecular weight | ~200–264 kDa | ~450 kDa |
| Molar substitution | 0.45–0.5 | 0.7 |
| Duration of action | ~2.5 hours | ~36 hours |
| Hydroxyethyl groups | Fewer | More |
| Anaphylaxis risk | Very low | Very low |
- "Penta-" refers to the pattern of hydroxyethylation (at the C2, C3, and C6 positions of glucose, with predominance at C2 over C6 — the C2:C6 ratio influences degradation speed)
- Fewer hydroxyethyl groups → less resistance to plasma amylase → faster degradation and shorter action
- Reported to have even fewer side effects than hetastarch
- Used as a plasma volume expander in hypovolemia and surgical resuscitation
Why Pentastarch Fell Out of Favor
The VISEP trial (NEJM 2008) — a landmark RCT of pentastarch 10% (250 kDa/0.45) in severe sepsis — demonstrated that pentastarch was an independent risk factor for acute kidney injury (AKI) and was associated with increased need for renal replacement therapy. This, combined with similar findings with other HES preparations (e.g., the 6S and CHEST trials for HES 130/0.4), led to a dramatic fall from favor of all starch-based colloids in critically ill patients, particularly those with sepsis.
"The use of starch-based colloids (i.e., pentastarch) has fallen out of favor because a trial demonstrated that they increased the incidence of renal failure in patients with severe sepsis." — Murray & Nadel's Textbook of Respiratory Medicine
Crystalloid vs. Colloid: Summary
| Feature | Crystalloid | Colloid |
|---|---|---|
| Intravascular retention | ~20% | ~70% (albumin) |
| Volume needed | Larger (3:1 rule) | Smaller |
| Pulmonary edema risk | Higher (↓ oncotic pressure) | Lower (theoretically) |
| Cost | Low | High |
| Coagulopathy | Less | More (HES, dextrans) |
| AKI risk | Low | Increased (HES) |
| Evidence of superiority | Neither proven superior overall | Neither proven superior overall |
Bottom line: Balanced crystalloids (LR, Plasmalyte) are first-line for resuscitation. Colloids offer no proven mortality benefit and carry significant risks — particularly HES in critically ill/septic patients.
— Barash Clinical Anesthesia 9e | Mulholland & Greenfield's Surgery 7e | Murray & Nadel's Respiratory Medicine
This is a shared conversation. Sign in to Orris to start your own chat.