Everything about breast abscess in detail with reference to standard textbooks
Reading File
Finding Sources
Finding Sources
Searching Images
breast abscess clinical presentation
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Finding Sources
Reading File
Reading File
I now have comprehensive information from multiple standard textbooks. Let me compile the detailed response:
Breast Abscess — A Comprehensive Review
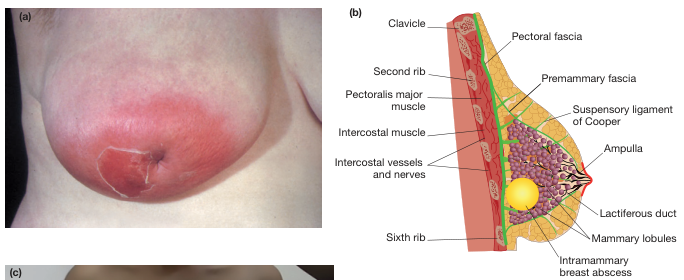
1. Definition
A breast abscess is a localized collection of pus within breast tissue, representing the end-stage of untreated or inadequately treated mastitis. It exists along a clinical continuum: cellulitis → mastitis → abscess formation.
— Tintinalli's Emergency Medicine, p. 3021; Roberts and Hedges' Clinical Procedures in Emergency Medicine, p. 3760
2. Classification
A. Lactational (Puerperal)
- Occurs in breastfeeding women, accounting for ~14% of breast abscesses
- Most common in the first 6 weeks postpartum or during weaning
- Entry of bacteria through cracked or abraded nipple skin
B. Non-Lactational (Non-Puerperal)
- More common — accounts for ~86% of all breast abscesses
- Includes:
- Peripheral abscess: occurs in breast parenchyma, not near the nipple
- Subareolar (periductal) abscess: centred on the areola/lactiferous ducts, strongly associated with smoking
- Complicating breast implants or nipple piercing
— Roberts and Hedges', p. 3760–3764
3. Epidemiology & Risk Factors
| Factor | Comment |
|---|---|
| Lactation | First 6 weeks or weaning phase |
| African American race | Higher incidence |
| Obesity | Non-lactational risk factor |
| Smoking | Strongly linked to periductal/subareolar disease; also a risk factor for recurrence |
| Diabetes mellitus | Risk factor; can lead to severe sepsis requiring hospitalization |
| Nipple piercing | Introduces skin flora |
| Breast implants | Contamination during/after procedure |
| MRSA colonization | Increasingly prominent |
The estimated incidence of mastitis in lactating women ranges from 2% to 33%; breast abscess complicates mastitis in approximately 3% of cases.
— Roberts and Hedges', p. 3762; Tintinalli's, p. 3012
4. Microbiology
Primary Organisms
- Staphylococcus aureus — most common overall (51.3% in one series); includes MRSA (~8.6–20% of cases; prevalence rising)
- Streptococcus pyogenes
Secondary / Recurrent Abscess Organisms
- Escherichia coli
- Bacteroides spp. (anaerobes)
- Corynebacterium spp. (associated with granulomatous mastitis)
- Coagulase-negative staphylococci (e.g., S. lugdunensis)
- Pseudomonas aeruginosa, Proteus mirabilis
- Mixed aerobic-anaerobic flora — more common in recurrent and subareolar abscesses
In lactating women, the source is typically bacteria from the mouth of the nursing infant.
— Roberts and Hedges', p. 3765–3766; Berek & Novak's Gynecology, p. 1776
5. Pathophysiology
- Bacteria enter through a cracked/abraded nipple or areolar skin
- Infection establishes in parenchymal tissue (interlobular connective tissue or periductal area)
- Inflammatory response → cellulitis → loculation of pus
- In non-lactating women with subareolar disease: squamous metaplasia of lactiferous duct epithelium → keratin plugging → duct occlusion → inspissated debris → superinfection → subareolar abscess → possible lactiferous duct fistula (mammillary fistula) to periareolar skin
- Milk stasis in lactating women promotes bacterial overgrowth
— Sabiston Textbook of Surgery, p. 2190–2198; Berek & Novak's, p. 1784
6. Clinical Features
Symptoms
- Localised breast pain, tenderness, swelling
- Fever and chills
- Malaise, rigors (in severe cases — toxic appearance)
Signs
- Erythema, warmth, induration over the affected area
- Fluctuance — pathognomonic of abscess (may be absent in deep abscesses)
- Tender palpable mass
- Nipple retraction or discharge (in subareolar/periductal disease)
- Peau d'orange or skin thinning (late — impending rupture)
- Leukocytosis
Key clinical point: In its early stages when cellulitis predominates, an abscess may be clinically indistinguishable; a trial of antibiotics may be appropriate, with ultrasound evaluation for equivocal cases. Women with significant breast abscess "can be quite ill and appear toxic."
— Roberts and Hedges', p. 3760
7. Investigations
1. Ultrasound (USS) — Investigation of Choice
- Distinguishes cellulitis (diffuse thickened hyperechoic skin/subcutaneous tissue, no cavity) from a true abscess (fluid collection)
- Characterises the abscess: size, unilocular vs. multiloculated, depth, wall thickness
- Guides needle aspiration or catheter drainage
- USS appearance: inhomogeneous, hypoechoic mass (sometimes described as hyperechoic), often with posterior acoustic enhancement
- Essential for documenting sonographic resolution after aspiration
2. Mammography
- Recommended in women >30 years after the acute phase resolves
- Rules out underlying carcinoma (inflammatory breast cancer can mimic abscess)
- Follow-up at 4–6 weeks post-treatment
3. Microbiological Culture
- Pus/aspirate — send for culture and sensitivity (aerobic + anaerobic)
- Guides antibiotic de-escalation/adjustment
- MRSA screening increasingly relevant
4. Biopsy
- Core needle biopsy if an abscess fails to resolve, or if inflammatory carcinoma is suspected
- Patients should NOT undergo prolonged antibiotic treatment without biopsy if erythema persists, to exclude inflammatory carcinoma
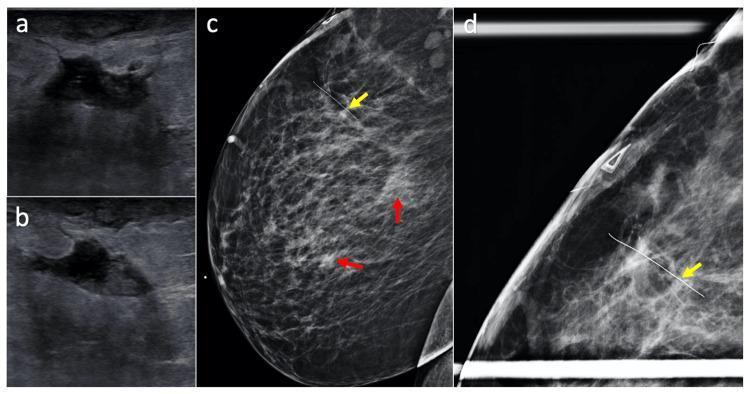
— Roberts and Hedges', p. 3770; Tintinalli's, p. 3023; Berek & Novak's, p. 1779
8. Management
A. Antibiotic Therapy
| Scenario | First-line | Alternatives |
|---|---|---|
| Non-severe, no MRSA risk | Dicloxacillin or cephalexin (oral) | Amoxicillin-clavulanate |
| High community MRSA prevalence | TMP-SMX or clindamycin | Doxycycline, tetracycline |
| β-lactam hypersensitivity | Clindamycin | — |
| Severe/systemic infection | IV vancomycin (empiric) | Linezolid |
| Deep abscess (anaerobic coverage needed) | Add metronidazole | Clindamycin (anaerobic cover) |
| Third-generation cephalosporins | Ceftazidime (parenteral) | Fluoroquinolones |
- Infections should respond within 48 hours; failure warrants imaging to exclude undrained collection or carcinoma
- Duration: typically 7–10 days for lactational mastitis/early abscess
- In lactating women: continue breastfeeding from the unaffected breast; continue emptying the affected breast with a pump
— Roberts and Hedges', p. 3768; Tintinalli's, p. 3023–3027; Sabiston, p. 2190–2198
B. Drainage — The Definitive Treatment
True abscesses require drainage — antibiotics alone are insufficient once pus has formed. — Sabiston, p. 2190
1. Ultrasound-Guided Needle Aspiration (First-line)
- Now the standard of care for most breast abscesses
- Advantages over surgical I&D: less scarring, does not interfere with breastfeeding, no general anaesthesia required
- May require repeat aspirations (mean 3.5 aspirations required for complete resolution)
- Aspirate daily or every other day until resolved
- Document sonographic resolution after each aspiration
Threshold-based approach:
- Abscess < 3 cm: ultrasound-guided needle aspiration
- Abscess ≥ 3 cm: ultrasound-guided catheter drainage (e.g., pigtail catheter)
2. Surgical Incision and Drainage (I&D) — Reserved for:
- Failure of needle aspiration and antibiotics
- Skin compromise overlying the abscess (threatened rupture)
- Complex multiloculated abscesses
- Recurrent abscesses
- Very deep/large collections (may require general anaesthesia)
3. Subareolar Abscess — Specific Considerations
- Percutaneous aspiration + antibiotics initially
- Recurrent subareolar abscesses → excision of involved lactiferous duct(s) at the nipple base during a quiescent (infection-free) interval
- Excision of the entire subareolar duct complex for repeated infections (with IV antibiotic cover)
- Rarely, nipple and areola excision for patients with persistent recurrence
— Roberts and Hedges', p. 3769–3773; Berek & Novak's, p. 1779–1792; Current Surgical Therapy, p. 879–883
C. Lactational-Specific Management
- Continue breastfeeding — do NOT wean; milk stasis worsens the infection
- Nurse from the non-infected breast; regularly empty the infected breast with a pump
- Apply warm compresses to aid drainage
- NSAIDs for pain and anti-inflammatory effect
- Dicloxacillin 250 mg QID or oxacillin 500 mg QID for 7–10 days
— Berek & Novak's, p. 1776
9. Complications
| Complication | Notes |
|---|---|
| Recurrence | Common after I&D alone; lower with US-guided aspiration |
| Lactiferous duct fistula (mammillary fistula) | Subareolar → periareolar skin tract; requires duct excision |
| Nipple retraction/inversion | Scarring from periductal disease |
| Skin necrosis | From pressure of untreated abscess |
| Systemic sepsis | Particularly in diabetics |
| Delayed diagnosis of malignancy | Inflammatory cancer mimics abscess |
| Difficulty breastfeeding | Post-surgical duct damage |
Although a breast abscess is rarely a harbinger of malignancy, it could be the initial manifestation of a metastatic process. Send aspirated pus for cytology if indicated. — Roberts and Hedges', p. 3771
10. Special Situations
Subareolar Abscess and Lactiferous Duct Fistula
- Strong association with tobacco smoking (squamous metaplasia of duct epithelium → keratin plugging → ductal obstruction)
- May present as a periareolar mass, nipple discharge, or fistula
- Recurrence rate is high with I&D alone
- Definitive treatment: excision of the lactiferous duct + fistula tract — Berek & Novak's, p. 1784–1792; Sabiston, p. 2191–2198
Idiopathic Granulomatous Mastitis (IGM)
- Mimics recurrent breast abscess or carcinoma
- More common in Hispanic, Middle Eastern, and Southeast Asian women
- Biopsy: non-necrotising granulomata (exclude TB, fungal, sarcoidosis)
- Treatment: antibiotics targeting Corynebacterium (doxycycline, clindamycin, azithromycin), systemic steroids, methotrexate; surgery avoided (poor wound healing) — Sabiston, p. 2203
Inflammatory Breast Cancer — Must Exclude
- Presents with breast enlargement, warmth, erythema, oedema, peau d'orange, nipple retraction
- Clinically indistinguishable from infection
- Diagnosis: mammography + skin/breast biopsy
- Must be considered if breast cellulitis or abscess fails to completely resolve with antibiotics — Tintinalli's, p. 3035–3037
11. Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Cellulitis (without abscess) | No fluctuance, no cavity on USS |
| Inflammatory breast cancer | No response to antibiotics; peau d'orange; biopsy needed |
| Periductal mastitis (duct ectasia) | Subareolar; nipple discharge/retraction; non-infective |
| Galactocele | Lactating woman; milk-filled cyst on USS |
| Ruptured breast cyst | Sudden onset; cyst history |
| Granulomatous mastitis | Chronic; biopsy distinguishes |
| Tuberculosis of breast | Rare; cold abscess; Mycobacterium on culture/PCR |
| Hidradenitis suppurativa | Inferior breast fold; apocrine distribution; sinus tracts |
| Metastatic carcinoma |
Summary Algorithm
Breast pain + erythema + swelling
↓
Ultrasound
/ \
Cellulitis Abscess confirmed
↓ ↓
Antibiotics Culture pus + Antibiotics
(cover MRSA +
if indicated) USS-guided needle aspiration
↓ (if <3 cm)
Repeat aspiration q24–48h (mean 3.5)
↓ (if ≥3 cm or fails aspiration)
USS-guided catheter drainage
↓ (fails or multiloculated/skin compromise)
Surgical I&D
↓
Follow-up mammogram >30 years (after resolution)
Subareolar recurrence → Duct excision (quiescent phase)
Key References (Standard Textbooks)
| Textbook | Edition | Key Content |
|---|---|---|
| Sabiston Textbook of Surgery | Current Ed. | Classification, pathophysiology, surgical management |
| Roberts and Hedges' Clinical Procedures in Emergency Medicine | Current Ed. | Incidence, microbiology, US-guided drainage technique, recurrence |
| Tintinalli's Emergency Medicine | Current Ed. | Differential diagnosis, MRSA management, inflammatory cancer |
| Berek & Novak's Gynecology | Current Ed. | Lactational vs. non-lactational, subareolar fistula, granulomatous mastitis |
| Current Surgical Therapy | 14th Ed. | Periductal mastitis, drainage thresholds, fistula management |
| Bailey and Love's Short Practice of Surgery | 28th Ed. | Anatomy, clinical photograph |
This is a shared conversation. Sign in to Orris to start your own chat.