diagnostic criteria for fungal pneumonia
Reading File
search_medical_text
fungal pneumonia diagnostic criteria clinical
search_medical_image
fungal pneumonia chest imaging CT findings
GradeResponse
GradeResponse
Here is a comprehensive overview of the diagnostic criteria for fungal pneumonia:
Fungal Pneumonia: Diagnostic Criteria
Fungal pneumonia encompasses several organisms with overlapping but distinct diagnostic frameworks. The major causative pathogens are Aspergillus spp., Candida spp., Cryptococcus neoformans, Histoplasma capsulatum, Coccidioides immitis, and Pneumocystis jirovecii (PCP).
I. General Diagnostic Framework
Diagnosis requires integration of three domains:
| Domain | Components |
|---|---|
| Host factors | Immunosuppression, neutropenia, SOT/HSCT, HIV, steroids |
| Clinical/radiologic | Symptoms + imaging findings |
| Mycologic evidence | Culture, histopathology, serology, molecular tests |
II. EORTC/MSGERC Diagnostic Categories (Invasive Fungal Disease)
The most widely used framework (European Organisation for Research and Treatment of Cancer / Mycoses Study Group) classifies invasive fungal disease (IFD) into three levels:
Proven IFD
- Histopathologic, cytopathologic, or direct microscopic evidence of fungal elements from a needle aspiration or biopsy specimen with associated tissue damage, OR
- Positive culture from a normally sterile site
Probable IFD
- At least one host factor + one clinical criterion + one mycologic criterion
Possible IFD
- Host factor + clinical criterion, but no mycologic evidence
III. Host Factors (Risk Criteria)
- Prolonged neutropenia (>10 days, ANC <500/μL)
- Allogeneic HSCT recipient
- Prolonged corticosteroid use (>0.3 mg/kg/day prednisone equivalent for >3 weeks)
- Treatment with T-cell immunosuppressants (calcineurin inhibitors, anti-TNF agents)
- Inherited severe immunodeficiency (e.g., chronic granulomatous disease, SCID)
- AIDS (particularly for Cryptococcus, PCP, Histoplasma)
IV. Clinical Criteria (Radiologic)
CT chest findings are the primary clinical criterion:
| Sign | Organism | Description |
|---|---|---|
| Halo sign | Aspergillus | Ground-glass opacity surrounding a nodule (perinodular hemorrhage) |
| Air crescent sign | Aspergillus | Radiolucent crescent separating necrotic tissue from cavity wall (late finding) |
| Tree-in-bud | Aspergillus, others | Bronchiolar impaction; centrilobular nodules |
| Wedge-shaped/pleural-based consolidation | Aspergillus | Angioinvasive infarction |
| Miliary nodules | Histoplasma, Cryptococcus | Diffuse small nodules |
| Diffuse bilateral GGO | PCP | Perihilar ground-glass opacities |
| Consolidation ± cavitation | Coccidioides, Mucor | Lobar or segmental |
The image below illustrates classic CT findings of invasive pulmonary aspergillosis (IPA):
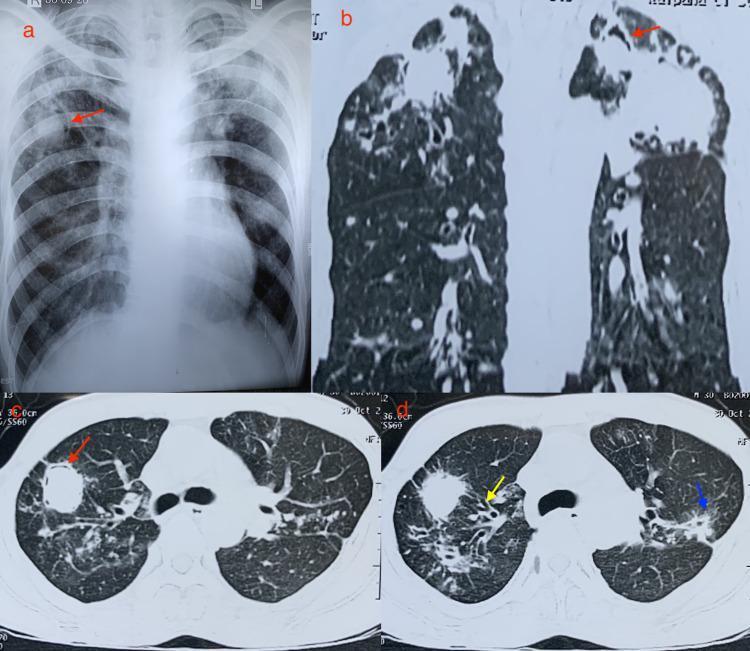
(a) PA chest X-ray and (b–d) CT in sagittal/axial planes. Red arrows = air crescent sign; yellow arrow = tree-in-bud pattern; blue arrow = halo sign.
V. Mycologic Criteria
As noted in the Guide to Utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases (p. 51), the following tests are helpful when radiographic or clinical evidence suggests fungal pneumonia:
Serum/BAL Biomarkers
| Test | Target Organism | Notes |
|---|---|---|
| Galactomannan (GM) | Aspergillus | Serum and BAL; BAL GM has higher sensitivity; cut-off: ODI ≥1.0 (BAL), ≥0.5 (serum) |
| 1,3-β-D-glucan | Most fungi (not Mucor/Cryptococcus) | Pan-fungal marker; frequently over-utilized in non-immunocompromised patients |
| Cryptococcal antigen | Cryptococcus | Serum or CSF; high sensitivity/specificity |
| Histoplasma urine antigen | Histoplasma | Best test for disseminated/acute pulmonary histoplasmosis |
| Coccidioides serology (IgM/IgG) | Coccidioides | Immunodiffusion, complement fixation |
| LDH + β-D-glucan | PCP | LDH elevation supportive; β-D-glucan elevated |
Culture
- Sputum culture: low sensitivity, poor specificity for most fungi
- BAL culture: preferred for most fungal pathogens
- Blood culture: positive in disseminated candidiasis and cryptococcosis
Molecular / PCR
- Aspergillus PCR (serum or BAL): emerging role; ESCMID guidelines support use alongside GM
- PCP PCR (BAL): highly sensitive but interpretation requires clinical context (colonization vs. infection)
Histopathology
- Gold standard for proven IFD
- Cytology and/or histopathology distinguish fungal infection from pulmonary hemorrhage, rejection, or other infiltrates
- Tissue obtained via:
- Transbronchial biopsy (BAL)
- CT-guided transthoracic needle aspiration (pleural-based lesions)
- Open lung biopsy (when less invasive tests are unrevealing)
VI. Organism-Specific Diagnostic Highlights
| Organism | Key Diagnostic Features |
|---|---|
| Aspergillus (IPA) | Halo/air crescent sign on CT; serum/BAL galactomannan; Aspergillus PCR; neutropenic host |
| Candida | Rarely causes primary pneumonia; β-D-glucan; blood culture; usually hematogenous |
| Cryptococcus | Cryptococcal antigen (serum/CSF); India ink of CSF; nodules/mass on CT |
| Histoplasma | Urine/serum antigen; complement fixation serology; endemic exposure history |
| Coccidioides | Serology (IgM/IgG); endemic area; thin-walled cavities on CT |
| PCP | BAL silver stain/immunofluorescence/PCR; elevated LDH + β-D-glucan; HIV or immunosuppressed; bilateral perihilar GGO on CT |
| Mucorales | Rhinosinusitis with black eschar, angioinvasion; CT halo sign (reverse halo/atoll sign); histopathology required; β-D-glucan often negative |
VII. Practical Diagnostic Algorithm
Suspected fungal pneumonia
│
├── Obtain: CT chest + serum biomarkers (GM, β-D-glucan, CrAg per host)
│
├── Immunocompromised?
│ YES → Apply EORTC/MSGERC criteria
│ Probable = Host + CT finding + positive biomarker
│
├── BAL bronchoscopy if CT abnormal
│ → Culture, GM, cytology, PCR
│
└── Biopsy if:
- BAL unrevealing
- Accessible lesion (CT-guided)
- Diagnosis remains uncertain → Open lung biopsy
Key takeaways:
- Diagnosis is almost never based on a single test; it requires host + clinical + mycologic convergence.
- Galactomannan and β-D-glucan are adjuncts, not standalone tests — they are most useful in high-pretest-probability (immunocompromised) patients.
- Histopathology remains the gold standard for proven disease.
- CT imaging, particularly the halo sign (early) and air crescent sign (late), is central to the clinical diagnosis of invasive aspergillosis.
etiologic factor for fungal pneumonia
search_medical_text
etiologic factors causative organisms fungal pneumonia pathogenesis
search_medical_text
risk factors host susceptibility invasive fungal infection immunocompromised
GradeResponse
GradeResponse
search_medical_text
Aspergillus Histoplasma Cryptococcus Coccidioides etiology epidemiology pulmonary infection
search_medical_text
Pneumocystis jirovecii Candida Mucor pulmonary infection etiology pathogenesis
Etiologic Factors of Fungal Pneumonia
Fungal pneumonia arises from an interplay of pathogen-specific factors and host susceptibility. The etiologic framework is best understood across three axes: the causative organism, the route of infection, and the host immune status.
I. Causative Organisms (Etiology by Pathogen)
1. Aspergillus spp. (most common cause of invasive fungal pneumonia)
- Species: A. fumigatus (most common), A. flavus, A. niger, A. terreus
- Habitat: Ubiquitous in soil, decaying organic matter, construction dust, hospital air
- Route: Inhalation of airborne conidia
- Pathogenesis: In immunocompetent hosts, conidia are cleared by alveolar macrophages and neutrophils. In immunocompromised hosts, conidia germinate into hyphae, invade pulmonary vasculature (angioinvasion), causing hemorrhagic infarction and dissemination
- Key risk factors (Harrison's, p. 6263): profound neutropenia, glucocorticoid use, underlying respiratory disease, CGD, advanced HIV, relapsed leukemia; risk increases with longer duration and higher steroid doses
2. Pneumocystis jirovecii (PCP)
- Classification: Previously classified as a protozoan; now recognized as an atypical fungus
- Habitat: Ubiquitous environmental organism; asymptomatic colonization common in humans
- Route: Airborne transmission; reactivation of latent infection
- Pathogenesis: Organism attaches to type I pneumocytes via surface glycoprotein A → alveolar damage → impaired gas exchange → progressive hypoxia
- Key risk factors: HIV/AIDS (CD4 <200 cells/μL), solid organ transplant, hematologic malignancies, prolonged steroids, primary immunodeficiencies
3. Histoplasma capsulatum
- Habitat: Endemic to Ohio and Mississippi River valleys (USA), Central and South America; found in soil enriched with bird or bat droppings (caves, chicken coops)
- Route: Inhalation of microconidia released from disturbed soil
- Pathogenesis: Microconidia convert to yeast form at 37°C in lungs → phagocytosed by macrophages but survive intracellularly → granuloma formation (in immunocompetent) vs. disseminated infection (in immunocompromised)
- Key risk factors: Endemic area exposure, activities like spelunking/construction/farming, HIV (CD4 <150), TNF-α inhibitor use
4. Coccidioides immitis / C. posadasii
- Habitat: Endemic to the arid southwestern USA (San Joaquin Valley), northern Mexico, parts of Central/South America; found in alkaline desert soil
- Route: Inhalation of arthroconidia from disturbed soil (dust storms, construction, agriculture)
- Pathogenesis: Arthroconidia → spherules → endospores released into lung tissue → granulomatous inflammation; in immunocompromised, dissemination to bones, meninges, skin
- Key risk factors: Endemic exposure, African-American or Filipino descent (higher risk of dissemination), pregnancy (3rd trimester), HIV, organ transplant, high-dose steroids
5. Cryptococcus neoformans / C. gattii
- Habitat: C. neoformans — worldwide in pigeon droppings and soil; C. gattii — endemic to tropical/subtropical regions (Pacific Northwest, Australia)
- Route: Inhalation of desiccated yeast or basidiospores
- Pathogenesis: Primary pulmonary infection often subclinical → hematogenous dissemination to CNS (cryptococcal meningitis) in immunocompromised; polysaccharide capsule impairs phagocytosis
- Key risk factors: HIV/AIDS (most common; CD4 <100), solid organ transplant, sarcoidosis, corticosteroid use
6. Mucorales (Mucormycosis)
- Species: Rhizopus, Mucor, Lichtheimia, Cunninghamella
- Habitat: Ubiquitous in soil, decaying vegetation, bread mold
- Route: Inhalation of sporangiospores; occasionally ingestion or direct inoculation
- Pathogenesis: Spores germinate → hyphae invade blood vessel walls → thrombosis → tissue necrosis (angioinvasion); iron availability (e.g., diabetic ketoacidosis, deferoxamine use) is a critical growth factor
- Key risk factors: Diabetic ketoacidosis (DKA), prolonged neutropenia, iron overload/deferoxamine therapy, hematologic malignancy, HSCT, trauma/burns
7. Candida spp.
- Species: C. albicans, C. glabrata, C. tropicalis, C. parapsilosis
- Route: Primarily hematogenous seeding of the lungs (true primary Candida pneumonia from inhalation is rare); aspiration in intubated patients
- Pathogenesis: Part of normal oropharyngeal flora → overgrowth with disruption of mucosal barriers → candidemia → pulmonary seeding
- Key risk factors: Prolonged broad-spectrum antibiotics, central venous catheters, TPN, abdominal surgery, neutropenia, ICU admission
8. Scedosporium / Lomentospora (Rare/Emerging)
- Per Harrison's (p. 6309): Scedosporium apiospermum complex and Lomentospora prolificans are major pathogens in immunocompromised hosts, causing pneumonia, disseminated infection, and brain abscess
- Found in tidal flats, swamps, ponds, manure, and soil in temperate climates
- Route: inhalation of conidia (primary); direct inoculation (secondary)
- Particularly associated with near-drowning and cystic fibrosis
II. Etiologic Factors by Category
A. Pathogen-Related Factors
| Factor | Significance |
|---|---|
| Thermotolerance | Ability to grow at 37°C (e.g., A. fumigatus grows up to 55°C) |
| Virulence factors | Capsule (Cryptococcus), gliotoxin (Aspergillus), melanin (multiple) |
| Spore/conidia size | Small conidia (2–3 μm) penetrate to alveoli (e.g., A. fumigatus) |
| Angioinvasion | Aspergillus, Mucor — vascular invasion drives hemorrhagic necrosis |
| Intracellular survival | Histoplasma, Cryptococcus evade macrophage killing |
| Environmental reservoir | Determines endemic vs. opportunistic patterns |
B. Host-Related Etiologic Factors
| Host Factor | Organisms Favored |
|---|---|
| Neutropenia (ANC <500) | Aspergillus, Candida, Mucor |
| T-cell dysfunction / HIV | PCP, Cryptococcus, Histoplasma, Coccidioides |
| Corticosteroids | Aspergillus, PCP, Mucor |
| HSCT / SOT | Aspergillus, Candida, PCP, Cryptococcus |
| DKA / iron overload | Mucor (iron is essential for growth) |
| Structural lung disease (COPD, bronchiectasis) | Aspergillus (chronic/ABPA forms) |
| CD4 <200 | PCP |
| CD4 <100 | Cryptococcus |
| CD4 <50 | Histoplasma, disseminated |
| TNF-α inhibitors | Histoplasma, Coccidioides, Aspergillus |
C. Environmental / Epidemiologic Factors
| Factor | Relevant Pathogen |
|---|---|
| Endemic area exposure (Ohio/Mississippi valleys) | Histoplasma |
| Arid southwest USA desert exposure | Coccidioides |
| Bird/bat droppings, spelunking, excavation | Histoplasma |
| Hospital construction, renovation, air ducts | Aspergillus |
| Pigeon droppings | Cryptococcus |
| Tropical travel (Pacific Northwest, Australia) | C. gattii |
| Near-drowning, contaminated water | Scedosporium |
| Decaying vegetation, organic compost | Mucorales |
III. Summary: Organism-to-Etiology Map
IMMUNOCOMPROMISED HOST
├── Neutropenic (hematologic malignancy, HSCT) → Aspergillus, Candida, Mucor
├── HIV/AIDS → PCP (CD4<200), Cryptococcus (CD4<100), Histoplasma, Coccidioides
├── SOT + steroids → Aspergillus, PCP, Cryptococcus
└── DKA / iron overload → Mucorales
IMMUNOCOMPETENT HOST (endemic exposure)
├── Ohio/Mississippi Valley → Histoplasma
├── Southwest USA/desert → Coccidioides
└── Worldwide (low inoculum) → subclinical Cryptococcus, Histoplasma
STRUCTURAL LUNG DISEASE
└── COPD, bronchiectasis, TB cavities → Aspergillus (chronic pulmonary aspergillosis, ABPA)
Key principle: Fungal pneumonia is almost always the result of a failure of host defenses against an environmental organism rather than person-to-person transmission. The specific etiology is heavily predicted by the type of immune defect combined with geographic and environmental exposure history.
This is a shared conversation. Sign in to Orris to start your own chat.