Structure of haemoglobin and functions
Reading File
Finding Sources
Finding Sources
Searching Images
haemoglobin structure quaternary alpha beta chains heme
Reading File
Reading File
Reading File
Loading Image
Loading Image
I have rich textbook content and diagrams. Let me now compile a comprehensive answer.
Structure and Functions of Haemoglobin
Overview
Haemoglobin (Hb) is an oxygen-transport protein found exclusively in red blood cells (RBCs). In adults, the predominant form is HbA (α₂β₂), with a molecular weight of approximately 64,500 Da. Its primary role is transporting O₂ from the lungs to peripheral tissues, but it also carries CO₂, H⁺, and modulates its own O₂ affinity through allosteric regulation.
Structure
1. Primary Structure (Amino Acid Sequence)
- HbA consists of four polypeptide (globin) chains: 2 α-chains (141 amino acids each) and 2 β-chains (146 amino acids each).
- Each chain is encoded by a separate gene; mutations in these genes cause haemoglobinopathies (e.g., HbS in sickle cell disease, where Glu→Val at position 6 of the β-chain).
2. Secondary Structure
- Each globin chain contains stretches of α-helical segments (conventionally labeled A through H from the amino terminus).
- These helices fold to create a hydrophobic heme-binding pocket near the surface of each subunit.
3. Tertiary Structure (Individual Subunit)
- Each globin chain folds around a single heme group — a protoporphyrin IX ring containing a central ferrous iron (Fe²⁺) atom.
- The Fe²⁺ forms six coordination bonds:
- Four bonds to the pyrrole nitrogens of the porphyrin ring
- One bond to the imidazole nitrogen of the proximal histidine (His F8 — 87th residue in α-chain, 92nd in β-chain) of the globin chain
- One bond reversibly available for O₂ binding
- A distal histidine (His E7) lies on the opposite side of the heme, stabilising the bound O₂ and preventing Fe²⁺ oxidation to Fe³⁺ (methaemoglobin).
4. Quaternary Structure (Tetramer)
This is the defining structural feature that distinguishes haemoglobin from myoglobin.
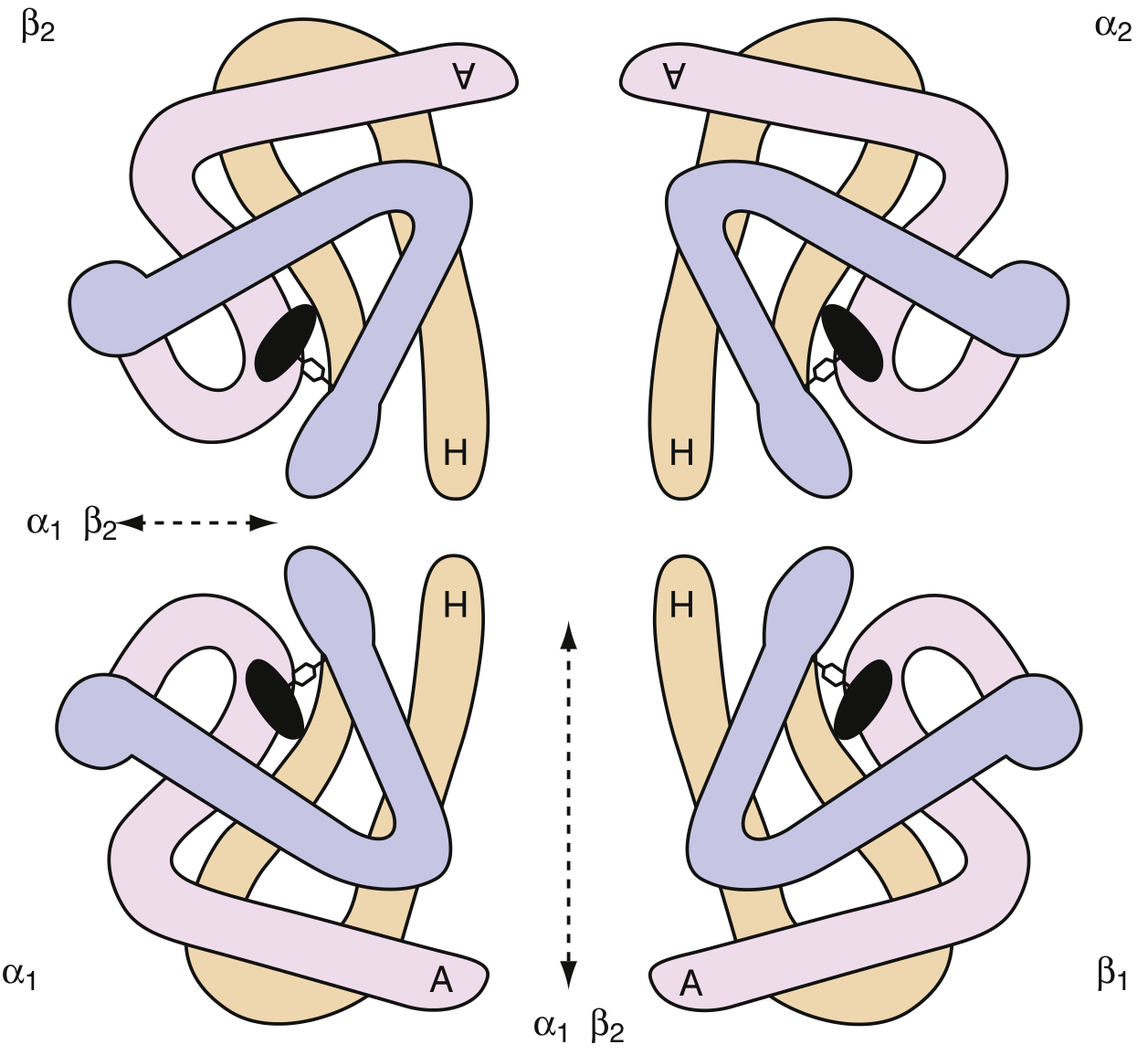
The four subunits are arranged as two αβ dimers (α₁β₁ and α₂β₂):
| Contact type | Location | Bond type | Significance |
|---|---|---|---|
| α₁β₁ (and α₂β₂) | Within each dimer | Strong hydrophobic | Stabilising — largely fixed |
| α₁β₂ (and α₂β₁) | Between dimers | Weaker polar/ionic | Functional — allows movement during O₂ binding |
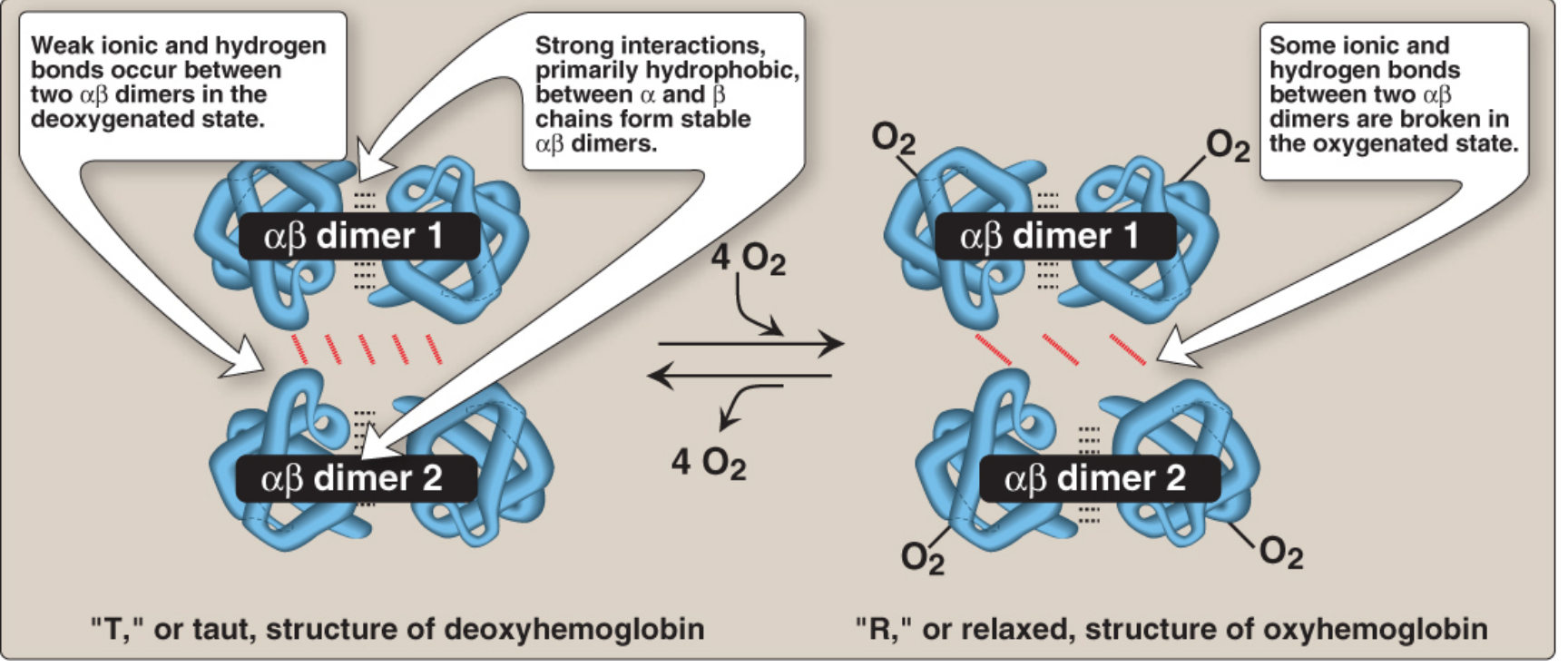
The movement at the α₁β₂ interface is the molecular basis of the T↔R conformational switch:
T form (Taut / Deoxy form)
- The two αβ dimers are constrained by an extensive network of ionic bonds and hydrogen bonds.
- Fe²⁺ is displaced slightly out of the plane of the heme ring.
- Low O₂ affinity — stabilised by H⁺, CO₂, 2,3-BPG, and Cl⁻.
R form (Relaxed / Oxy form)
- Binding of O₂ pulls Fe²⁺ into the plane of the heme; this movement pulls the proximal histidine and the attached helix, breaking some of the inter-dimer polar bonds.
- High O₂ affinity — once one O₂ binds, subsequent binding is facilitated (cooperativity).
Heme Group
- Heme = iron protoporphyrin IX: a porphyrin ring (four pyrrole units linked by methine bridges) with a central Fe²⁺.
- Synthesised from succinyl-CoA + glycine → δ-aminolevulinic acid (ALA) → porphobilinogen → protoporphyrin IX → heme.
- Fe must remain as Fe²⁺ (ferrous) to bind O₂ reversibly. Oxidation to Fe³⁺ produces methaemoglobin, which cannot carry O₂.
Developmental Variants
Different globin chains are expressed at different stages:
| Stage | Haemoglobin | Chains |
|---|---|---|
| Embryonic | HbGower 1, HbGower 2, HbPortland | ζ/ε, α/ε, ζ/γ |
| Fetal | HbF | α₂γ₂ |
| Adult (major) | HbA | α₂β₂ (~97%) |
| Adult (minor) | HbA₂ | α₂δ₂ (~2.5%) |
HbF has a higher O₂ affinity than HbA because its γ-chains bind 2,3-BPG less avidly, facilitating O₂ transfer from maternal to fetal blood.
Functions
1. Oxygen Transport
- Each Hb molecule carries up to 4 O₂ molecules (one per heme).
- O₂ binding is cooperative (sigmoidal dissociation curve), meaning the affinity increases as each successive O₂ binds — the basis of efficient loading in the lungs and unloading in the tissues.
- Without haemoglobin, plasma can carry only ~0.3 mL O₂ per 100 mL blood; with HbA, this rises to ~20 mL/100 mL.
2. Carbon Dioxide Transport
- ~20–23% of CO₂ is carried as carbamino-haemoglobin (CO₂ bound to the N-terminal amino groups of globin chains as carbamate).
- CO₂ binding to the T form stabilises deoxyhaemoglobin and promotes O₂ release (Bohr effect).
3. Buffering (Bohr Effect)
- Hb is one of the most important blood buffers.
- In tissues, rising CO₂ → ↑ H⁺ (via carbonic anhydrase) → H⁺ binds to specific histidine residues on globin chains → T form stabilised → O₂ released.
- In the lungs, CO₂ is expelled → ↓ H⁺ → R form favoured → O₂ loaded.
- This reciprocal relationship between H⁺/CO₂ and O₂ affinity is the Bohr effect.
4. Regulation via 2,3-Bisphosphoglycerate (2,3-BPG)
- 2,3-BPG is a product of glycolysis in RBCs (Rapoport–Luebering shunt).
- It binds in the central cavity between the β-chains of the T form, forming ionic bonds and reducing O₂ affinity (right-shifts the O₂ dissociation curve).
- Elevated at high altitude and in chronic anaemia → enhances O₂ delivery to tissues.
- HbF binds 2,3-BPG poorly → higher O₂ affinity (important for placental gas exchange).
5. Nitric Oxide (NO) Transport
- Haemoglobin can bind NO at the Fe²⁺ of heme (forming nitrosyl-Hb) or at cysteine β-93 (forming S-nitrosyl-Hb), modulating vascular tone.
6. Proton Transport (Haldane Effect)
- Deoxyhaemoglobin has a higher affinity for H⁺ than oxyhaemoglobin; this helps carry H⁺ from tissues to the lungs for excretion as H₂O after combining with HCO₃⁻.
Summary Table
| Property | Detail |
|---|---|
| Molecular weight | ~64,500 Da |
| Subunits | α₂β₂ (adult HbA) |
| Prosthetic group | Heme (Fe²⁺-protoporphyrin IX) |
| O₂ binding sites | 4 per molecule |
| O₂ dissociation curve | Sigmoidal (cooperative) |
| Low-affinity form | T (taut/deoxy) — stabilised by H⁺, CO₂, 2,3-BPG |
| High-affinity form | R (relaxed/oxy) |
| Key allosteric effectors | H⁺, CO₂, 2,3-BPG, Cl⁻, temperature |
Sources: Lippincott's Illustrated Reviews: Biochemistry, 8th ed., pp. 94–96; Henry's Clinical Diagnosis and Management by Laboratory Methods, pp. 660–663; Harrison's Principles of Internal Medicine, 22nd ed., pp. 813–815
Ok please make it in ppt format
Reading File
Reading File
Reading File
Reading File
Running Command
Running Command
Writing File
Running Command
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.