Dressing procedure deep drive mind blowing answer
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
wound dressing types procedure steps
Reading File
Now I have comprehensive material from multiple authoritative textbooks. Let me compile this into a thorough, well-structured answer.
🩹 Wound Dressing Procedure — Deep Dive
1. Historical Background
The use of wound dressings stretches back at least 4,000 years. The Edwin Smith Surgical Papyrus (~1615 BCE) describes linen strips and plaster to dress wounds, noting that closed wounds heal faster. Ancient Egyptians coated linen strips with grease or honey to create semi-occlusive, adherent dressings.
Gauze-based dressings were not standardized until 1871. The paradigm shifted dramatically in 1962 when George Winter demonstrated a 30% faster re-epithelialization rate under occlusive dressings compared to air-dried wounds — the foundation of modern "moist wound healing."
— Dermatology 2-Volume Set 5e
2. Why Moist Wound Healing Works
| Mechanism | Effect |
|---|---|
| Prevents tissue desiccation & crust formation | Epidermal cells migrate laterally without burrowing under dry eschar |
| Maintains growth factors in wound fluid | Promotes cellular proliferation |
| Low tissue oxygen tension at wound surface | Stimulates angiogenesis and collagen deposition |
| Maintains electrical gradient | Further promotes healing |
| Mildly acidic pH | Favors fibroblast proliferation and granulation tissue formation |
| Rate of epithelialization | 2× faster under occlusive vs. uncovered wounds |
A thicker crust forces regenerating epidermis to migrate deeper to find living cells — longer migration = slower healing.
— Sabiston Textbook of Surgery 7e | Dermatology 2-Volume Set 5e
3. Functions of an Ideal Wound Dressing
An ideal dressing must:
- Maintain a moist healing environment
- Absorb excess exudate without causing maceration of surrounding skin
- Provide a bacterial barrier
- Leave no residual debris (avoids foreign body reactions)
- Be non-adherent and pain-free to remove
- Be cost-effective and easy to use
- Adapt to changing wound conditions (exudate, infection, necrosis)
No perfect single dressing exists for all wounds — dressing choice must evolve as the wound heals.
— Dermatology 2-Volume Set 5e
4. Four Categories of Dressings (Sabiston Classification)
| Category | Examples | Best For |
|---|---|---|
| Nonadherent fabrics | Scarlet red, Vaseline gauze, Xeroform, Mepitel, Adaptic, Telfa | Primary contact layer; protection |
| Absorptive gauze | Wide-mesh gauze | Exudate removal; wet-to-dry debridement |
| Foams | Allevyn, Curafoam, Lyofilm | Protection + high exudate absorption |
| Creams, ointments, solutions | Silver sulfadiazine, bacitracin, mupirocin | Antimicrobial coverage |
— Sabiston Textbook of Surgery 7e
5. Moisture-Retentive Dressing Types (The "Big Six")
🔵 A. Film Dressings
- Thin, transparent, self-adhesive polyurethane sheets
- Gas-permeable (O₂/CO₂ exchange) but impermeable to water and bacteria
- Best for: Shallow, minimally exudative wounds, IV site protection
- Limit: Cannot absorb exudate; may cause maceration in high-exudate wounds
🟡 B. Foam Dressings
- Hydrophilic polyurethane sheets — absorb exudate while keeping wound moist
- Comfortable, conforming, can expand to fill wound cavity
- Best for: Moderate-to-high exudate wounds, pressure ulcers, donor sites
- Easily removed for wound inspection/cleansing
🟢 C. Hydrogels
- High water content (>90%) — donate moisture to wound
- Best for: Dry wounds, necrotic wounds, radiation dermatitis, painful wounds (cooling effect)
- Not ideal for high-exudate wounds (will over-hydrate)
🟠 D. Alginates / Gelling Fibers
- Derived from brown seaweed (alginates) or carboxymethyl cellulose (hydrofibers)
- React with wound exudate via ion exchange (Na⁺ in exudate replaces Ca²⁺ in alginate) → form a gel
- Hemostatic effect: Released free calcium activates the clotting cascade
- Gelling fibers are 3× more absorbent than alginates; vertical fluid uptake reduces periwound maceration
- Highly absorbent — can stay in place several days; removed painlessly with saline irrigation
- Best for: High-exudate wounds, post-debridement, bleeding wounds
🔴 E. Hydrocolloids
- Inner layer: hydrophilic colloid base (pectin, karaya, carboxymethyl cellulose + adhesive)
- Outer layer: semipermeable polyurethane
- Form a gel in presence of wound exudate; semipermeable to water vapor and gases
- Best for: Partial-thickness wounds, pressure ulcers (Stage II–III), low-to-moderate exudate
- Classic example: DuoDERM®
🟣 F. Superabsorbent Dressings
- Most recent category; highest fluid-locking capacity
- Prevents fluid back-leak even under compression
- Best for: Highly exudative chronic wounds
— Dermatology 2-Volume Set 5e
6. Dressing Selection by Wound Characteristics
| Wound Factor | Dressing of Choice |
|---|---|
| High exudate | Alginates, hydrofibers, foams, superabsorbents |
| Low/no exudate (dry) | Hydrogels, films |
| Necrotic tissue / eschar | Hydrogels (autolytic debridement), enzymatic agents |
| Infected wound | Antimicrobial (silver, cadexomer-iodine, honey, DACC) |
| Bleeding | Alginates (hemostatic) |
| Malodorous | Activated charcoal cloths, medicated impregnated dressings |
| Partial-thickness / graft | Nonadherent fabrics (Mepitel, Adaptic) |
| Painful wound | Hydrogels, soft silicone dressings |
— Dermatology 2-Volume Set 5e | Textbook of Family Medicine 9e
7. Antimicrobial Dressings
| Agent | Mechanism | Key Points |
|---|---|---|
| Silver | Disrupts bacterial cell membranes | Broad-spectrum; nanocrystalline silver maintains therapeutic levels for days |
| Cadexomer-iodine | Slow iodine release from dextran beads | 1g absorbs up to 7 mL fluid; iodine released as fluid absorbed; avoids cytotoxicity of povidone-iodine; superior to hydrocolloid in venous ulcers |
| Honey (Manuka) | High osmolality + flavonoids + H₂O₂ release | Anti-infective, anti-odor, analgesic; stimulates macrophages; Cochrane review supports use in superficial burns |
| DACC | Hydrophobic binding irreversibly sequesters bacteria | Reduces postoperative skin infections vs. standard dressings |
| Methylene blue/Gentian violet | Broad-spectrum (Gram+, Gram−, Candida) in polyvinyl alcohol sponge | Not released into wound — no tissue toxicity |
| Copper oxide | Bactericidal + pro-angiogenic | Enhanced VEGF and HIF-1α upregulation in diabetic wounds |
⚠️ Cadexomer-iodine: contraindicated in thyroid disease, pregnancy, lactation, children, iodine allergy.
— Dermatology 2-Volume Set 5e
8. Dressing Layers: Primary vs. Secondary
| Layer | Role | Examples |
|---|---|---|
| Primary (contact) layer | Directly touches wound bed; non-adherent | Mepitel, Adaptic, Vaseline gauze, alginates, hydrocolloids |
| Secondary (outer) layer | Absorption, protection, secures primary | Foam, gauze, bandage, film, Tegaderm |
Many highly absorbent dressings (alginates) require a secondary dressing. Some single-layer dressings (hydrocolloids, films) function independently.
9. Wet-to-Dry Dressings (Mechanical Debridement)
- Gauze moistened with saline is applied to wound → allowed to dry → removed
- As it dries, it mechanically debrides necrotic tissue upon removal
- Painful and non-selective (removes both dead and viable tissue)
- Should be discontinued once the wound bed is clean — continued use desiccates granulating tissue
- Safe wound irrigation pressure: 4–15 psi (e.g., 35-mL syringe + 19-gauge needle)
— Textbook of Family Medicine 9e
10. Negative Pressure Wound Therapy (NPWT)
A major advancement in wound care. Subatmospheric pressure is applied to wound via open-cell foam sponge + airtight drape.
Mechanisms:
- Removes chronic edema and inflammatory mediators (MMP-1, MMP-2, MMP-9, TNF-α)
- Increases local blood flow 5×
- Stimulates capillary growth, endothelial proliferation, angiogenesis
- Causes microstrain/macrostrain → increased cellular proliferation, higher microvessel density
- Upregulates VEGF, TGF-β1, TIMP-1
Clinical benefits: Decreased wound volume, accelerated granulation, improved graft take, reduced drainage time, 78% decrease in hospital stay, 76% cost reduction vs. conventional dressings.
Applications: Diabetic ulcers, ischemic ulcers, open abdomen, complex surgical wounds, contaminated wounds.
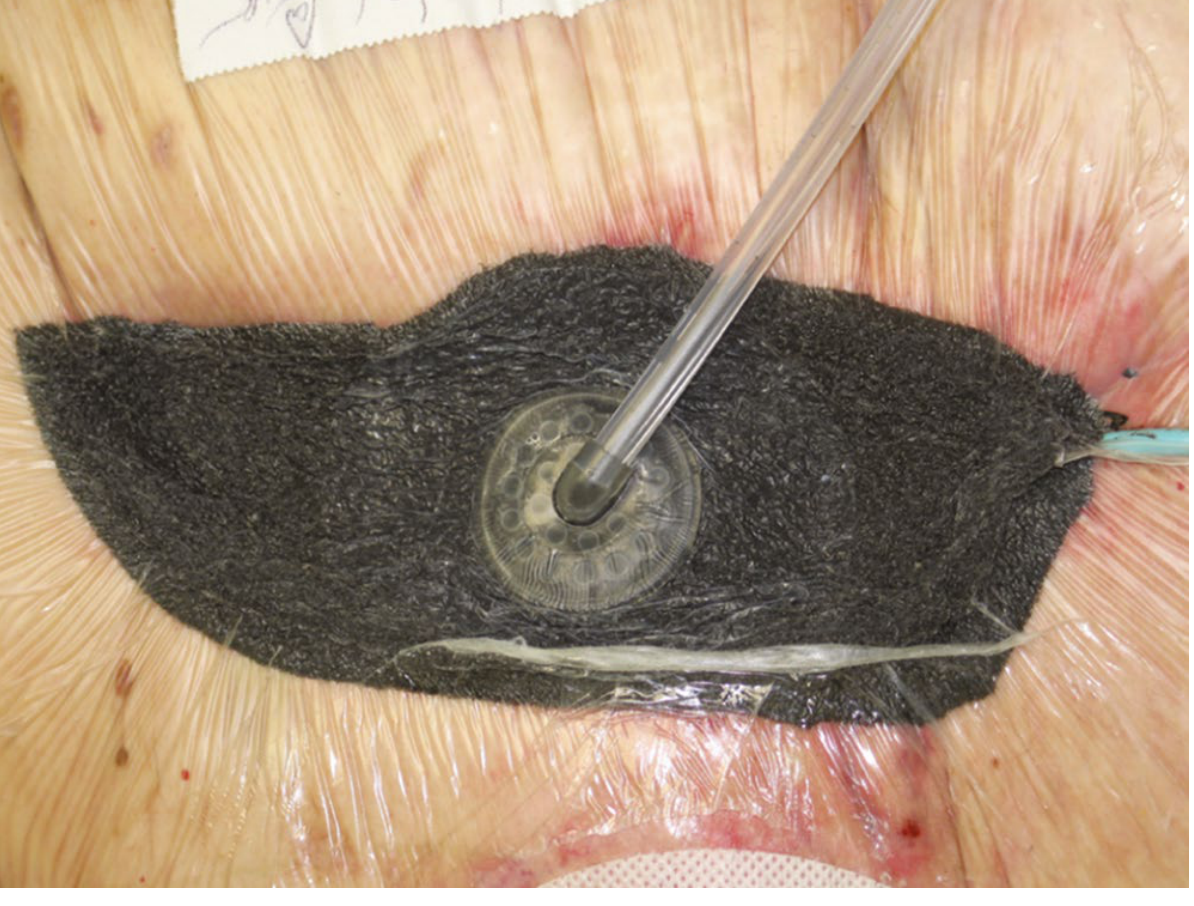
— Sabiston Textbook of Surgery 7e
11. Step-by-Step Dressing Change Procedure
Pre-procedure:
- Explain procedure to patient; obtain consent
- Assemble sterile field: gloves, dressing materials, saline/irrigation, forceps
- Skin disinfectants (e.g., betadine) are no longer routinely recommended — delay wound healing
Removal of old dressing:
- Gently loosen edges — moisten if adherent to prevent re-injury
- Inspect wound: note size, depth, exudate character, odor, surrounding skin
Wound cleansing:
- Irrigate with saline at 4–15 psi
- Debride if necrotic tissue present (sharp, mechanical, enzymatic, or autolytic)
Application of new dressing:
- Select dressing matched to current wound state (exudate, infection, depth)
- Apply primary contact layer directly over wound bed
- Apply secondary absorptive layer if needed
- Secure without excessive compression (can act as tourniquet → increasing pain = remove dressing)
Post-procedure:
- Leave initial pressure dressings intact 24–48 hours (post-surgical)
- Instruct patient: do not wet dressing; replace if wet/contaminated with fresh antibiotic + Tegaderm
- Increasing pain hours after dressing application → suspect tourniquet effect → remove
— Fitzpatrick's Dermatology | Pfenninger and Fowler's Procedures for Primary Care
12. Special Wounds
| Wound Type | Preferred Approach |
|---|---|
| Pressure ulcers (Stage II–III) | Moist dressing (hydrocolloid); reposition every 2 hrs; support surface; protein 1.0–1.5 g/kg/day; calories 30–35 kcal/kg/day |
| Venous ulcers | Compression + cadexomer-iodine or foam |
| Diabetic foot ulcers | Offloading + NPWT; cadexomer-iodine cost-effective |
| Burn wounds | Biologic/nonadherent primary layer; NPWT for complex burns; early excision and grafting for >40% TBSA |
| Post-graft wounds | Initial pressure dressing → change to lighter dressing within 24 hrs |
| Children | Special consideration — children can work free of dressings; plan accordingly |
Summary Mnemonic: "MOIST-CARE"
| Letter | Principle |
|---|---|
| M — Moist environment | Core of modern wound healing |
| O — Occlusion balanced | Match dressing occlusion to exudate level |
| I — Infection control | Antimicrobial dressings when needed |
| S — Selection by wound phase | Dressing changes as wound evolves |
| T — Trauma minimization | Non-adherent layers, gentle removal |
| C — Clean wound bed | Debridement of necrotic tissue |
| A — Absorb exudate appropriately | Prevent maceration |
| R — Re-assess regularly | Change strategy with wound changes |
| E — Evidence-based adjuncts | NPWT, bioactive dressings when indicated |
Key Sources:
- Dermatology 2-Volume Set 5e, Chapter 145 — Traditional and Moisture-Retentive Wound Dressings
- Sabiston Textbook of Surgery, Biological Basis of Modern Surgical Practice — Chapter 23 (Wound Healing)
- Pfenninger and Fowler's Procedures for Primary Care, 3e
- Textbook of Family Medicine 9e — Pressure Sore Management
Disinfectant
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Now I have comprehensive, authoritative content. Let me deliver the full deep-dive answer.
🧪 Disinfectants — Deep Dive
1. Core Definitions (Spaulding & CDC Framework)
| Term | Definition |
|---|---|
| Antisepsis | Use of chemical agents on living tissue (skin, mucosa) to inhibit or eliminate microbes; no sporicidal action implied |
| Disinfection | Use of physical or chemical agents to destroy most microbial forms; bacterial spores and some resistant organisms may survive |
| Sterilization | Destruction of all microbial forms including bacterial spores |
| Germicide | Any chemical that kills microbes — includes bactericide, virucide, sporicide, tuberculocide, fungicide |
| High-level disinfectant | Kills all microbial pathogens except large numbers of bacterial spores |
| Intermediate-level disinfectant | Kills all microbial pathogens except bacterial endospores |
| Low-level disinfectant | Kills most vegetative bacteria and lipid-enveloped, medium-size viruses |
Key Rule: Organic material (serum, feces, sputum, urine) can inactivate most disinfectants — surfaces must be mechanically cleaned before disinfection.
— Medical Microbiology 9e
2. Spectrum of Microbial Resistance (Hardest → Easiest to Kill)
Prions (most resistant)
↓
Bacterial spores (Clostridium, Bacillus)
↓
Mycobacteria (waxy lipid cell wall)
↓
Non-enveloped (non-lipid) viruses (e.g., poliovirus, norovirus)
↓
Fungi
↓
Vegetative (non-spore-forming) bacteria
↓
Enveloped (lipid) viruses (HIV, HBV, influenza) ← EASIEST to kill
This hierarchy explains why spores survive intermediate-level disinfection but enveloped viruses are destroyed even by low-level agents.
3. Disinfection Levels & Clinical Use
| Level | What It Kills | Agents | Use Case |
|---|---|---|---|
| High | All pathogens except large spore burdens | Glutaraldehyde 2–3.2%, H₂O₂ 3–25%, Chlorine compounds 100–1000 ppm, Peracetic acid, Moist heat 75–100°C | Endoscopes, surgical instruments that can't be autoclaved |
| Intermediate | All pathogens except spores | Alcohols 70–95%, Iodophors 30–50 ppm, Phenolics 0.4–5% | Surface/instrument cleaning where spore contamination is unlikely |
| Low | Most vegetative bacteria, lipid viruses | Quaternary ammonium compounds 0.4–1.6% | General environmental surfaces (floors, walls) |
— Medical Microbiology 9e
4. Classification of Chemical Disinfectants by Mechanism
🔵 A. Alcohols
Examples: Ethyl alcohol (ethanol), Isopropyl alcohol, n-Propanol
Working concentration: 60–90% (optimal when diluted with water — pure alcohol is less effective)
Mechanism: Act as "liquid desiccants" — remove water from biological systems, denature proteins, disrupt lipid membranes
Spectrum: Rapid, broad-spectrum — bactericidal, virucidal (enveloped viruses), fungicidal
NOT sporicidal
Clinical use: Hand antisepsis, skin prep before injections, surface wiping
Water is essential for activity — 70% ethanol kills faster than 100% ethanol because water facilitates protein denaturation.
🟢 B. Aldehydes
Examples: Glutaraldehyde, Formaldehyde
Mechanism: Alkylation — react with free amino (–NH₂), hydroxyl (–OH), carboxyl (–COOH), and sulfhydryl (–SH) groups → cross-link proteins and nucleic acids → cell death
Glutaraldehyde:
- Used as 2% solution → achieves sporicidal activity
- More active at alkaline pH ("activated" with NaOH)
- Less toxic than formaldehyde to living tissue but still causes burns on skin/mucous membranes
- Inactivated by organic material — items must be cleaned first
- High-level disinfectant / chemical sterilant for endoscopes and surgical instruments
Formaldehyde:
- Low concentrations = bacteriostatic; high concentrations (20%) = bactericidal/sporicidal
- Combining with alcohol enhances microbicidal activity
- Vapors are carcinogenic — rarely used clinically now
🔴 C. Halogens
Iodine / Iodophors
Mechanism: Highly reactive element — precipitates proteins and oxidizes essential enzymes; microbicidal against virtually all organisms including spores and mycobacteria
- Activity not affected by pH or concentration of solution
- Efficiency increased in acidic pH (more free iodine liberated)
- Iodine acts faster than other halogens or quaternary ammonium compounds
- Inactivated by: serum, feces, sputum, urine, sodium thiosulfate, ammonia
Iodophor (iodine + carrier polymer):
- Povidone-iodine (PVP-I): iodine complexed with polyvinylpyrrolidone → stable, nontoxic to tissues and metals
- Slower iodine release → sustained activity
- ⚠️ Can inhibit wound healing at wound sites — not ideal as wound antiseptic in high concentrations
Chlorine Compounds
Mechanism: Three active forms in water:
- Elemental chlorine (Cl₂) — strong oxidizing agent
- Hypochlorous acid (HOCl) — primary active species
- Hypochlorite ion (OCl⁻)
Chlorine also forms chloramines with nitrogenous compounds.
Acts by irreversible oxidation of sulfhydryl (–SH) groups of essential enzymes; hypochlorites form toxic N-chloro compounds that disrupt cellular metabolism.
Key kinetics:
- Activity inversely proportional to pH (HOCl > OCl⁻ → more active at acid pH)
- Twofold ↑ in concentration → 30% ↓ in killing time
- 10°C ↑ temperature → 50–65% ↓ in killing time
- Inactivated by organic matter and alkaline detergents
Clinical applications of chlorine:
| Agent | Concentration | Use |
|---|---|---|
| Household bleach (NaOCl) | 10% for 10 min | HIV deactivation on surfaces |
| Dilute bleach | 0.5% | Environmental disinfection |
| Chlorhexidine + chlorine | Various | Wound care, skin prep |
HIV-specific: 10% bleach for 10 min at room temperature inactivates ≥10⁵ units of HIV infectivity. In needles/syringes with blood → undiluted bleach for ≥30 seconds required. — Jawetz Melnick & Adelberg's Medical Microbiology 28e
🟡 D. Biguanides — Chlorhexidine
Mechanism: Disrupts cell membrane integrity → leakage of cytoplasmic contents
Spectrum: Bactericidal (Gram+ and Gram−), virucidal (enveloped viruses)
NOT sporicidal; mycobacteria are highly resistant (waxy cell envelope)
Applications: Hand washing, surgical scrub, oral rinses, wound antisepsis, catheter site care, skin prep
Advantages: Persistent activity ("residual effect") on skin — builds up with repeated use; active in presence of whole blood
Hazards:
- Ototoxic (avoid near tympanic membrane)
- Conjunctivitis risk (avoid in eyes)
- ⚠️ Chlorhexidine in alcohol solution allowed to dry before epidural catheterization — several cases of severe arachnoiditis linked to wet contact with neural tissue
🟠 E. Oxidizing Agents
Examples: Hydrogen peroxide (H₂O₂), Peracetic acid, Ozone
Hydrogen Peroxide:
- Active species: Not H₂O₂ itself, but the hydroxyl free radical (•OH) formed by its decomposition
- 3–6%: kills most bacteria
- 10–25%: kills all organisms including spores (sterilant)
- Uses: Plastic implants, contact lenses, surgical prostheses
- Plasma gas sterilization: H₂O₂ vaporized → reactive free radicals generated by microwave/radiofrequency energy → efficient sterilization without toxic byproducts; now replaces ethylene oxide in many settings
- HIV: 0.3% H₂O₂ for 10 min = complete inactivation
Peracetic acid (CH₃CO₃H):
- Oxidizing agent with excellent activity
- End products (acetic acid + oxygen) = nontoxic — major safety advantage
- Used as chemical sterilant (0.2%)
🟣 F. Phenolics
Examples: Phenol, cresol, hexachlorophene, triclosan, PCMX (p-chloro-metaxylenol)
Mechanism: Denature proteins; disrupt cell membranes; coagulate cytoplasm
Spectrum: Bactericidal, fungicidal, virucidal (enveloped viruses) — intermediate level
NOT sporicidal
Uses: Environmental surface disinfection, antiseptic soaps and hand rinses
- Hexachlorophene / Triclosan (bisphenols): bactericidal, sporostatic (not sporicidal); little activity against P. aeruginosa and molds
⚪ G. Quaternary Ammonium Compounds (QACs)
Examples: Benzalkonium chloride, cetrimide
Mechanism: Cationic surfactants — disrupt cell membrane phospholipid bilayer → leakage of intracellular contents
Spectrum: Active against Gram+ bacteria, lipid-enveloped viruses — low-level only
NOT effective against: Gram− bacteria (especially Pseudomonas), mycobacteria, spores, non-enveloped viruses
Uses: Disinfection of non-critical surfaces, food-contact surfaces, skin antisepsis (limited)
⚠️ Can actually support growth of Gram− organisms if contaminated
🔵 H. Heavy Metals
Mercury, Silver, Copper:
- Mercury (merbromin, thimerosal): binds –SH groups → enzyme inactivation; largely abandoned due to toxicity
- Silver: bactericidal — oligodynamic effect; used in wound dressings (silver sulfadiazine) and catheters
- Copper oxide: bactericidal + pro-angiogenic (see wound dressings)
- Reversal: mercuric ion inactivated by adding sulfhydryl compounds (thioglycolic acid)
5. Physical Disinfection & Sterilization Methods
| Method | Mechanism | Parameters | Notes |
|---|---|---|---|
| Moist heat / Autoclave | Protein denaturation, membrane disruption | 121°C / 15 psi / 15 min (spores); 132°C for shorter cycles | Standard; 1.7°C drop needs 48% longer exposure |
| Dry heat | Protein oxidation/denaturation | 150–170°C / 1 hour | Less efficient than moist heat; damages instruments |
| Boiling (100°C) | Kills vegetative bacteria in 2–3 min | Not sporicidal | Disinfection only |
| Pasteurization | Moist heat 75–100°C for 30 min | High-level disinfection | |
| UV radiation (254–260 nm) | Thymine dimer formation → DNA replication failure | Variable | Bactericidal; not reliably sporicidal; surface use only |
| Ionizing radiation (gamma/X-ray, ≤1 nm) | Free radical formation → protein/DNA/lipid damage | Variable | Bactericidal AND sporicidal; industrial sterilization of single-use items |
| Filtration | Mechanical exclusion | 0.22–0.45 μm pore; HEPA | Sterilizes heat-sensitive liquids; HEPA filters air |
| Ethylene oxide gas | Alkylation of DNA/proteins | 450–1200 mg/L at 29–65°C for 2–5 hr; aerate 12 hr after | Carcinogenic, explosive, flammable — regulated; temperature/pressure-sensitive items |
| Plasma gas | Ionized H₂O₂ → free radicals | 30% H₂O₂ at 55–60°C | Replaced ethylene oxide in many settings; no toxic byproducts |
6. Factors Affecting Disinfectant Efficacy
- Organic load — blood, pus, serum, feces inactivate most agents → clean before disinfecting
- Concentration — higher concentration generally = faster kill (follow manufacturer specs)
- Contact time — inadequate exposure = incomplete kill
- Temperature — higher temperature = faster kill (e.g., 10°C rise → 50–65% less time for chlorine)
- pH — chlorine more active at acid pH; glutaraldehyde more active at alkaline pH
- Type of organism — see resistance spectrum above
- Biofilm — organisms in biofilm are far more resistant than planktonic forms
7. Sterilization vs. Disinfection: Methods Comparison Table
| Method | Level | Sporicidal | Mycobactericidal | Virucidal (non-lipid) | Equipment |
|---|---|---|---|---|---|
| Autoclave (steam) | Sterilization | ✅ | ✅ | ✅ | Heat-stable items |
| Ethylene oxide | Sterilization | ✅ | ✅ | ✅ | Heat/pressure-sensitive |
| Glutaraldehyde 2% | High | ✅ (prolonged) | ✅ | ✅ | Endoscopes |
| H₂O₂ 10–25% | High/Sterilant | ✅ | ✅ | ✅ | Implants, lenses |
| Chlorine 100–1000 ppm | High | ✅ | ✅ | ✅ | Surfaces, water |
| Alcohol 70–90% | Intermediate | ❌ | ✅ | ✅ (lipid only) | Skin, surfaces |
| Iodophors | Intermediate | ❌ | ✅ | ✅ | Skin, surfaces |
| Phenolics | Intermediate | ❌ | ✅ | ✅ (lipid only) | Surfaces |
| QACs | Low | ❌ | ❌ | ❌ (non-lipid) | Non-critical surfaces |
8. Disinfectant Kinetics
Biocide activity is time- and concentration-dependent, governed by:
$$C^n \cdot t = K$$
Where:
- C = concentration
- t = time to kill a fixed fraction of organisms
- n = dilution coefficient (reflects how sensitive the agent is to dilution)
- K = constant
If n is large → small dilutions dramatically reduce efficacy (hypochlorites)
If n is small → efficacy relatively preserved over dilution ranges (quaternary ammonium compounds)
Reversal of disinfectant action can occur by:
| Mechanism | Example |
|---|---|
| Agent removal | Washing organisms off the surface |
| Substrate competition | High substrate concentration displaces competitive inhibitor from enzyme |
| Agent inactivation | Sulfhydryl compounds (thioglycolic acid) neutralize mercuric ion |
— Jawetz Melnick & Adelberg's Medical Microbiology 28e
9. Specific Clinical Applications
| Setting | Agent of Choice | Rationale |
|---|---|---|
| Surgical hand scrub | Chlorhexidine or iodophor | Persistent activity (chlorhexidine), broad spectrum |
| Skin prep before injection | 70% isopropyl alcohol | Fast, broad-spectrum, no residue |
| Preoperative skin prep | Chlorhexidine-alcohol | Superior to povidone-iodine for SSI prevention |
| Wound antisepsis | Dilute cadexomer-iodine or chlorhexidine | Avoid concentrated povidone-iodine (delays healing) |
| Endoscope reprocessing | Glutaraldehyde 2% or peracetic acid | High-level disinfection; can't autoclave |
| IV catheter site care | Chlorhexidine-based antiseptic | Change minimum every 7 days |
| Environmental surfaces (hospitals) | Quaternary ammonium, hypochlorite | Ease of use; WHO recommendations |
| HIV needle/syringe decontamination | 10% household bleach (undiluted, ≥30 sec) | Complete inactivation in blood-containing needles |
| Water disinfection | Chlorine | Most widely used globally |
10. Key Agents — Pros, Cons, Pitfalls
| Agent | ✅ Advantages | ❌ Disadvantages / Pitfalls |
|---|---|---|
| Chlorhexidine | Persistent residual activity; active in blood | Ototoxic; conjunctivitis; arachnoiditis risk if wet near epidural |
| Povidone-iodine | Broad-spectrum, fast acting | Inhibits wound healing; inactivated by organics |
| Glutaraldehyde | Sporicidal; works at room temp | Toxic burns; inactivated by organics; requires aeration |
| Alcohols | Fast, safe, cheap | Not sporicidal; flammable; dries skin |
| Hypochlorite (bleach) | Cheap, broad-spectrum, sporicidal | Inactivated by organics; corrosive at high concentrations |
| QACs | Gentle on surfaces, cheap | Low spectrum; supports Gram− growth if contaminated |
| Peracetic acid | Excellent activity, nontoxic byproducts | Corrosive to skin/eyes; short shelf life in solution |
Summary Mnemonic: "A-HAPH-QO" (Agents with Ascending kill power)
| Letter | Agent | Key Fact |
|---|---|---|
| A | Alcohols | Intermediate; NOT sporicidal |
| H | Halogens (iodine, chlorine) | Intermediate-to-high; fast acting |
| A | Aldehydes (glutaraldehyde) | High / sterilant; sporicidal |
| P | Peracetic acid | Sterilant; safest byproducts |
| H | H₂O₂ (hydrogen peroxide) | High; works via hydroxyl radical |
| Q | QACs | Low level only |
| O | Oxidizers (ozone, plasma) | Highest level; no toxic residue |
Key Sources:
- Medical Microbiology 9e — Chapter 3 (Sterilization, Disinfection, Antisepsis)
- Jawetz, Melnick & Adelberg's Medical Microbiology 28e — Chapter 4 (Biocides and Kinetics)
- Jawetz — Chapter 44 (HIV Inactivation data)
This is a shared conversation. Sign in to Orris to start your own chat.