Explain how folic acid deficiency leads to impaired DNA synthesis in RBC precursors.
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Loading Image
I now have all the material needed for a complete, well-sourced answer.
How Folic Acid Deficiency Impairs DNA Synthesis in RBC Precursors
1. The Role of Folate: Tetrahydrofolate as a One-Carbon Carrier
Folic acid (pteroylmonoglutamic acid) must first be reduced in two steps — first to dihydrofolate (FH₂) by dihydrofolate reductase, and then to tetrahydrofolate (FH₄) by the same enzyme. FH₄ is the biologically active form and functions as an acceptor and donor of one-carbon fragments acquired from compounds such as serine and formiminoglutamic acid.
These one-carbon transfers are essential for three key biosynthetic reactions:
- Purine synthesis — required for both adenine and guanine, the building blocks of DNA and RNA
- Methionine synthesis — conversion of homocysteine → methionine (also requires vitamin B₁₂)
- dTMP (thymidylate) synthesis — the single most critical step for DNA production
In the first two reactions, FH₄ is regenerated and re-enters the one-carbon donor pool. In dTMP synthesis, however, FH₄ is oxidised to FH₂ (dihydrofolate) as a byproduct and must be re-reduced by dihydrofolate reductase to re-enter the active pool. — Robbins, Cotran & Kumar: Pathologic Basis of Disease, p. 611
2. The Critical Bottleneck: dTMP Synthesis
The reaction most severely disrupted by folate deficiency is catalysed by thymidylate synthase:
dUMP + 5,10-methylene-FH₄ → dTMP + FH₂
- 5,10-methylene-THF donates a methylene group and two hydrogen atoms to convert dUMP to deoxythymidine monophosphate (dTMP).
- Without sufficient FH₄, thymidine (T) cannot be synthesised endogenously.
- Since thymidine is unique to DNA (RNA uses uracil), its absence specifically and selectively cripples DNA synthesis.
"Among the molecules whose synthesis is dependent on folates, dTMP is perhaps the most important biologically because it is required for DNA synthesis." — Robbins, Cotran & Kumar, p. 611
3. Why RBC Precursors Are Preferentially Affected
Erythroid precursors in the bone marrow undergo rapid, successive cell divisions to produce the ~200 billion new red cells needed daily. This places an exceptionally high demand on DNA replication. When folate is deficient:
- Thymidine availability drops, and DNA synthesis slows or stalls.
- Cells attempt to grow and accumulate haemoglobin (cytoplasmic maturation proceeds normally) but cannot complete nuclear division.
- This creates characteristic nuclear-cytoplasmic asynchrony: a large, haemoglobinised cell with an immature, finely dispersed chromatin pattern — the megaloblast.
"Deficiency of folic acid... affects cells that are dividing rapidly because they have a large requirement for thymidine for DNA synthesis. Clinically, this affects the bone marrow, leading to megaloblastic anemia." — Harper's Illustrated Biochemistry, 32nd Ed, p. 557
4. Consequences in the Bone Marrow and Blood
The impaired DNA synthesis has several downstream effects:
| Feature | Mechanism |
|---|---|
| Megaloblasts | Nuclear maturation blocked; cytoplasm matures normally → nuclear-cytoplasmic asynchrony |
| Ineffective erythropoiesis | Most precursors cannot complete division and undergo apoptosis in the marrow |
| Macroovalocytes in blood | Cells that do escape are abnormally large (macro-ovalocytes), lacking central pallor |
| Hypersegmented neutrophils | Same DNA synthesis defect affects granulocyte precursors → giant metamyelocytes and 5+ nuclear lobes |
| Pancytopenia | Granulocyte and platelet precursors also affected |
| Marrow hypercellularity | Driven by elevated erythropoietin compensating for ineffective output |
— Robbins, Cotran & Kumar, pp. 608–611
Bone marrow aspirate in megaloblastic anaemia:
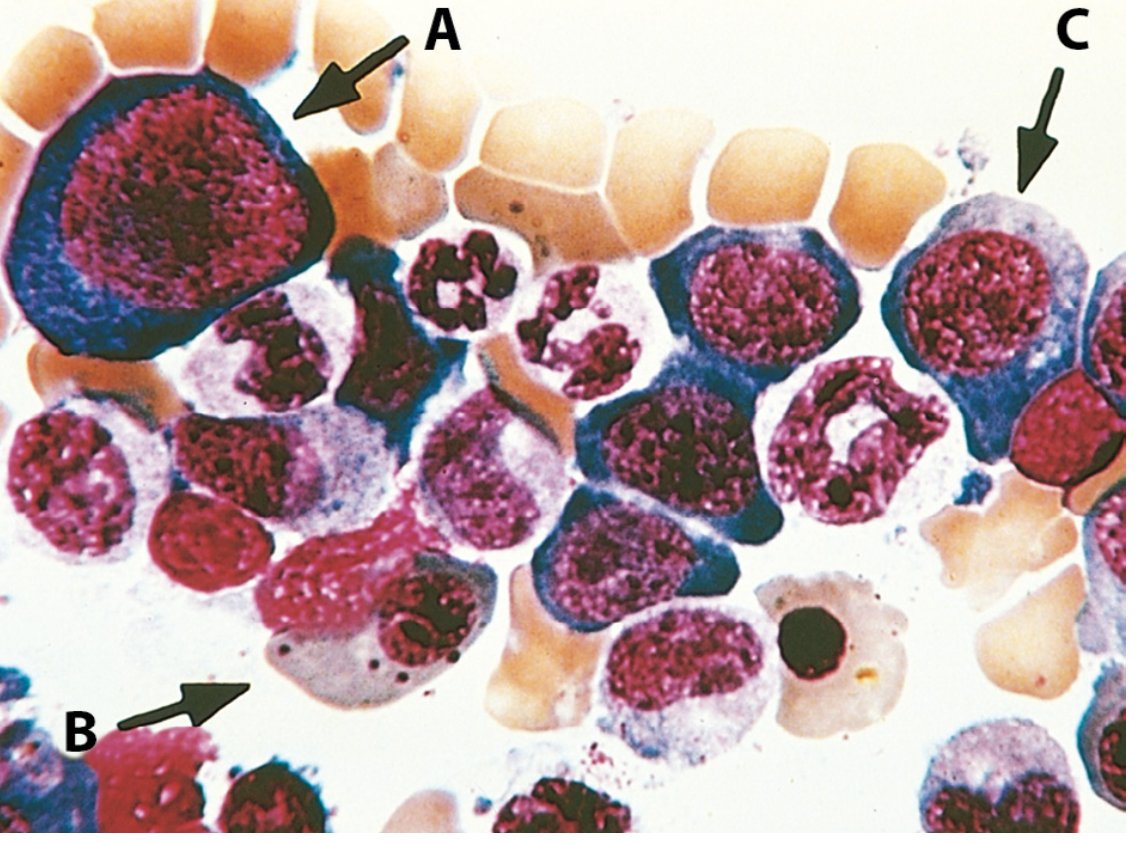
Fig. 14.16 — Megaloblastic anaemia, bone marrow aspirate. A and C are early erythroid precursors with abnormally immature chromatin. B is an orthochromatic megaloblast — haemoglobinised but without the pyknotic nucleus expected of a normal normoblast. (Robbins, p. 609)
5. The Vitamin B₁₂ Connection: "Folate Trap"
Vitamin B₁₂ deficiency produces the identical haematological picture through a different route. Methionine synthase (B₁₂-dependent) normally demethylates 5-methylTHF back to free THF. Without B₁₂, 5-methylTHF accumulates and cannot be converted to the active forms needed for dTMP synthesis — folate is effectively trapped in a form the cell cannot use. The final common pathway is still folate unavailability → thymidine deficit → impaired DNA synthesis. — Harper's Illustrated Biochemistry, 32nd Ed, p. 557
Summary
The chain of events is:
Folate deficiency → ↓ active FH₄ → ↓ 5,10-methylene-THF → ↓ thymidylate synthase activity → ↓ dTMP → ↓ DNA synthesis → arrest of nuclear maturation in rapidly dividing RBC precursors → ineffective erythropoiesis + megaloblastic anaemia
This is a shared conversation. Sign in to Orris to start your own chat.