Ivabradin
Reading File
Checking Date & Time
Finding Sources
Searching PubMed
ivabradine clinical use heart failure
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of ivabradine.
Ivabradine (Corlanor)
Class: HCN (hyperpolarization-activated cyclic nucleotide-gated) channel blocker — the only drug in this class.
Mechanism of Action
Ivabradine selectively blocks the funny current (I_f) — a mixed Na⁺/K⁺ inward current that is activated by hyperpolarization and is responsible for spontaneous diastolic depolarization in the sinoatrial (SA) node. It blocks the intracellular portion of the ion pore, reducing the slope of diastolic depolarization and prolonging the time between action potentials, which slows heart rate in a dose- and use-dependent manner.
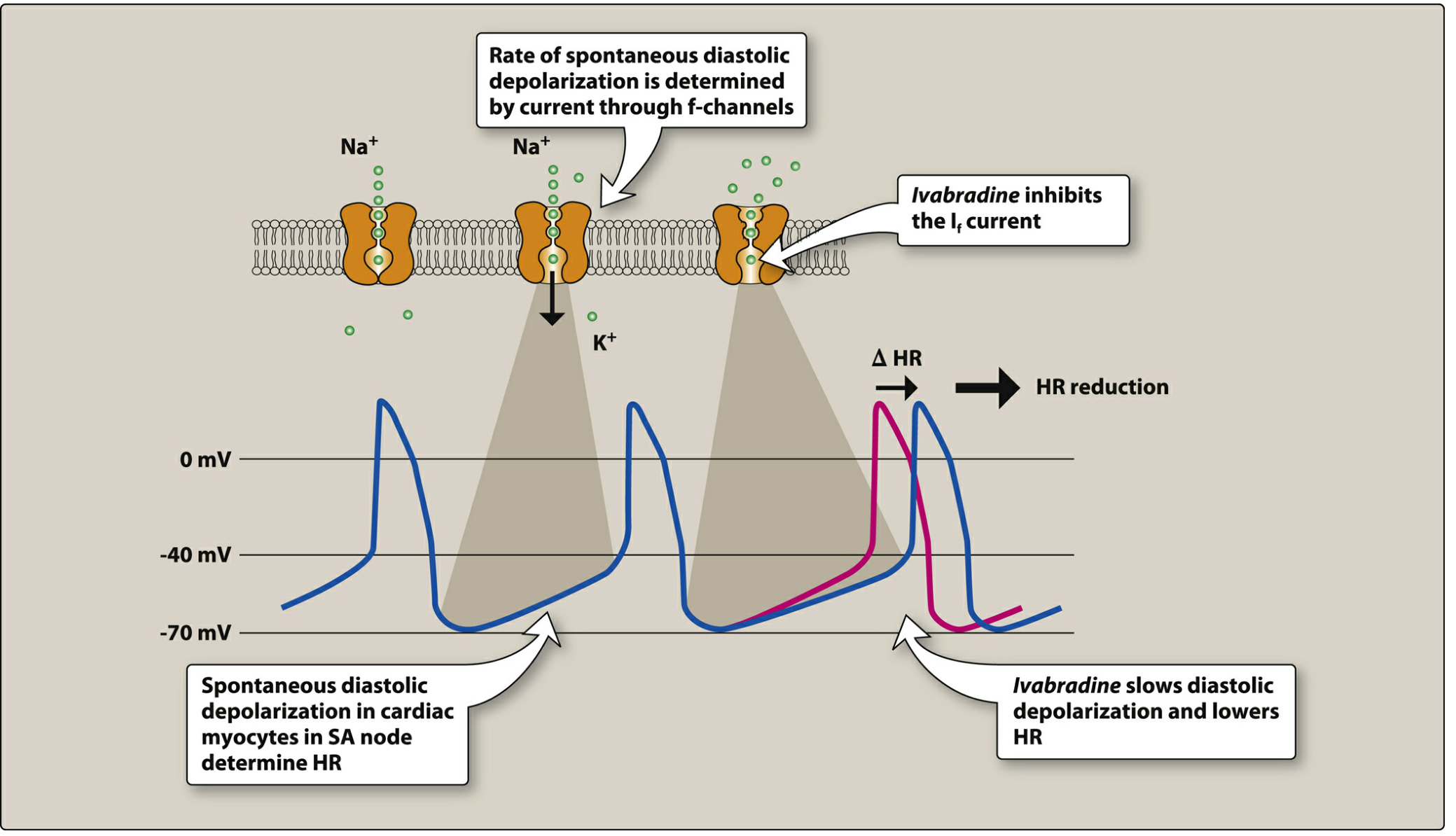
Ivabradine slows diastolic depolarization in the SA node, reducing heart rate without affecting contractility or repolarization — Lippincott Illustrated Reviews: Pharmacology
Key selectivity: The effect is entirely confined to the sinus node. Ivabradine has:
- No effect on blood pressure
- No effect on myocardial contractility
- No effect on AV conduction or ventricular repolarization
- No effect on intracardiac conduction
Because it requires an open channel to bind (use-dependence), its heart rate–lowering effect is greatest at higher heart rates.
FDA-Approved Indication
Heart failure with reduced ejection fraction (HFrEF):
Indicated to reduce the risk of hospitalization for worsening heart failure in patients with:
- Stable, symptomatic HF (NYHA class II–IV)
- LVEF ≤ 35%
- Sinus rhythm with resting heart rate ≥ 70 bpm
- On maximally tolerated doses of a beta-blocker (or with a contraindication to beta-blockers)
Key Trial: SHIFT
The SHIFT trial (n = 6,588) randomized patients meeting the above criteria to ivabradine vs. placebo. Ivabradine produced an 18% reduction in the composite of cardiovascular death or HF hospitalization (primarily driven by reductions in HF hospitalization). It also improved LVEF and quality of life.
Other Uses (Off-label)
- Inappropriate sinus tachycardia — especially when beta-blockers and non-dihydropyridine calcium channel blockers have failed or are not tolerated
- Stable angina (approved in Europe, not the US) — reduces oxygen demand by lowering heart rate without affecting contractility
Dosing
| Step | Dose |
|---|---|
| Starting dose | 5 mg twice daily (with meals) |
| If resting HR < 60 bpm at baseline | 2.5 mg twice daily |
| Titration (after 14 days) | Increase to 7.5 mg twice daily if HR > 60 bpm |
| If HR < 50 bpm or symptomatic bradycardia | Reduce to 2.5 mg twice daily or discontinue |
Pharmacokinetics
| Parameter | Detail |
|---|---|
| Administration | With meals (increases absorption) |
| Metabolism | Extensive first-pass hepatic metabolism via CYP3A4 → active metabolite (also a CYP3A4 substrate) |
| Volume of distribution | High |
| Protein binding | ~70% |
| Half-life | ~6 hours → allows twice-daily dosing |
| Dose adjustment | Required in severe hepatic or renal impairment |
Adverse Effects
| Effect | Notes |
|---|---|
| Bradycardia | Most common cardiac adverse effect; often managed with dose reduction |
| Phosphenes / luminous phenomena | Transient brightness, halos, or flashes in the visual field (inhibition of I_f-like channels in the retina); usually mild, dose-reducible |
| Atrial fibrillation | Increased risk; if AF develops, discontinue ivabradine (ineffective and potentially harmful in AF) |
| AV block | Caution with pre-existing sinus bradycardia or first-degree AV block |
Contraindications & Cautions
- Pregnancy / breastfeeding — contraindicated (fetal toxicity)
- Atrial fibrillation — not effective for rate control; not indicated
- Advanced heart block (without pacemaker)
- Potent CYP3A4 inhibitors (e.g., azole antifungals, macrolides, diltiazem, grapefruit juice) — increase ivabradine levels → risk of severe bradycardia
- CYP3A4 inducers (e.g., rifampin, St. John's Wort) — reduce efficacy
- QT-prolonging agents (e.g., amiodarone) — combination may increase TdP risk
- Not a substitute for beta-blockers — it does not have the mortality benefit or the sympathetic blockade of beta-blockers
Summary Table
| Property | Detail |
|---|---|
| Drug class | HCN channel blocker (I_f inhibitor) |
| Target | SA node funny current (I_f) |
| Effect | Pure heart rate reduction (negative chronotropy) |
| Approved use | HFrEF + sinus rhythm + HR ≥ 70 |
| Mechanism diagram | Slows diastolic depolarization slope |
| Key trial | SHIFT (18% ↓ CV death/HF hospitalization) |
| Main side effects | Bradycardia, phosphenes, ↑AF risk |
Sources:
- Braunwald's Heart Disease, 12th Ed.
- Fuster and Hurst's The Heart, 15th Ed.
- Goldman-Cecil Medicine
- Lippincott Illustrated Reviews: Pharmacology
This is a shared conversation. Sign in to Orris to start your own chat.