Essay answer on craniopharyngioma for ent post graduation exam
Reading File
Finding Sources
Finding Sources
Searching PubMed
craniopharyngioma management treatment
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Loading Image
Reading File
Finding Sources
Reading File
Loading Image
I now have comprehensive, authoritative content from multiple major textbooks. Let me compose the full essay answer.
Craniopharyngioma — Essay Answer for ENT Postgraduate Examination
Introduction
Craniopharyngioma is a histologically benign (WHO Grade I), slow-growing epithelial tumour arising from vestigial remnants of Rathke's pouch — the embryological evagination of the primitive stomodeum (ectoderm of the roof of the mouth) that gives rise to the anterior pituitary. Despite its benign histology, it carries significant morbidity and mortality because of its intimate relationship with the optic chiasm, hypothalamus, pituitary stalk, and third ventricle.
Epidemiology
- Accounts for 1%–5% of all intracranial tumours and 3%–5% of all paediatric brain tumours
- Most common non-glial, suprasellar tumour in children
- Bimodal age distribution: first peak at 5–15 years (adamantinomatous type); second peak in adults ≥65 years (papillary type)
- No significant sex predilection
- Incidence: fewer than 500 cases/year in adults in Western populations
(Bradley and Daroff's Neurology in Clinical Practice; Robbins, Cotran & Kumar Pathologic Basis of Disease)
Embryology & Pathogenesis
Rathke's pouch arises at approximately 3–4 weeks of gestation as an ectodermal evagination from the roof of the primitive stomodeum. It ascends toward the diencephalon and comes to lie at the base of the brain, where its stalk normally obliterates. Persistence or incomplete regression of epithelial cell rests along this migratory path — from the oro-nasopharynx up to the sella turcica — gives rise to craniopharyngioma. This explains why rare ectopic cases can present as nasopharyngeal masses (Cummings Otolaryngology; The Developing Human, Clinically Oriented Embryology).
Molecular pathogenesis distinguishes the two subtypes:
- Adamantinomatous type: near-universal (92–94%) CTNNB1 (β-catenin) gene mutations → aberrant nuclear β-catenin accumulation → activation of Wnt signalling pathway
- Papillary type: high rates (92–95%) of BRAF p.V600E mutations → activation of the RAS–MAP kinase pathway
(Bradley and Daroff; Robbins & Cotran; Grainger & Allison)
Pathology
Gross Appearance
- Average diameter: 3–4 cm
- Usually suprasellar with or without intrasellar extension; 5% purely intrasellar
- May be encapsulated and solid, but more often cystic and multiloculated
- Cysts contain a characteristic thick, dark, cholesterol-rich, machine-oil–like fluid
- Tumour frequently encroaches on the optic chiasm, cranial nerves, and floor of the third ventricle
Histological Variants
| Feature | Adamantinomatous | Papillary |
|---|---|---|
| Age | Children & young adults | Adults (almost exclusively) |
| Architecture | Nests/cords of stratified squamous epithelium; stellate reticulum; peripheral palisading | Solid sheets + papillae lined by well-differentiated squamous epithelium |
| Wet keratin | Present — diagnostic (compact lamellar keratin; "ghost" keratinocyte nests) | Absent |
| Calcification | Frequent (~75%); dystrophic | Rare |
| Cysts | Common; machine-oil fluid | Uncommon |
| Goblet cells | Absent | Present |
| Mutation | CTNNB1 (β-catenin) | BRAF V600E |
| Infiltration | Sends finger-like extensions → glial reaction | Less infiltrative |
Wet keratin is pathognomonic — it is diagnostic even on a small biopsy lacking viable epithelium, distinguishing craniopharyngioma from epidermoid cysts (dry, flaky keratin).
(Robbins & Cotran; Bradley and Daroff)
Histology — Adamantinomatous Craniopharyngioma:
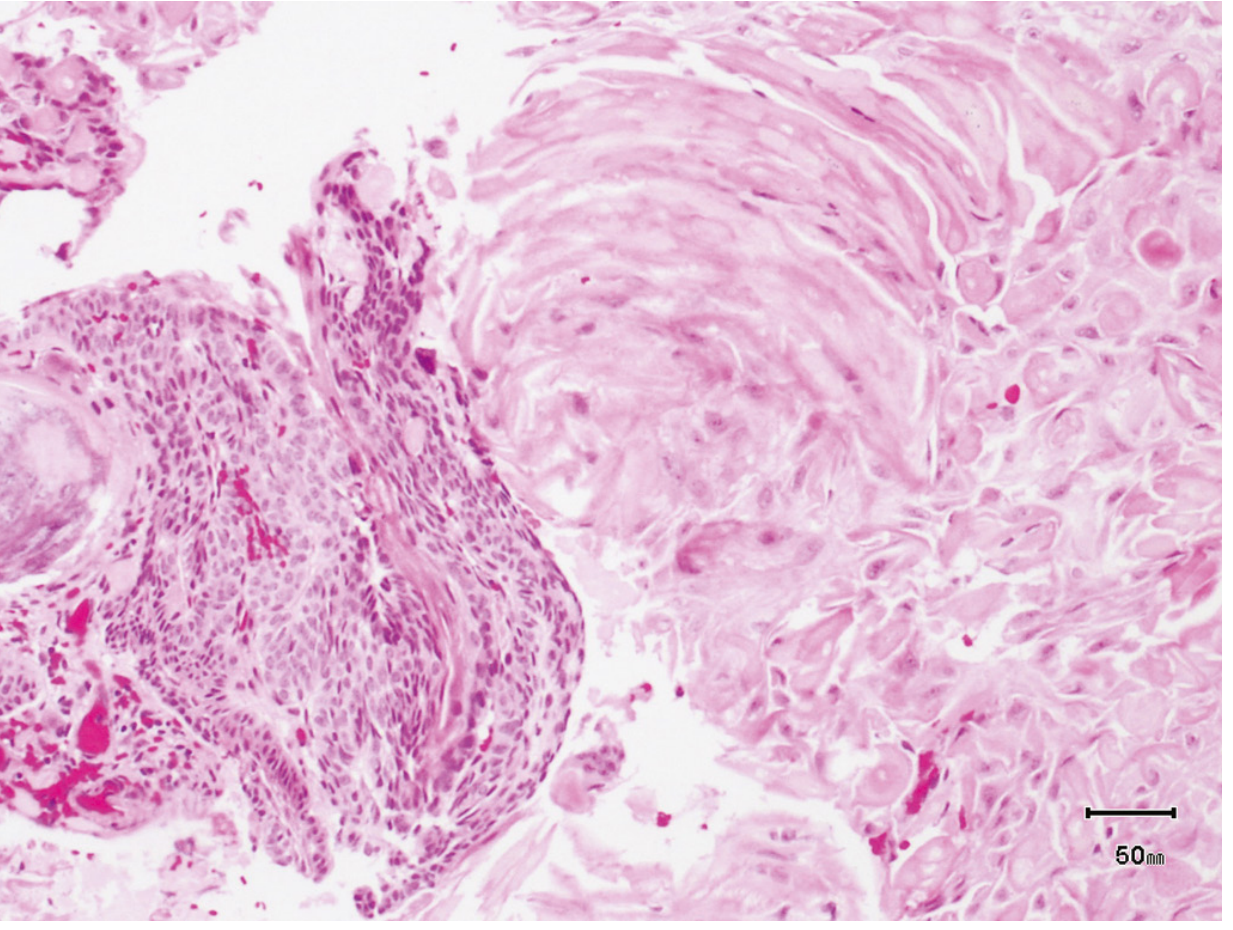
Clinical Features
Onset
- Insidious — a 1–2 year history of slowly progressive symptoms is typical
- Symptoms arise from compression of adjacent structures
Symptom Triad
- Raised intracranial pressure (headache, vomiting, papilloedema) — from hydrocephalus due to third ventricle obstruction
- Visual disturbance — bitemporal hemianopia (chiasmal compression); may progress to optic atrophy
- Endocrine dysfunction — hypopituitarism (most common endocrine manifestation):
- Growth hormone deficiency → growth retardation, short stature (>70% of children at diagnosis)
- Gonadotrophin deficiency → delayed puberty, amenorrhoea
- TSH, ACTH, and LH/FSH deficiencies (panhypopituitarism)
- Diabetes insipidus (hypothalamic/stalk involvement)
- Hyperprolactinaemia (stalk effect — loss of dopaminergic inhibition)
Hypothalamic Involvement (particularly severe)
- Severe obesity (damage to satiety centres)
- Memory impairment, psychomotor slowing
- Altered sleep–wake rhythms
- Thermoregulatory dysfunction
- Loss of impulse control, behavioural changes
ENT-Relevant Presentation
- Rare ectopic presentation as a nasopharyngeal mass — can masquerade as hypertrophied adenoid tissue, especially in children
- Presents with nasal obstruction, epistaxis, or features mimicking adenoidal hypertrophy
- Must be considered in differential of nasopharyngeal lesions listed in Box 95.1 (Cummings Otolaryngology)
(Bradley and Daroff; Grainger & Allison; Goldman-Cecil Medicine; Cummings Otolaryngology)
Investigations
Neuroimaging
CT Scan:
- Suprasellar calcification in ~75% of cases — virtually pathognomonic in children
- Calcified, mixed cystic-solid mass
- Non-contrast CT best demonstrates calcification
MRI (investigation of choice):
- T1: Cystic component often hyperintense (due to proteinaceous/cholesterol-rich fluid); solid component isointense
- T2/T2 FLAIR: Cystic components hyperintense
- T1 + contrast: Enhancement of solid component and cyst wall
- Identifies relationship to optic chiasm, hypothalamus, and pituitary stalk
- MR spectroscopy: high lipid peaks
- Large tumours → hydrocephalus, forward displacement/stretching of optic nerves and chiasm
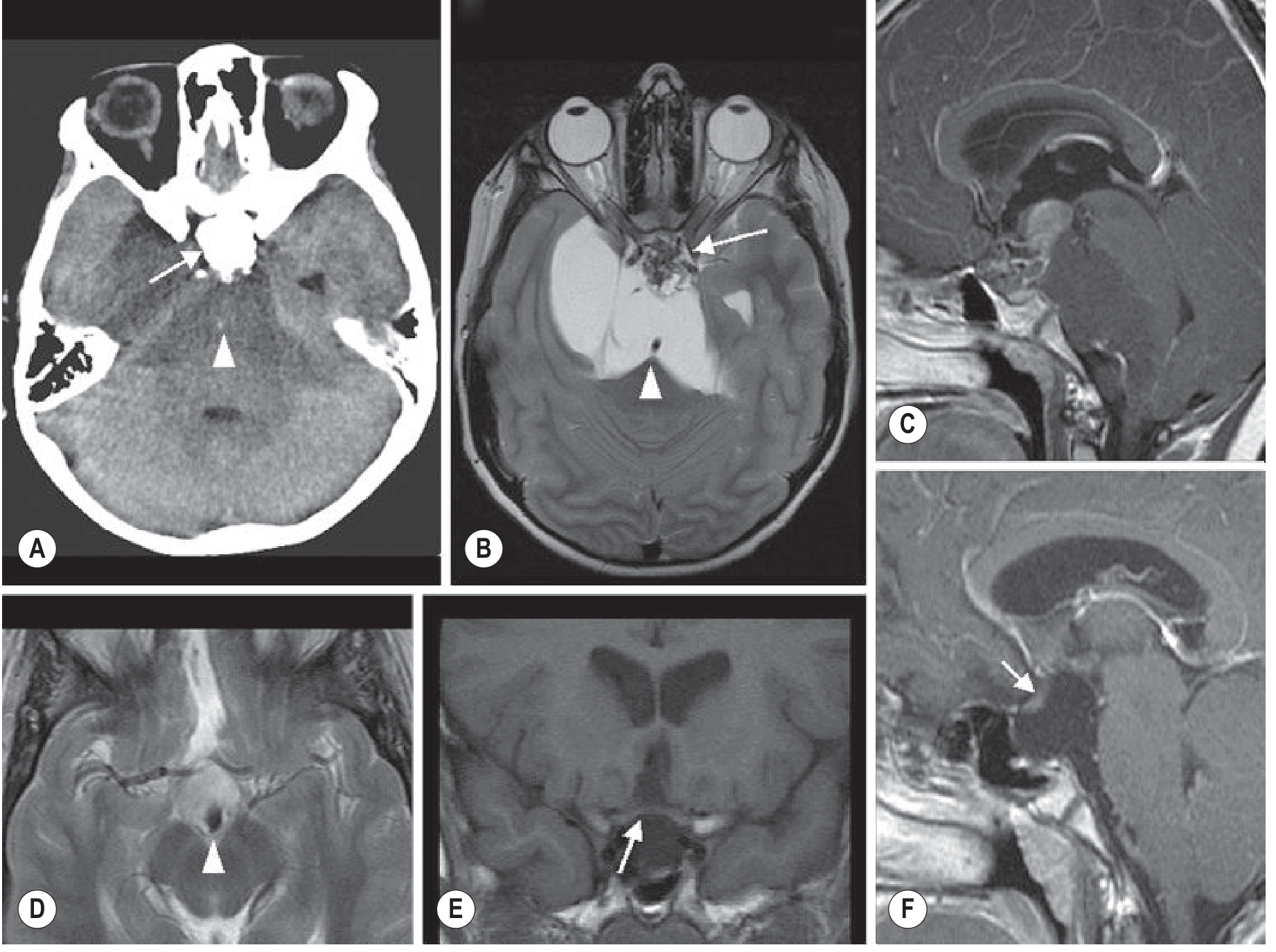
Endocrine Evaluation
- Basal pituitary hormone panel: GH/IGF-1, LH, FSH, TSH, free T4, ACTH/morning cortisol, prolactin
- Water deprivation test / paired plasma and urine osmolality (diabetes insipidus)
- Visual field assessment (Humphrey perimetry — bitemporal hemianopia)
Differential Diagnosis of Suprasellar Mass
- Pituitary adenoma
- Germinoma / dysgerminoma
- Rathke's cleft cyst (cuboidal epithelium; white mucoid fluid; no calcification)
- Meningioma
- Optic pathway glioma
- Langerhans cell histiocytosis
- Arachnoid cyst
- Hypothalamic hamartoma
- Nasopharyngeal carcinoma (ectopic craniopharyngioma mimicry)
Management
Management is multidisciplinary (neurosurgery, endocrinology, radiation oncology, ophthalmology). The strategy balances maximal tumour control against preservation of the hypothalamic–pituitary axis.
1. Surgery — Primary Treatment
Transsphenoidal resection:
- Preferred for subdiaphragmatic (intrasellar) tumours and small suprasellar extensions
- Associated with lower incidence of postoperative diabetes insipidus
- Endoscopic endonasal extended transsphenoidal approach increasingly used
Transcranial resection:
- Subfrontal, pterional, or interhemispheric approaches
- Required for large suprasellar/third ventricular tumours
Extent of resection — the central controversy:
- Radical (gross total) resection: achieves best local control but risks profound hypothalamic damage → severe obesity, panhypopituitarism, visual loss, memory impairment, behavioural change
- Hypothalamic-sparing (limited) resection: less morbidity, similar or acceptable recurrence rates when combined with radiation; advocated by UK guidelines
- Surgical margins are deceptive — microscopic finger-like extensions are common; complete resection does not guarantee recurrence-free survival
2. Radiotherapy
- External beam radiotherapy (EBRT): extends progression-free survival after incomplete resection; long-term survival achievable with surgery + radiation
- Stereotactic radiosurgery (Gamma Knife): suitable for small residual/recurrent disease; good local control
- Intracavitary irradiation: ³²P or ⁹⁰Y instilled stereotactically into cystic component — effective for predominantly cystic tumours
- Intracavitary bleomycin: for cystic components following stereotactic aspiration
- Radiation dose typically 50–55 Gy in conventional fractionation
- Combined limited surgery + adjuvant radiotherapy: maximises quality of life while achieving >90% long-term survival in children
(2025 systematic review — Harary et al., J Neurooncol 2025 [PMID 40063185]: spectrum of RT options confirms multiple modalities)
3. Targeted Molecular Therapy (Emerging)
- Papillary craniopharyngiomas with BRAF V600E mutation: BRAF inhibitors (vemurafenib, dabrafenib) ± MEK inhibitors (trametinib) have shown remarkable responses in case series and small studies
- Phase II trials underway combining BRAF + MEK inhibition
- Potentially transformative — may offer non-surgical cytoreduction pre-operatively or as primary treatment in unresectable cases
- Adamantinomatous type: drugs targeting β-catenin/Wnt pathway under development
4. Postoperative Endocrine Replacement
- Virtually all patients require lifelong hormone replacement following surgery:
- Hydrocortisone (cortisol replacement)
- Levothyroxine (thyroid replacement)
- Sex steroids (oestrogen/testosterone)
- GH replacement (especially in children)
- Desmopressin (DDAVP) for diabetes insipidus
- Management of hypothalamic obesity (dietary, behavioural, pharmacological measures)
5. Management of Hydrocephalus
- Ventriculoperitoneal (VP) shunting or external ventricular drain if hydrocephalus is severe
- Often resolves after tumour resection
Prognosis
- Overall survival: >90% at 10 years in children with combined modality treatment
- Recurrence rate: 25–50% after radical resection; higher after subtotal resection alone
- Main cause of morbidity: hypothalamic dysfunction (obesity, cognitive impairment, endocrine sequelae) — often more debilitating than the tumour itself
- Recurrence management: surgery + radiation, stereotactic radiosurgery, or targeted therapy (BRAF tumours)
- Quality of life is significantly impaired in many long-term survivors, primarily due to metabolic and neurocognitive sequelae
(Systematic review: Zheng et al., J Clin Neurosci 2023 [PMID 37922728]; Tang et al., Childs Nerv Syst 2024 [PMID 37466685])
Summary Table
| Feature | Detail |
|---|---|
| Origin | Rathke's pouch epithelial remnants |
| WHO Grade | I (histologically benign) |
| Incidence | 1–5% intracranial tumours; most common suprasellar tumour in children |
| Age | Bimodal: 5–15 yr (adamantinomatous) + >65 yr (papillary) |
| Location | Suprasellar (majority) ± intrasellar; rare nasopharyngeal |
| Classic triad | Raised ICP + visual field defect + endocrine dysfunction |
| Key imaging sign | Suprasellar calcification on CT (75%) |
| Diagnostic histology | Wet keratin (pathognomonic) |
| Adamantinomatous mutation | CTNNB1 (β-catenin) / Wnt pathway |
| Papillary mutation | BRAF V600E |
| Primary treatment | Surgery (transsphenoidal or transcranial) |
| Adjuvant | Radiotherapy (EBRT / stereotactic / intracavitary) |
| Targeted therapy | BRAF ± MEK inhibitors (papillary type) |
| Prognosis | >90% 10-yr survival; high endocrine and hypothalamic morbidity |
ENT-Specific Relevance
From an ENT perspective, craniopharyngioma is relevant in the following contexts:
- Differential diagnosis of nasopharyngeal masses — ectopic craniopharyngioma can arise anywhere along the Rathke pouch migratory path from the nasopharynx to the sella. Cases of extra-sellar craniopharyngioma masquerading as hypertrophied adenoid tissue in children have been documented (Cummings Otolaryngology).
- Skull base surgery — transsphenoidal endoscopic approaches are performed by ENT-trained skull base surgeons in many centres. The expanded endonasal approach to the sella and suprasellar space is a core ENT skull base procedure.
- Visual assessment — bitemporal field defects require collaboration with ophthalmology.
- CSF rhinorrhoea — a recognised complication of transsphenoidal resection requiring ENT management.
Sources: Cummings Otolaryngology Head and Neck Surgery; Robbins, Cotran & Kumar Pathologic Basis of Disease; Bradley and Daroff's Neurology in Clinical Practice; Grainger & Allison's Diagnostic Radiology; Goldman-Cecil Medicine; The Developing Human (Moore & Persaud); Adams and Victor's Principles of Neurology; PMID 37922728; PMID 37466685; PMID 40063185
This is a shared conversation. Sign in to Orris to start your own chat.