Pulmonary embolism definition etiology pathogenesis classification in details
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive content from multiple authoritative textbooks to construct a detailed answer. Let me compile it:
Pulmonary Embolism — Definition, Etiology, Pathogenesis & Classification
1. Definition
Pulmonary embolism (PE) is the obstruction of one or more pulmonary arteries by embolic material — most commonly a thrombus originating from the deep venous system of the lower extremities — leading to partial or complete occlusion of the pulmonary vasculature.
PE is the most serious clinical presentation of venous thromboembolism (VTE), a spectrum that also includes deep vein thrombosis (DVT). In 70–80% of cases, the source is leg DVT; fragmented thrombi are carried through progressively larger veins and the right side of the heart before lodging in the pulmonary arterial vasculature.
- Estimated incidence: 60–120 cases per 100,000 per year in the United States
- Causes 60,000–100,000 deaths per year in the U.S.
- VTE is the third most frequent cardiovascular disease globally, with overall annual incidence of 75–270 per 100,000 inhabitants
- 1-month mortality: 9–11%; 3-month mortality: up to 17%
- Approximately one-third of patients die within the first hours of presentation, often before diagnosis is confirmed
- ~20% die before or shortly after diagnosis
— Robbins, Cotran & Kumar Pathologic Basis of Disease; Fuster and Hurst's The Heart, 15th Edition
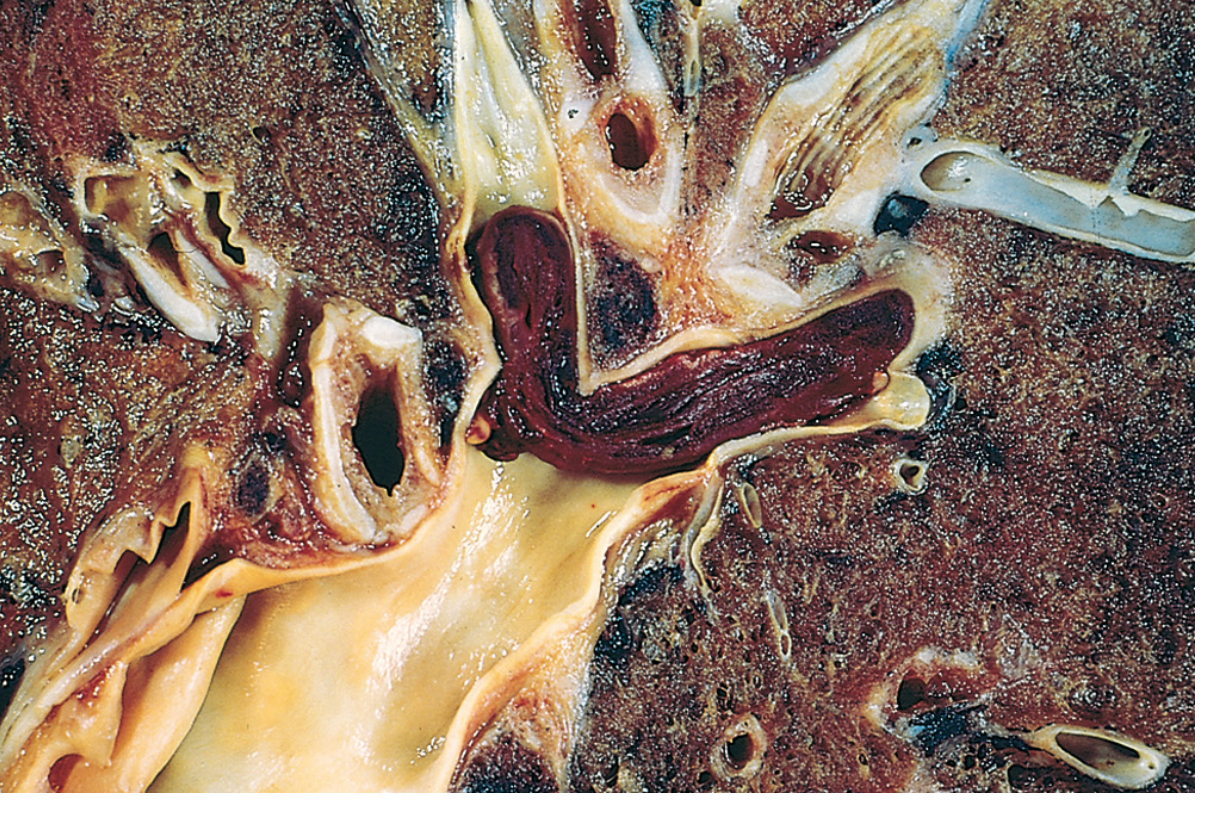
Embolus from a lower extremity deep venous thrombosis, lodged at a pulmonary artery branchpoint — Robbins Pathologic Basis of Disease
2. Etiology & Risk Factors
2.1 Virchow's Triad
In 1856, Rudolf Virchow described the three fundamental predisposing conditions for intravascular coagulation. All risk factors for VTE/PE map onto one or more of these three elements:
| Element | Mechanism |
|---|---|
| Venous stasis | Reduced flow → allows clotting proteins and cells to accumulate and bond; caused by immobility, CHF, venous obstruction, obesity |
| Endothelial injury | Trauma or surgery exposes subendothelial collagen → platelet adhesion, activation, secretion of prothrombotic mediators, and aggregation → fibrin formation |
| Hypercoagulability | Inherited or acquired imbalance favoring procoagulant state; clotting cascade produces thrombin which converts soluble fibrinogen → insoluble fibrin |
— Murray & Nadel's Textbook of Respiratory Medicine; Miller's Review of Orthopaedics, 9th ed.; Fuster and Hurst's The Heart, 15th ed.
2.2 Risk Factors
Hereditary (Inherited Thrombophilias)
| Factor | Mechanism |
|---|---|
| Factor V Leiden | Mutated Factor V not inactivated by activated protein C → prolonged clotting |
| Prothrombin G20210A mutation | Elevated prothrombin levels → more thrombin |
| Antithrombin III deficiency | Loss of key natural anticoagulant |
| Protein C deficiency | Failure to inactivate Factors Va and VIIIa |
| Protein S deficiency | Cofactor for protein C; loss of anticoagulant function |
| Plasminogen deficiency | Impaired fibrinolysis |
| Dysfibrinogenemia | Abnormal fibrinogen molecule |
| Elevated factors VIII, IX, XI | Increased procoagulant drive |
| Hyperhomocysteinemia | Endothelial damage + hypercoagulability |
Acquired Risk Factors
| Category | Specific Factors |
|---|---|
| Immobility / stasis | Prolonged bed rest, surgery, long-distance travel (>5,000 km or >8 hours), paralysis, stroke |
| Surgery & trauma | Major surgery (especially orthopedic, pelvic, spinal, neurosurgical); trauma — risk highest in first 2 postoperative weeks, may persist 2–3 months |
| Malignancy | Active cancer (lung, brain, pancreas, leukemia) — cancer itself is thrombogenic; chemotherapy adds risk |
| Hormonal | Oral contraceptives (especially high-estrogen), hormone replacement therapy, high-dose fertility injections, prostate cancer hormonal therapy |
| Pregnancy & puerperium | Increased coagulation factors (V, VIII, X, vWF), fall in protein S, uterine IVC compression, pelvic vein trauma at delivery; risk ~5× nonpregnant controls |
| Cardiovascular | Heart failure, MI, AF, central venous catheters, pacemakers/ICDs |
| Obesity | BMI ≥29 kg/m²; elevated fibrinogen, Factor VII, PAI-1; platelet activation |
| Antiphospholipid syndrome | Acquired thrombophilia; antibodies promote coagulation |
| Heparin-induced thrombocytopenia (HIT) | Paradoxical thrombosis |
| Other | Prior VTE (strongest independent predictor), inflammatory bowel disease, nephrotic syndrome, polycythemia vera, varicose veins, COVID-19 |
In the DVT FREE registry (5,451 patients with ultrasound-confirmed DVT), the most common comorbidities were: hypertension (50%), recent surgery (38%), immobility (34%), cancer (32%), and obesity (27%).
— Fuster and Hurst's The Heart, 15th ed.; Sabiston Textbook of Surgery; Murray & Nadel's Respiratory Medicine
3. Pathogenesis
3.1 Formation of the Source Thrombus (DVT)
Thrombi most commonly originate in valve pockets of the deep veins. The veins of the calf are the most common initial site. Thrombus formation begins with platelet adhesion to exposed subendothelial collagen → platelet activation and secretion of prothrombotic mediators → platelet aggregation → activation of the coagulation cascade → thrombin generation → fibrin deposition and clot consolidation.
Thrombus propagation: The thrombus may extend both retrogradely and proximally to fill the vessel entirely. Thrombi below the popliteal space carry very low PE risk; those above the popliteal (proximal DVT) carry significantly higher PE risk. If embolization does not occur, the thrombus undergoes resolution via recanalization, organization, or lysis.
3.2 Embolization
When part or all of a DVT detaches (typically from a proximal lower extremity vein), it travels through progressively larger veins → inferior vena cava → right atrium → right ventricle → pulmonary artery, where it lodges based on its size:
- Large emboli: Occlude the main pulmonary artery
- Saddle emboli: Straddle the pulmonary artery bifurcation
- Medium emboli: Lodge in segmental or subsegmental arteries
- Small emboli: Reach smaller, more peripheral branches
- Paradoxical embolism: Rare — venous embolus passes through a patent foramen ovale or septal defect into the systemic arterial circulation
Frequently there are multiple emboli, either sequential or simultaneous ("shower of emboli") from a single large mass.
3.3 Hemodynamic & Pulmonary Consequences
The mechanical obstruction of pulmonary arterial flow triggers a cascade of pathophysiological changes:
Mechanical obstruction
Newly formed thrombi are coated with fibrin and platelets, causing mechanical obstruction of pulmonary blood flow → increased pulmonary vascular resistance → acute pulmonary hypertension → acute right ventricular (RV) strain.
Right ventricular failure
The sudden increase in pulmonary vascular resistance imposes an acute pressure overload on the RV — an unprepared (thin-walled) chamber not conditioned for high-pressure work. This causes:
- RV dilation and hypokinesis
- Tricuspid regurgitation
- Interventricular septal shift (D-sign) → compromised LV filling
- Decreased cardiac output → systemic hypotension → coronary hypoperfusion → RV ischemia → further RV failure (a vicious cycle)
When emboli obstruct ≥60% of the pulmonary circulation: sudden death, acute cor pulmonale, or cardiovascular collapse.
Gas exchange abnormalities
-
Dead space: Obstructed segments are ventilated but not perfused → ↑ alveolar dead space → hypercapnia if ventilation cannot compensate
-
Hypoxemia (most patients): Paradoxically, most patients are hypocapnic (hyperventilating). Hypoxemia occurs through:
- Redistribution of perfusion to non-obstructed units → low V/Q areas in previously normally ventilated zones (overperfusion)
- Bronchoconstriction (mediated by serotonin, histamine) → ↓ ventilation in affected segments
- Shunt — develops later with atelectasis (loss of surfactant) and infarction (11–17% of cases)
- Low mixed venous PO₂ — depressed cardiac output increases O₂ extraction, further lowering arterial PO₂
- Intracardiac shunt — elevated RV pressure can open a patent foramen ovale
-
12% of PE patients in the Urokinase PE Trial had PaO₂ >80 mmHg, confirming hypoxemia is not universal
Pulmonary infarction
- The lung has dual blood supply (pulmonary + bronchial arteries), so infarction is not inevitable
- Occurs in ~10% of PE cases; more likely when bronchial circulation is also compromised (e.g., left-sided heart failure)
- Obstruction of medium-sized arteries → pulmonary hemorrhage but usually not infarction
- Obstruction of small end-arteriolar branches → hemorrhage or infarction
- Hampton's hump — classic wedge-shaped pleural-based opacity on CXR representing infarction
Neurohormonal/vasoactive mediators
Platelet activation within the thrombus releases serotonin, thromboxane A₂, and histamine → pulmonary vasoconstriction and bronchoconstriction → worsening V/Q mismatch.
Chronic sequelae
- Recurrent PE → progressive pulmonary hypertension and cor pulmonale
- Chronic thromboembolic pulmonary hypertension (CTEPH): Incomplete resolution → organized fibrous obstruction; occurs in ~1% of acute PE episodes
— Robbins, Cotran & Kumar Pathologic Basis of Disease; Murray & Nadel's Respiratory Medicine; Fuster and Hurst's The Heart; Sabiston Textbook of Surgery
4. Classification
4.1 By Etiology / Provoking Status
| Type | Definition |
|---|---|
| Provoked PE | Presence of a temporary/reversible risk factor within 3 months before diagnosis (surgery, trauma, immobilization, pregnancy) |
| Unprovoked (idiopathic) PE | No identifiable temporary risk factor; carries higher recurrence risk; may warrant long-term anticoagulation |
| Provoked by persistent factor | Inherited thrombophilia, active malignancy — affects duration of anticoagulation |
4.2 By Anatomical Location
| Type | Description |
|---|---|
| Saddle embolus | Straddles the main pulmonary artery bifurcation; typically causes massive PE |
| Central PE | Main pulmonary artery or lobar arteries |
| Segmental PE | Segmental arteries |
| Subsegmental PE | Peripheral small arteries; often clinically silent |
4.3 By Clinical Severity (ESC 2019 vs AHA 2011)
This is the most clinically relevant classification, based on hemodynamic status, RV dysfunction, myocardial injury biomarkers, and PESI score:
| ESC 2019 Term | AHA 2011 Term | Hemodynamic Status | PESI / sPESI | Evidence of RV Dysfunction | Treatment Strategy |
|---|---|---|---|---|---|
| High Risk | Massive | Unstable (sustained hypotension, shock, cardiac arrest) | High | Typically abnormal RV on imaging + elevated troponin, or both | Anticoagulation + advanced therapy (systemic thrombolysis, catheter-directed therapy, embolectomy, ECMO) |
| Intermediate-High Risk | Submassive | Stable (normotensive) | High | Abnormal RV on imaging AND elevated troponin | Anticoagulation; advanced therapy if clinical deterioration |
| Intermediate-Low Risk | — | Stable | High | Abnormal RV on imaging OR elevated troponin (but not both) | Anticoagulation |
| Low Risk | Low risk | Stable | Low | None | Anticoagulation; home therapy in select patients |
Key definitions:
- Hypotension/instability = sustained SBP <90 mmHg, or a drop ≥40 mmHg for ≥15 min, not explained by other causes
- RV dysfunction = RV dilation on echo or CT (RV:LV ratio >0.9), elevated BNP/NT-proBNP, or new right bundle branch block on ECG
- Myocardial injury = elevated troponin I or T
- PESI (Pulmonary Embolism Severity Index) incorporates age, sex, cancer, heart failure, chronic lung disease, HR, SBP, RR, temperature, mental status, O₂ saturation
— Braunwald's Heart Disease, 2-Volume Set; Fuster and Hurst's The Heart, 15th ed.; Miller's Anesthesia, 10th ed.
4.4 By Temporal Onset
| Type | Description |
|---|---|
| Acute PE | Sudden onset, no chronic adaptive changes |
| Subacute PE | Onset over days to weeks |
| Chronic PE / CTEPH | Recurrent or incompletely resolved emboli → organized fibrous vascular occlusion → chronic pulmonary hypertension; ~1% of acute PE episodes |
4.5 By Clinical Presentation (PIOPED Classification)
| Category | Features |
|---|---|
| Pleuritic pain/hemoptysis | Peripheral PE with pulmonary infarction; 56% have pleural effusion |
| Isolated dyspnea | Central PE without infarction; 26% have pleural effusion |
| Circulatory collapse | Massive PE; no pleural effusion; highest mortality |
5. Special Types of Pulmonary Embolism (Non-thrombotic)
Though most PE is thrombotic, emboli can also consist of:
| Type | Source/Mechanism |
|---|---|
| Fat embolism | Long bone fractures, orthopedic surgery |
| Air embolism | IV catheter placement, surgery, trauma |
| Amniotic fluid embolism | Obstetric complication during labor/delivery |
| Septic embolism | Infected thrombi (right-sided endocarditis, IV drug use) |
| Tumor embolism | Malignant cells entering venous circulation |
| Foreign body embolism | Catheter fragments, talc (IV drug users) |
Summary
| Aspect | Key Points |
|---|---|
| Definition | Obstruction of pulmonary arterial vasculature, most often by thrombus from lower extremity DVT |
| Etiology | Virchow's triad: stasis + endothelial injury + hypercoagulability; both acquired and hereditary risk factors |
| Pathogenesis | DVT → embolization → mechanical obstruction → acute RV pressure overload → RV failure → ↓ cardiac output; V/Q mismatch → hypoxemia |
| Classification | By severity (ESC: high/intermediate-high/intermediate-low/low risk); by anatomy (saddle, central, segmental); by acuity (acute, subacute, chronic/CTEPH) |
This is a shared conversation. Sign in to Orris to start your own chat.