Myocardial infarction
Reading File
Finding Sources
Searching PubMed
"myocardial infarction"[MeSH Terms] AND "management"[MeSH Terms]
Searching Images
myocardial infarction pathology ECG diagram
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Searching PubMed
"myocardial infarction"[MeSH Terms] AND "percutaneous coronary intervention"[MeSH Terms]
Reading File
Finding Sources
Loading Image
Excellent content gathered. I have enough for a comprehensive answer. Let me compile the complete response now.
Myocardial Infarction (MI)
Definition
MI — commonly called a "heart attack" — is the death of cardiac muscle due to prolonged ischemia. In the United States, approximately 800,000 individuals experience an MI each year (nearly one every 40 seconds), causing ~400,000 deaths annually. — Robbins, Cotran & Kumar Pathologic Basis of Disease
Epidemiology & Risk Factors
- 10% occur in people <40 years; 45% in people <65 years
- Frequency rises progressively with age and with increasing atherosclerotic burden
- Through middle age, male sex confers higher relative risk; females are generally protected during reproductive years
- Post-menopausal decline in estrogen is associated with worsening CAD — IHD becomes the most common cause of death in older women
- Hormone replacement therapy has not been shown to be protective and may be detrimental (pro-thrombotic effect)
Pathogenesis
The Classic Sequence (90% of cases)
- An atheromatous plaque is eroded or disrupted by endothelial injury, intraplaque hemorrhage, or mechanical forces — exposing subendothelial collagen and necrotic plaque contents to blood
- Platelets adhere and activate, releasing thromboxane A₂, ADP, and serotonin → further platelet aggregation and vasospasm
- Coagulation is activated via tissue factor → growing thrombus
- Within minutes, the thrombus can completely occlude the coronary artery lumen
When angiography is performed within 4 hours of MI onset, coronary thrombosis is found in ~90% of cases. By 12–24 hours (without intervention), this falls to 60%, as some occlusions clear spontaneously.
Non-Atherosclerotic Causes (~10% of cases)
- Vasospasm (with or without atherosclerosis) — e.g., cocaine, ephedrine
- Embolism — from LA thrombus in AF, infective endocarditis vegetations, prosthetic material, or paradoxical emboli through a PFO
- Uncommon: vasculitis, sickle cell disease, amyloid deposition, aortic dissection, marked ventricular hypertrophy, shock
Myocardial Response to Ischemia
The temporal progression of ischemic injury:
| Feature | Time |
|---|---|
| Onset of ATP depletion | Seconds |
| Loss of contractility | <2 minutes |
| ATP reduced to 50% of normal | 10 minutes |
| ATP reduced to 10% of normal | 40 minutes |
| Irreversible cell injury (necrosis) | 20–40 minutes |
| Microvascular injury | >1 hour |
— Robbins, Cotran & Kumar Pathologic Basis of Disease
Only severe ischemia (blood flow ≤10% of normal) lasting 20–40 minutes or longer leads to irreversible necrosis. An infarct achieves its full extent by 6–12 hours. This provides the rationale for rapid diagnosis and reperfusion.
Progression of Necrosis
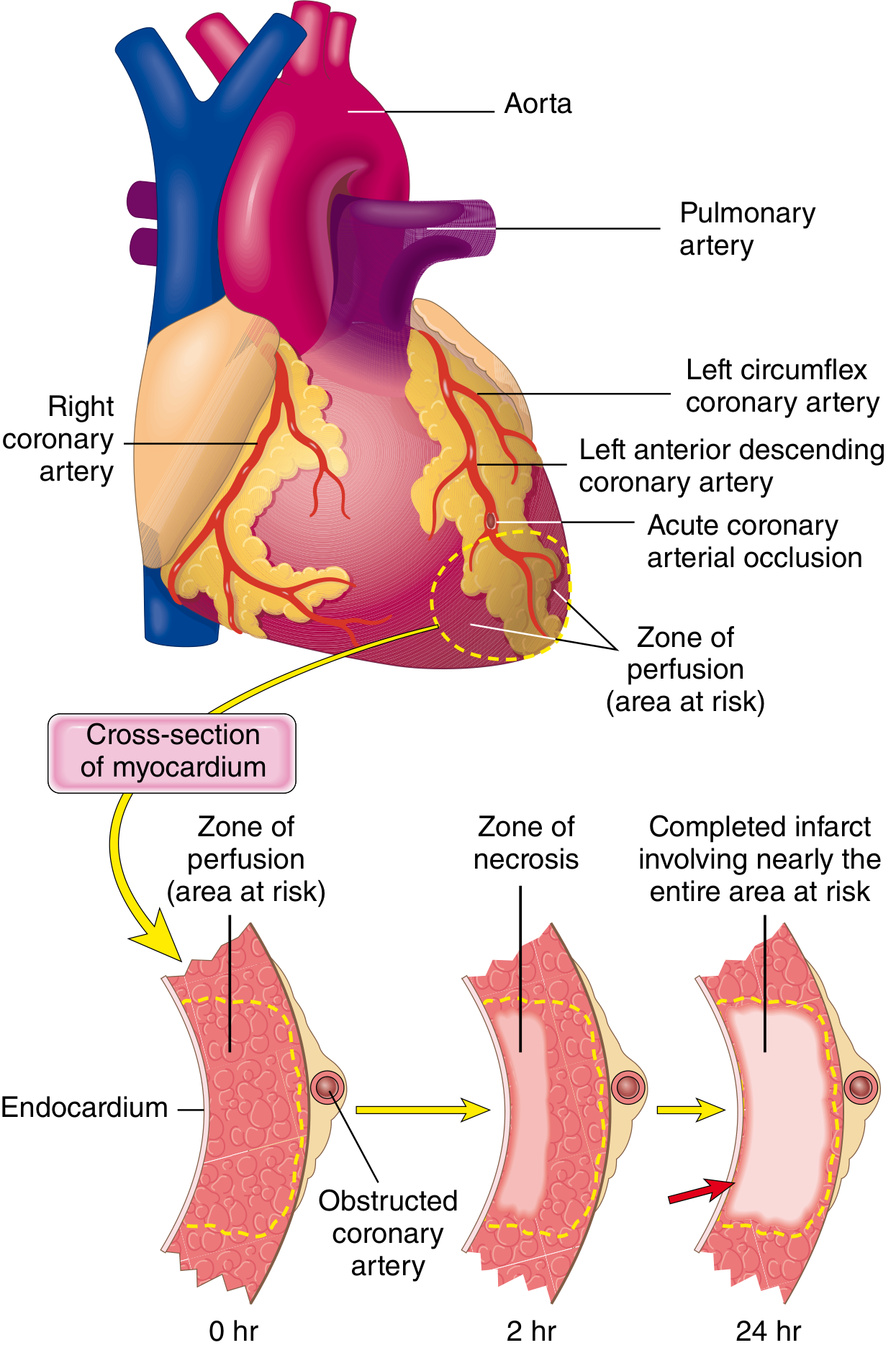
Progression of myocardial necrosis after coronary artery occlusion. Necrosis begins in the subendocardial zone and progresses as a wavefront toward the epicardium over 0–24 hours. A narrow rim immediately beneath the endocardium is spared because it receives oxygen by diffusion from the ventricular cavity. — Robbins, Cotran & Kumar
Patterns of Infarction
| Pattern | Mechanism | Correlate |
|---|---|---|
| Transmural infarct | Complete epicardial vessel occlusion | Atherothrombosis; typically causes STEMI |
| Subendocardial (nontransmural) | Partial/transient occlusion or global hypoperfusion | Often circumferential; correlates with NSTEMI |
| Multifocal microinfarction | Small intramural vessel pathology | Microembolization, vasculitis, catecholamine excess |
Coronary Artery Territories
- LAD: apex, anterior LV wall, anterior 2/3 of ventricular septum
- RCA (right-dominant, ~80% of people): entire RV free wall, posterobasal LV wall, posterior 1/3 of septum
- LCX: lateral LV wall
ECG Changes
Three electrical mechanisms underlie ST changes in acute MI (Ganong's Review of Medical Physiology):
| Defect in Infarcted Cells | Current Flow | ECG Change |
|---|---|---|
| Rapid repolarization | Out of infarct | ST elevation |
| Decreased resting membrane potential | Into infarct | TQ depression → recorded as ST elevation |
| Delayed depolarization | Out of infarct | ST elevation |
The hallmark of acute MI is ST-segment elevation in leads overlying the infarct, with reciprocal ST depression in opposite leads.
- After days to weeks: ST normalizes; dead muscle becomes electrically silent → Q waves appear (or R-wave progression fails in anterior MI)
- Non-Q-wave infarcts tend to be less severe but carry a high risk of re-infarction
STEMI ECG Examples
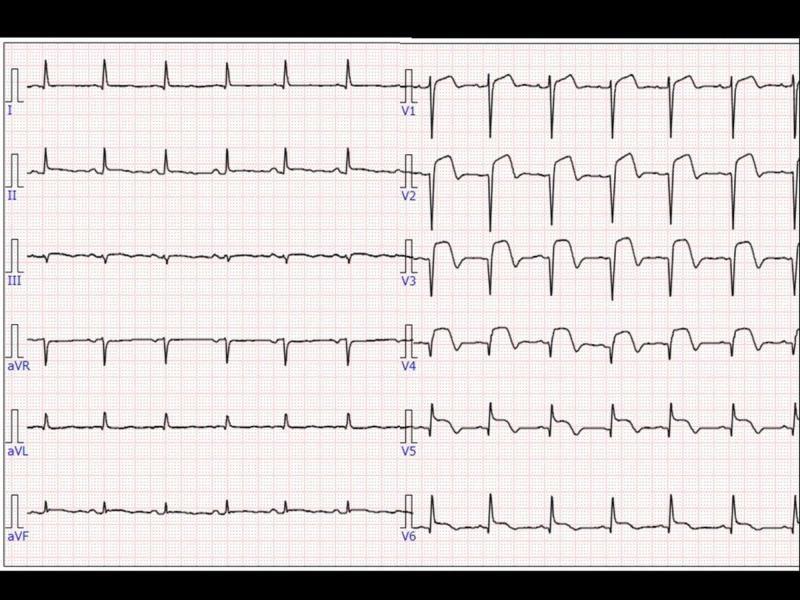
Anterior STEMI: tombstone ST elevation across V1–V6, indicating extensive anterior wall MI.
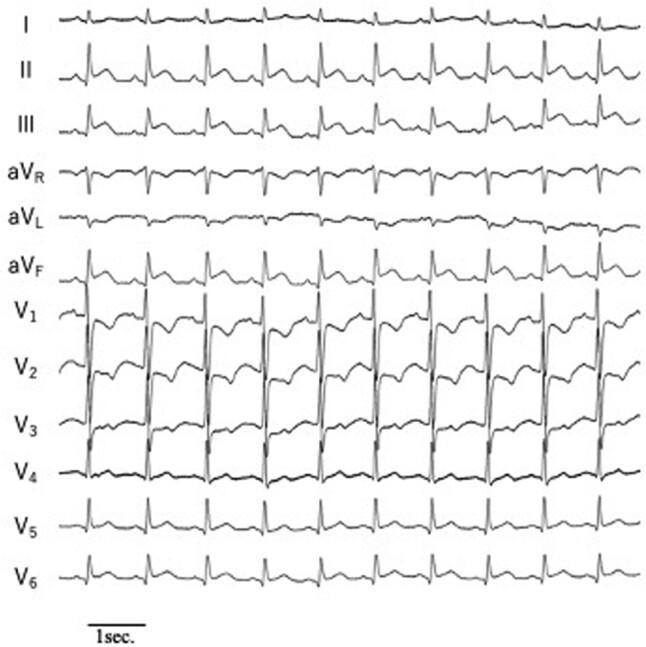
Inferior STEMI: ST elevation in II, III, aVF with reciprocal depression in I and aVL.
Morphological Stages of MI
| Time After Occlusion | Gross Appearance | Microscopic Appearance |
|---|---|---|
| 0–30 min | None | None (reversible) |
| 30 min–4 hr | None (may see pallor) | Early waviness of fibers at border |
| 4–12 hr | Pallor/mottling | Coagulation necrosis begins; edema, hemorrhage |
| 12–24 hr | Tan-yellow pallor | Continued coagulation necrosis; neutrophil infiltration starts |
| 1–3 days | Pallor with yellow center | Neutrophil infiltration prominent |
| 3–7 days | Hyperemic border | Macrophage infiltration; early granulation tissue |
| 1–3 weeks | Soft, yellow-white with firm red-tan rim | Granulation tissue with neovascularization |
| >2 months | White scar | Dense collagen scar |
Cardiac Biomarkers
The escape of intracellular proteins through disrupted sarcolemmal membranes into the circulation allows detection:
- Troponin I and T — most sensitive and specific; begin rising at 2–4 hours, peak at 24–48 hours, remain elevated for 7–10 days (cTnI) or 10–14 days (cTnT). Gold standard.
- CK-MB — rises at 4–8 hours, peaks at 24 hours, normalizes by 48–72 hours. Useful for detecting re-infarction (returns to baseline faster).
- Myoglobin — earliest to rise (~1–2 hr), but not cardiac-specific.
Classification: STEMI vs. NSTEMI
| Feature | STEMI | NSTEMI |
|---|---|---|
| ECG | ST elevation | ST depression or T-wave changes |
| Pathology | Complete transmural occlusion | Partial/transient or subendocardial |
| Troponin | Elevated | Elevated |
| Urgency | Emergency PCI within 90 min (door-to-balloon) | Risk-stratified; early invasive within 24–72 hr |
Management
Immediate ("Time is Muscle")
- Aspirin 162–325 mg (chewed) immediately
- P2Y12 inhibitor (clopidogrel, ticagrelor, or prasugrel) — dual antiplatelet therapy (DAPT)
- Anticoagulation (heparin, bivalirudin, or enoxaparin)
- Oxygen if SpO₂ <90%
- Nitrates for ongoing chest pain (avoid if RV infarct or recent PDE5 inhibitor use)
- Beta-blockers — early oral (unless contraindicated: HR <60, hypotension, severe HF, heart block)
Reperfusion — the cornerstone of treatment
STEMI:
- Primary PCI is the preferred strategy — target door-to-balloon time ≤90 minutes (≤120 min if transfer required)
- Fibrinolysis (e.g., alteplase, tenecteplase) if PCI unavailable within 120 minutes of first medical contact; most effective within 3 hours of symptom onset
NSTEMI/UA:
- Early invasive strategy (coronary angiography + PCI) within 24 hours for high-risk patients (GRACE score, elevated troponin, TIMI risk ≥3)
- Ischemia-guided (conservative) strategy for low-risk patients
Recent Evidence (2024–2025)
A network meta-analysis in JACC (2024) confirmed the superiority of complete revascularization over culprit-only PCI in MI with multivessel disease (PMID: 38986670). A subsequent JACC network meta-analysis (2025) specifically in STEMI with multivessel disease supports staged complete revascularization as the optimal strategy (PMID: 39779054).
Long-Term Medical Therapy
| Drug Class | Rationale |
|---|---|
| Dual antiplatelet (aspirin + P2Y12) | Prevent stent thrombosis and recurrent MI |
| High-intensity statin | Plaque stabilization, LDL reduction |
| ACE inhibitor / ARB | Reduce LV remodeling, prevent HF |
| Beta-blocker | Reduce mortality, especially if EF reduced |
| Aldosterone antagonist (eplerenone) | If EF ≤40% with HF or diabetes |
Complications
| Category | Complication | Time Frame |
|---|---|---|
| Electrical | Ventricular fibrillation, VT, AV block (especially inferior MI) | Hours–days |
| Mechanical | Free wall rupture, VSD, papillary muscle rupture (acute MR) | 3–5 days |
| Ischemic | Extension of infarct, re-infarction | Days–weeks |
| Inflammatory | Pericarditis (Dressler syndrome) | Days; 1–8 weeks |
| Structural | LV aneurysm, mural thrombus | Weeks–months |
| Pump failure | Cardiogenic shock, acute HF | Hours–days |
Prognosis
Mortality is highest in the first hour (largely arrhythmic — VF). Overall in-hospital mortality with modern reperfusion therapy is <5–7% for STEMI. Predictors of poor prognosis include advanced age, large infarct size, anterior location, reduced EF, Killip class, and time to reperfusion.
Sources: Robbins, Cotran & Kumar Pathologic Basis of Disease; Ganong's Review of Medical Physiology (26th ed.); Robbins & Kumar Basic Pathology; Braunwald's Heart Disease; Goldman-Cecil Medicine. Recent evidence: JACC PMID 38986670 (2024), JACC PMID 39779054 (2025).
This is a shared conversation. Sign in to Orris to start your own chat.