Anesthesia for Video-Assisted Thoracoscopic Surgery (VATS)
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Searching PubMed
VATS anesthesia one-lung ventilation
Loading Image
Loading Image
I now have all the material needed for a comprehensive, well-organized response. Let me compose it.
Anesthesia for Video-Assisted Thoracoscopic Surgery (VATS)
Sources: Morgan and Mikhail's Clinical Anesthesiology, 7e; Barash Clinical Anesthesia, 9e
Overview
VATS is now the preferred approach for most lung resections (lobectomy, segmentectomy, wedge), pleurodesis, pleural biopsies, mediastinal procedures, and esophageal surgery. Procedures are performed through several small incisions in the chest wall with the patient in the lateral decubitus position. Anesthetic management parallels that of open thoracotomy with one critical distinction: one-lung ventilation (OLV) is required (not merely desirable) for virtually all VATS procedures to provide the collapsed, still operative lung the surgeon needs for visualization and safe instrumentation. Increasingly, "tubeless" VATS techniques (NIVATS) are also performed.
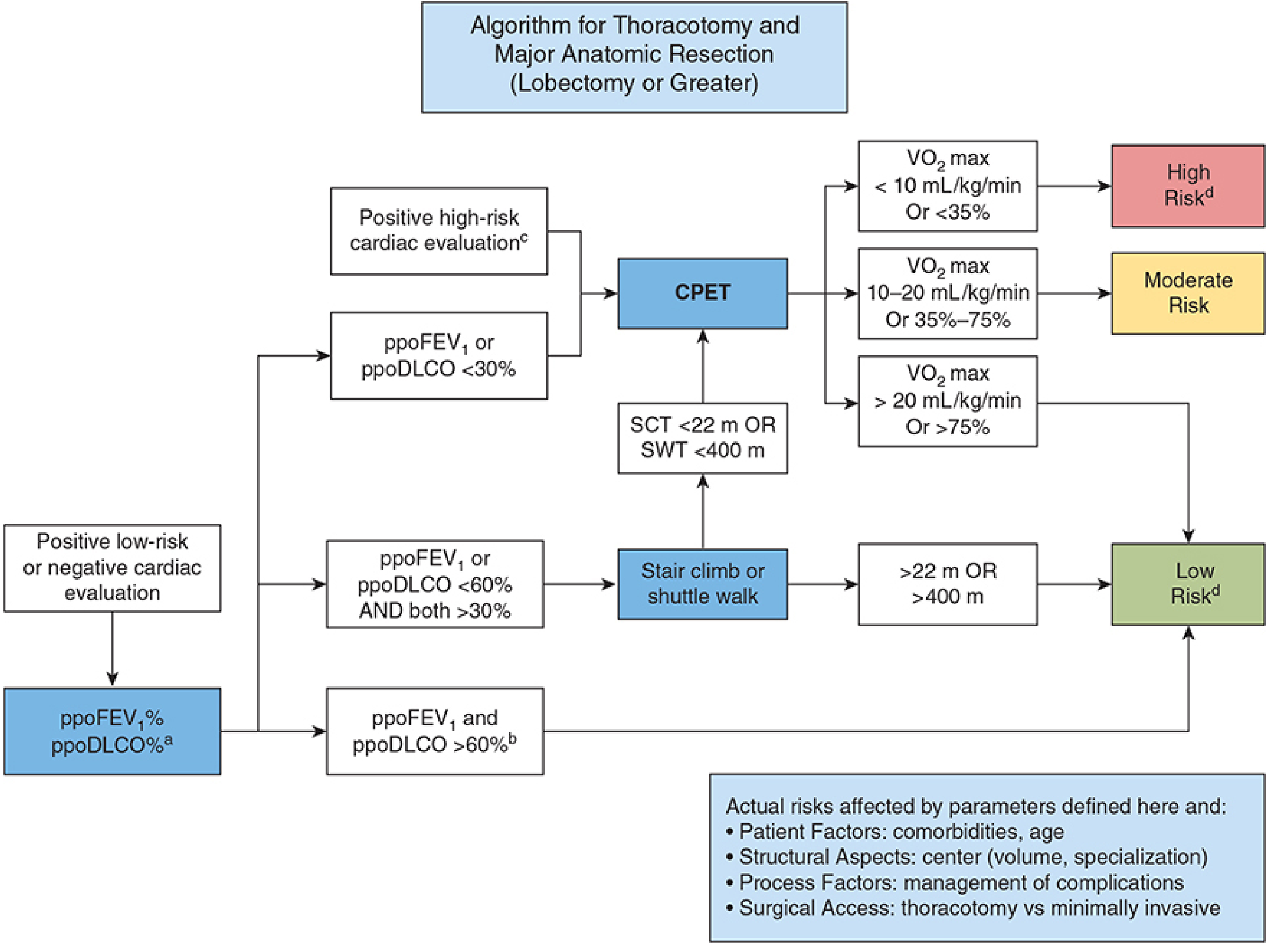
1. Preoperative Assessment
Patients undergoing VATS for lung resection require thorough cardiopulmonary evaluation to predict postoperative pulmonary reserve.
Pulmonary Function
The predicted postoperative (PPO) values of FEV₁ and DLCO are the key metrics:
- Low risk (proceed directly): ppoFEV₁ and ppoDLCO >60% predicted
- Moderate risk (exercise testing needed): ppoFEV₁ or ppoDLCO 30–60% → stair-climb or shuttle walk test → CPET
- High risk: VO₂ max <10 mL/kg/min or ppoFEV₁/ppoDLCO <30% → mortality after major resection may exceed 10%; consider minimally invasive alternatives or non-surgical options
Other Considerations
- Paraneoplastic syndromes: Lambert–Eaton syndrome (proximal myopathy, augmented with repetition—important for NMB dosing), SIADH, Cushing syndrome, hypercalcemia
- Pancoast tumors: involvement of C7–T2 nerve roots; may affect regional technique options
- Concurrent therapy: prior chemotherapy (bleomycin → pulmonary toxicity; adriamycin → cardiomyopathy) and radiation must be assessed
2. One-Lung Ventilation (OLV)
Physiological Consequences
Intentional collapse of the operative (nondependent) lung creates a large right-to-left intrapulmonary shunt (20–30%). The collapsed lung continues to be perfused but is no longer ventilated, widening the A–a O₂ gradient and causing hypoxemia.
Hypoxic pulmonary vasoconstriction (HPV) is the primary compensatory mechanism — it reduces blood flow to the non-ventilated lung and attenuates shunting. Factors that inhibit HPV (worsening shunt) include:
| HPV Inhibitors | Effect on Shunt |
|---|---|
| Inhalation anesthetics | ↑ shunt |
| Pulmonary hypertension | ↑ shunt |
| Vasodilators (NTG, nitroprusside, nitric oxide) | ↑ shunt |
| Phosphodiesterase inhibitors (milrinone) | ↑ shunt |
| β-adrenergic agonists | ↑ shunt |
| Calcium channel blockers | ↑ shunt |
| Hypocapnia/alkalosis, hypothermia | ↑ shunt |
| High cardiac output / high mixed venous PO₂ | ↑ shunt |
Factors that decrease blood flow to the ventilated (dependent) lung also worsen shunting indirectly:
- High mean airway pressures (excessive PEEP, hyperventilation)
- Low FiO₂ in the ventilated lung (induces HPV there)
- Vasoconstrictors with greater effect on normoxic vessels
- Intrinsic PEEP from inadequate expiratory time
CO₂ elimination is usually preserved as long as minute ventilation is maintained; arterial CO₂ tension is typically not significantly altered.
Lung-Protective Ventilation Strategy During OLV
Both the ventilated and non-ventilated lungs are at risk for injury:
- Dependent lung: hyperfusion + ventilator-induced trauma from large tidal volumes
- Non-dependent lung: surgical trauma + ischemia-reperfusion injury
Current recommendations:
- Tidal volume: 4–5 mL/kg predicted body weight (NOT the same TV used during two-lung ventilation)
- FiO₂: 50–80% (avoid 100% O₂ — evidence of oxygen toxicity has accumulated)
- Plateau pressure: <25 cm H₂O; peak airway pressure: <35 cm H₂O
- Pressure-controlled ventilation preferred — limits peak/plateau pressures, more homogeneous tidal volume distribution, reduced dead-space
- Apply PEEP to the dependent lung to prevent atelectasis; use recruitment maneuvers
- Permissive hypercapnia is acceptable in rare patients with elevated CO₂ despite adequate saturation
3. Lung Isolation Techniques
Option 1: Double-Lumen Bronchial Tube (DLT) — Most Common
DLTs provide two separate lumens for independent ventilation of each lung and are the most commonly employed technique.
Key features:
- Longer endobronchial lumen enters a main bronchus; shorter tracheal lumen terminates in lower trachea
- Both tracheal and endobronchial cuffs
- Available in sizes: 35F, 37F, 39F, 41F
- Left-sided DLTs preferred due to longer length of left main bronchus (longer "safe zone" for placement)
- Right-sided DLTs require a proximal portal for right upper lobe ventilation
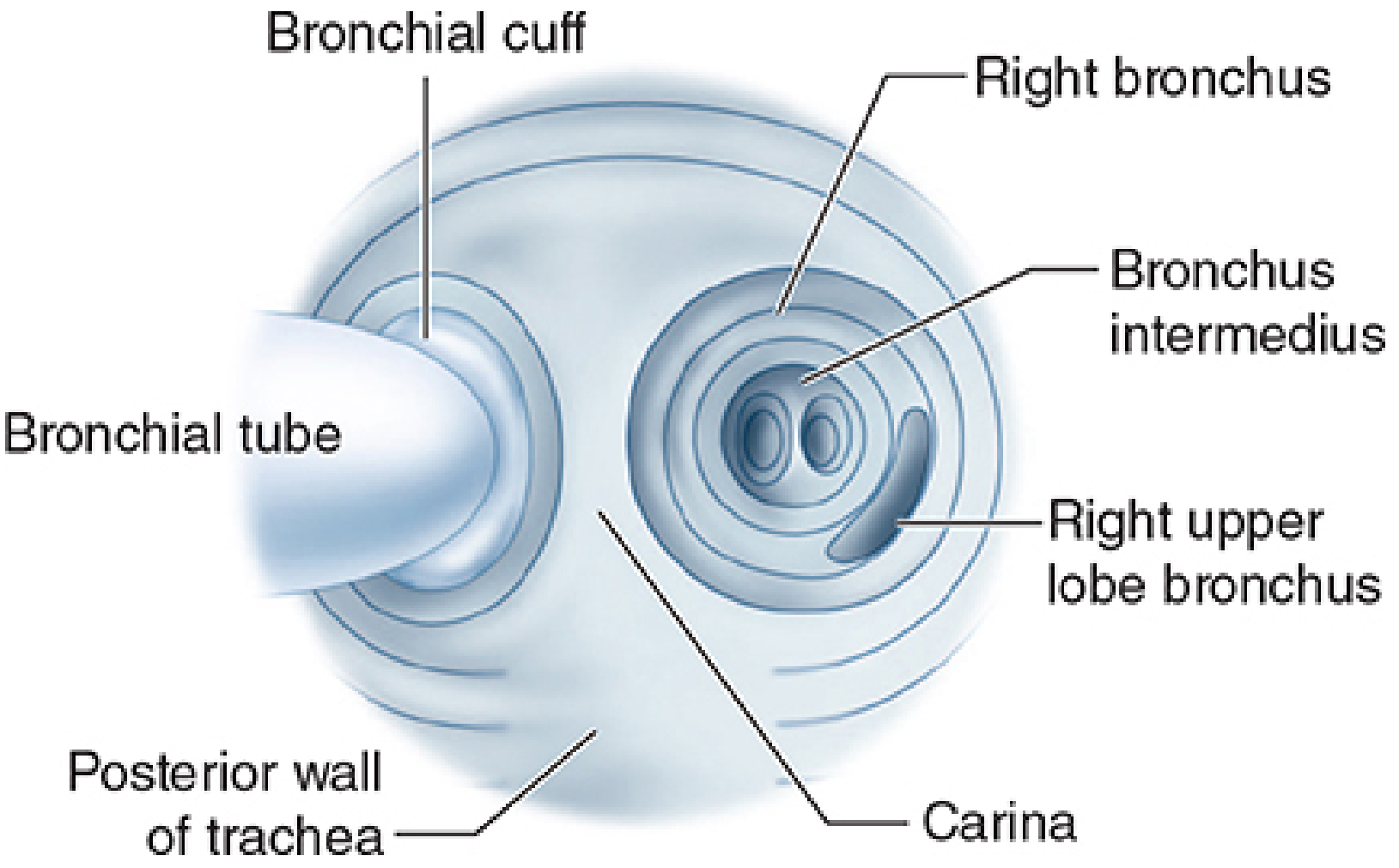
Placement and confirmation:
- After intubation, confirm position with fiberoptic bronchoscopy — the gold standard
- Reconfirm position after lateral positioning, as the tube may migrate relative to the carina when the patient is turned
- Malpositioning → failure of operative lung to collapse, poor compliance, low exhaled tidal volume
Complications:
- Hypoxemia (tube malplacement, occlusion, excessive venous admixture)
- Traumatic laryngitis
- Tracheobronchial rupture (traumatic placement or endobronchial cuff overinflation)
- Inadvertent suturing or stapling of the tube to a bronchus (detected as inability to remove tube at extubation)
Option 2: Single-Lumen Tube + Bronchial Blocker
A bronchial blocker (7F or 9F, with 1.4-mm inner lumen) is placed through or alongside a conventional ETT and positioned under fiberoptic guidance.
Advantages: No need for tube exchange if postoperative ventilation is required; preferred for difficult airways, ICU patients, prior tracheostomies
Disadvantages:
- Slow/incomplete lung collapse (small inner lumen)
- More prone to dislodgement than DLT
- Inefficient suctioning of secretions
Option 3: Endobronchial Intubation
A conventional single-lumen ETT is advanced into a mainstem bronchus — simple but crude, with no independent access to the collapsed lung.
Option 4: Tubeless VATS (NIVATS)
Thoracoscopic surgeries are increasingly performed under paravertebral blocks or thoracic epidural anesthesia with sedation (propofol or dexmedetomidine). Introduction of the thoracoscope creates a spontaneous pneumothorax → lung collapse. Studies suggest NIVATS may reduce surgical morbidity and shorten hospital stay. Close monitoring is essential and some patients will not tolerate the approach.
4. Intraoperative Management
Positioning
Lateral decubitus position — the operative (nondependent) lung is uppermost and collapsed; the dependent lung is ventilated.
Monitoring
Standard ASA monitoring plus arterial line for continuous blood pressure and blood gas sampling. End-tidal CO₂ is useful as a trend monitor but may not be accurate due to increased dead space — there is an unpredictable gradient between arterial and end-tidal CO₂. Periodic ABG analysis is essential.
Managing Intraoperative Hypoxemia
When hypoxemia develops during OLV, a stepwise approach is used:
- Confirm tube position — re-examine with fiberoptic bronchoscopy through the tracheal lumen; suction both lumens
- Increase FiO₂ to 1.0
- Recruitment maneuvers on the dependent ventilated lung
- PEEP to the dependent lung (adequate but not excessive — high PEEP increases mean airway pressure and diverts flow to the collapsed lung)
- CPAP or blow-by oxygen to the operative lung — reduces shunting but partially inflates the lung; in VATS, this impairs surgical visualization — use cautiously and communicate with surgeon
- Revert to two-lung ventilation for severe hypoxemia; during pneumonectomy, a pulmonary artery clamp can also eliminate shunt
- Suspect ipsilateral pneumothorax in COPD patients with sudden severe hypoxemia — requires aborting surgery, reexpanding the operative lung, and inserting a contralateral chest tube
Lung Re-expansion
At end of procedure, the operative lung is inflated gradually, targeting peak inspiratory pressure <30 cm H₂O to prevent disruption of the staple/suture line. Clamping the dependent lumen during reinflation helps limit overdistension.
5. Postoperative Management
Extubation
Most patients are extubated shortly after VATS to reduce the risk of pulmonary barotrauma ("blowout" of the bronchial suture line). When postoperative mechanical ventilation is required, the DLT must be exchanged for a standard single-lumen ETT (using a tube exchanger/catheter guide, especially if initial laryngoscopy was difficult).
Postoperative Care
- Observation in PACU, then monitored unit or ICU at least overnight
- Semi-upright positioning (>30°)
- Supplemental oxygen to maintain adequate saturation
- Incentive spirometry
- ECG and hemodynamic monitoring
- Postoperative chest X-ray — confirm thoracostomy tube position, central line position, bilateral lung expansion
- Watch for: atelectasis/splinting, gravity-dependent transudation into dependent lung, reexpansion edema of collapsed lung
Postoperative Complications
- Hemorrhage (~3% of thoracotomies; up to 20% mortality): chest tube drainage >200 mL/h, hypotension, tachycardia, falling hematocrit
- Supraventricular tachyarrhythmias: common; require immediate treatment
- Bronchopleural fistula: sudden large air leak; early (<72 h) = inadequate stump closure; late = suture line necrosis
- Lobar torsion: remaining lung occludes pulmonary vein → hemoptysis/infarction
- Cardiac herniation through pericardial defect (after pneumonectomy): herniation into right hemithorax → severe hypotension + elevated CVP; left hemithorax → myocardial compression/infarction
- Nerve injuries: phrenic (elevated hemidiaphragm, ventilator weaning difficulty), recurrent laryngeal, vagus, brachial plexus
6. Postoperative Analgesia
Adequate analgesia is critical — inadequate pain control leads to splinting, poor respiratory effort, inability to clear secretions, atelectasis, shunting, and hypoxemia.
| Modality | Notes |
|---|---|
| Thoracic epidural (most effective) | Continuous infusion: opioid (fentanyl, morphine, hydromorphone) + local anesthetic (bupivacaine/ropivacaine); requires 24/7 acute pain team coverage |
| Paravertebral block | Excellent for VATS; good alternative to epidural with fewer hemodynamic effects |
| Erector spinae plane block | Increasingly used; systematic reviews show benefit for VATS |
| Intercostal nerve blocks | Useful but limited duration; must be combined with other modalities |
| Serratus anterior plane block | Meta-analysis supports analgesic effectiveness for VATS |
| Wound infiltration | Liposomal bupivacaine or catheter infusion at closure; reduces parenteral opioid requirement |
| Multimodal systemic analgesia | Oral/IV acetaminophen, NSAIDs, gabapentinoids, low-dose ketamine, IV lidocaine infusion |
Summary Table
| Phase | Key Considerations |
|---|---|
| Preoperative | ppoFEV₁, ppoDLCO, CPET; paraneoplastic syndromes; prior chemo/radiation |
| Lung isolation | DLT (preferred), bronchial blocker, or tubeless (NIVATS) |
| OLV physiology | 20–30% shunt; HPV compensation; inhibited by inhalation agents, vasodilators |
| Ventilation strategy | TV 4–5 mL/kg, PCV, PEEP, FiO₂ 50–80%, plateau <25 cm H₂O |
| Hypoxemia management | Stepwise: confirm position → FiO₂ 1.0 → PEEP → recruitment → CPAP to operative lung → 2-lung VT |
| Extubation | Immediate post-op preferred; DLT exchange if ventilation needed |
| Analgesia | Thoracic epidural or PVB as cornerstone; multimodal regimen |
— Morgan and Mikhail's Clinical Anesthesiology, 7e, pp. 1024–1059
This is a shared conversation. Sign in to Orris to start your own chat.