consequences of cell injury
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have everything needed to write a comprehensive answer. Here is the full synthesis:
Consequences of Cell Injury
All injurious stimuli produce a stereotypic sequence of cellular responses that range from fully reversible functional disturbances to irreversible death. The outcome depends on the nature, duration, and severity of the injury.
Temporal Sequence of Changes
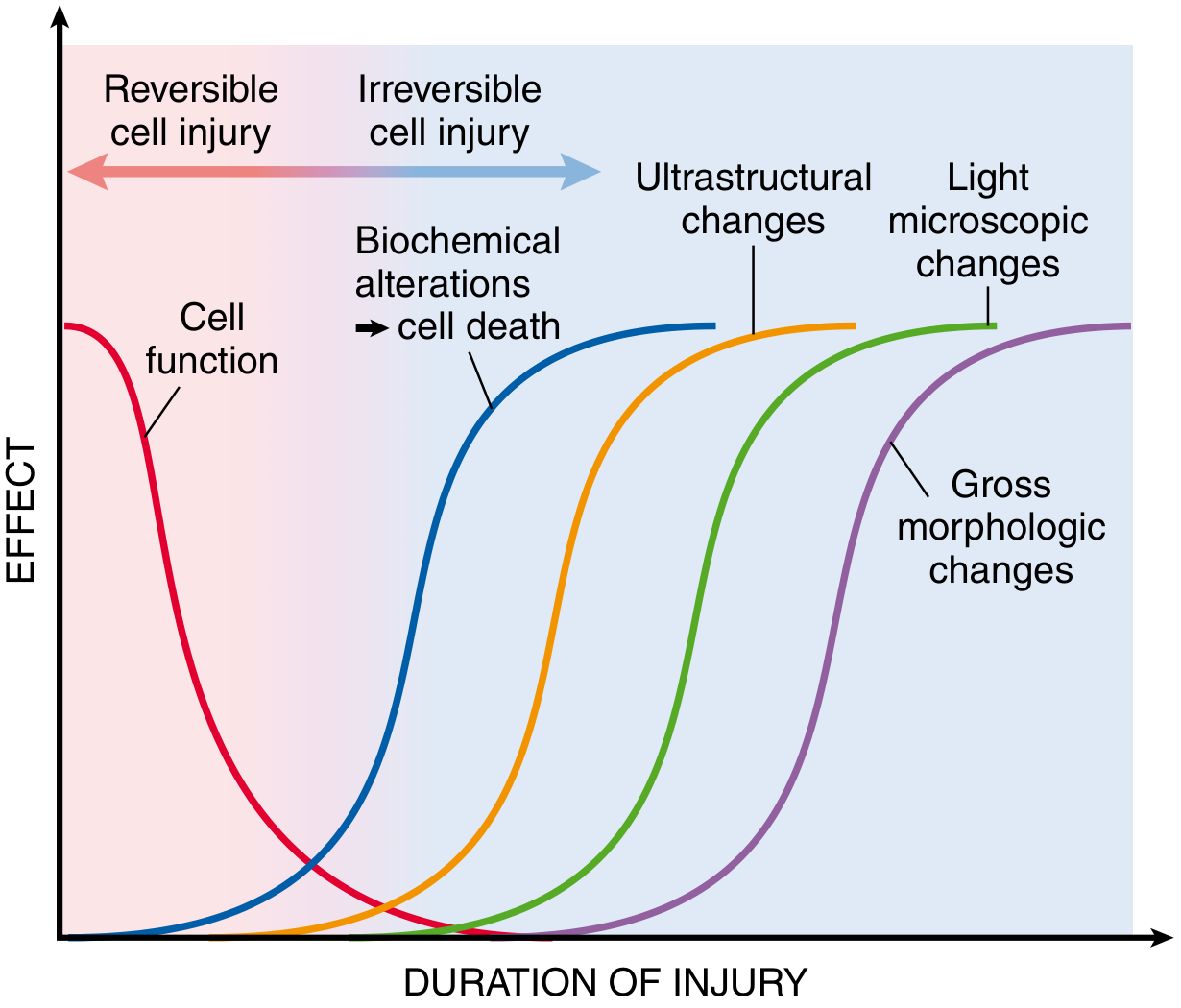
Fig. 2.6 — Robbins & Cotran Pathologic Basis of Disease
Key principle: cell function fails before morphology changes. The sequence from injury onset:
- Functional loss (seconds–minutes)
- Biochemical alterations (minutes–hours) — detectable by biochemical assays
- Ultrastructural changes — detectable only by electron microscopy
- Light microscopic changes — hours to days
- Gross morphologic changes — latest to appear
1. Reversible Cell Injury
If the injurious stimulus is removed in time, injured cells can recover to normalcy. Two consistent hallmarks:
Cellular Swelling (Hydropic Change / Vacuolar Degeneration)
- The earliest and most universal manifestation of cell injury
- Caused by failure of the ATP-dependent Na⁺/K⁺ plasma membrane pump → Na⁺ accumulates intracellularly → osmotic water influx
- ATP depletion is caused by: hypoxia (↓ oxidative phosphorylation), mitochondrial damage (toxins/radiation)
- Gross: pallor, increased turgor, increased organ weight
- Light microscopy: small clear cytoplasmic vacuoles (distended pinched-off ER segments); cytoplasm becomes eosinophilic (loss of RNA)
- Electron microscopy reveals:
- Plasma membrane blebs, blunting, loss of microvilli
- Mitochondrial swelling with small amorphous densities
- "Myelin figures" — phospholipid whorls from damaged membranes
- ER dilation with detachment of ribosomes/polysomes
- Nuclear chromatin clumping
Fatty Change (Steatosis)
- Occurs in organs active in lipid metabolism (e.g., liver, heart)
- Toxic injury disrupts lipid metabolic pathways → accumulation of triglyceride-filled lipid vacuoles
2. Irreversible Cell Injury → Cell Death
Persistent or excessive injury crosses the "point of no return" — a threshold beyond which mitochondrial and membrane damage is irreparable. Three hallmarks mark irreversibility:
- Inability to restore mitochondrial function (oxidative phosphorylation/ATP generation)
- Altered structure and loss of function of plasma and intracellular membranes
- Loss of structural integrity of DNA and chromatin
Cell death occurs by two principal mechanisms:
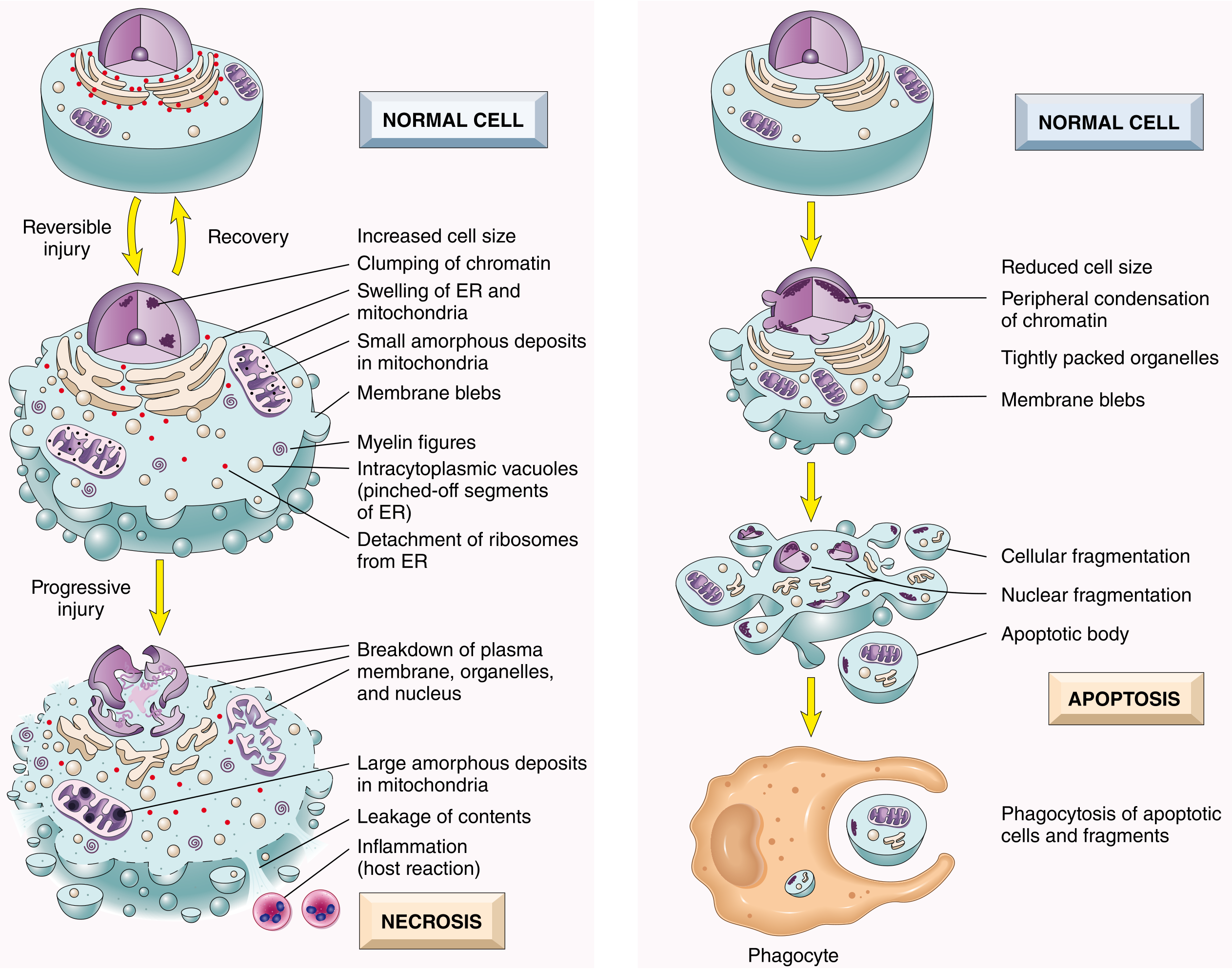
Fig. 2.7 — Robbins & Cotran Pathologic Basis of Disease
A. Necrosis
Historically considered "accidental" cell death following severe injury. Characterized by:
| Feature | Finding |
|---|---|
| Cell size | Enlarged (swelling) |
| Nucleus | Pyknosis (condensation) → Karyorrhexis (fragmentation) → Karyolysis (dissolution) |
| Plasma membrane | Disrupted |
| Cellular contents | Leak out (enzymatic digestion) |
| Inflammation | Frequent (host reaction to leaked contents) |
| Role | Pathologic — culmination of irreversible injury |
Patterns of Tissue Necrosis
| Type | Description | Example |
|---|---|---|
| Coagulative | Architecture preserved; cells "ghost outlines" (denatured proteins resist digestion) | Myocardial infarction |
| Liquefactive | Complete dissolution; pus formation | Brain infarct, abscess |
| Caseous | Cheesy, amorphous; combination of coagulative + liquefactive | Tuberculosis |
| Fat necrosis | Calcium soap deposits (saponification) | Acute pancreatitis |
| Fibrinoid | Immune complex deposition in vessel walls | Vasculitis, malignant HTN |
| Gangrenous | Coagulative + superimposed infection (wet gangrene = liquefactive component) | Limb ischemia |
B. Apoptosis
"Regulated" cell death — mediated by defined molecular pathways. Surgical precision without inflammation.
| Feature | Finding |
|---|---|
| Cell size | Reduced (shrinkage) |
| Nucleus | Fragmentation into nucleosome-size fragments (DNA laddering) |
| Plasma membrane | Intact — altered lipid orientation (phosphatidylserine flips to outer leaflet) |
| Cellular contents | Packaged into apoptotic bodies — phagocytosed by neighboring cells or macrophages |
| Inflammation | Absent |
| Role | Physiologic (embryogenesis, immune selection, tissue homeostasis) OR pathologic (DNA damage, infections) |
Mechanisms of Apoptosis
- Intrinsic (mitochondrial) pathway: DNA damage, misfolded proteins, growth factor withdrawal → pro-apoptotic Bcl-2 family proteins (Bax, Bak) overcome anti-apoptotic members (Bcl-2, Bcl-XL) → cytochrome c release → apoptosome → caspase-9 → caspase-3 activation
- Extrinsic (death receptor) pathway: FasL binds Fas, TNF binds TNFR → DISC formation → caspase-8 → caspase-3 activation
C. Other Forms of Cell Death (Newer Mechanisms)
| Type | Mechanism | Key Feature |
|---|---|---|
| Necroptosis | "Programmed necrosis" via RIPK1/RIPK3/MLKL; morphologically resembles necrosis but is genetically regulated | Causes inflammation (unlike apoptosis) |
| Pyroptosis | Caspase-1/11-dependent; releases IL-1β and IL-18; plasma membrane rupture | Highly pro-inflammatory; occurs in infections |
| Ferroptosis | Iron-dependent accumulation of lipid peroxides; distinct from apoptosis and necrosis | Associated with ischemia-reperfusion injury |
3. Cellular Adaptations
When injury is sublethal and chronic, cells may adapt rather than die:
| Adaptation | Definition | Example |
|---|---|---|
| Hypertrophy | ↑ cell size (↑ workload/trophic signals) | Cardiac hypertrophy in hypertension |
| Hyperplasia | ↑ cell number (growth factor stimulation) | Endometrial hyperplasia (estrogen) |
| Atrophy | ↓ cell size/number (↓ workload, ↓ trophic signals) | Muscle wasting, denervation |
| Metaplasia | One differentiated cell type → another | Barrett esophagus (squamous → columnar) |
Summary Diagram
CELL INJURY
│
├─ Mild/Transient → REVERSIBLE INJURY
│ • Cellular swelling (hydropic change)
│ • Fatty change
│ • Membrane blebs, ER dilation
│ • Recovery if stimulus removed
│
└─ Severe/Persistent → IRREVERSIBLE INJURY → CELL DEATH
│
├─ NECROSIS (pathologic, inflammatory)
│ Patterns: coagulative, liquefactive, caseous,
│ fat, fibrinoid, gangrenous
│
├─ APOPTOSIS (often physiologic, no inflammation)
│ Intrinsic + Extrinsic pathways → caspases
│
└─ REGULATED NECROSIS
Necroptosis / Pyroptosis / Ferroptosis
Sources:
- Robbins & Cotran Pathologic Basis of Disease, 10th ed., Ch. 2 (pp. 35–55)
- Robbins & Kumar Basic Pathology, 11th ed., Ch. 1
This is a shared conversation. Sign in to Orris to start your own chat.