Thrichinococcus infection
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive information. Let me compile a thorough response on Trichinella infection (trichinosis/trichinellosis).
Note on terminology: "Thrichinococcus" does not correspond to a recognized organism. Based on the spelling, this most likely refers to Trichinella infection (trichinosis/trichinellosis). If you meant Echinococcus (hydatid disease), please let me know.
Trichinella Infection (Trichinosis / Trichinellosis)
Trichinosis is a systemic zoonotic illness caused by nematodes of the genus Trichinella — primarily T. spiralis in most of the world, though 8 closely related species can infect humans.
Life Cycle
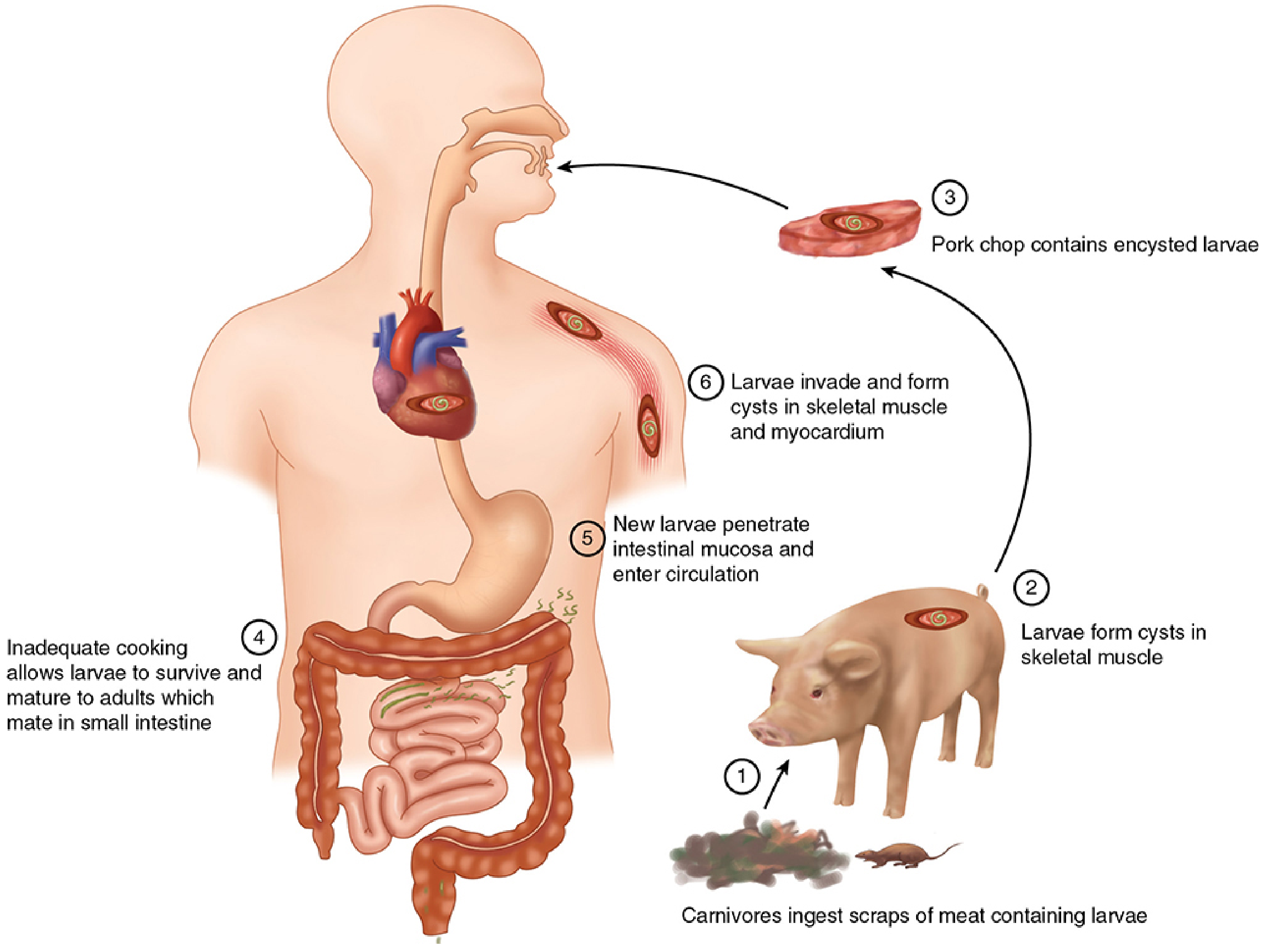
The same host harbors both the adult and larval forms:
- Ingestion of raw/undercooked meat containing encysted larvae
- Cysts dissolve in the GI tract; larvae invade the small intestinal mucosa, living within a syncytium of ~45 villus cells
- Adults mature rapidly; males (60 µm × 1.2 mm) and females (90 µm × 2.2 mm) mate within 30 hours
- Viviparous females begin releasing larvae ~1 week post-ingestion; adults are expelled within 4 weeks
- Larvae (6 × 100 µm) enter intestinal blood and lymphatics → distributed systemically
- Larvae develop only within striated muscle → enter a myocyte → induce transformation into a "nurse cell" → coiled larva remains viable for years
— Sleisenger and Fordtran's Gastrointestinal and Liver Disease, p. 2274
Epidemiology
| Species | Distribution |
|---|---|
| T. spiralis | Americas, Europe, Russia (most common globally) |
| T. nativa | Arctic/subarctic (infects walrus, bear; freeze-resistant) |
| T. britovi | Europe, North Africa, Middle East, Asia |
| T. nelsoni | Equatorial Africa |
| T. pseudospiralis | Americas, Europe, Russia (no encapsulated cysts; infects birds) |
| T. papuae | Papua New Guinea (infects reptiles) |
| T. zimbabwensis | Zimbabwe, Ethiopia, Mozambique (only species not yet implicated in human disease) |
Sources of human infection: undercooked pork, wild boar, bear meat, walrus, horse meat, cougar. Outbreaks are often cluster events — butcher shops, hunting feasts, subsistence feeding.
The incidence has declined markedly in industrialized countries due to: thorough cooking of pork, commercial freezing, and grain-only pig feeding. Trichinosis is a re-emerging illness in Eastern Europe due to relaxed enforcement.
— Sleisenger and Fordtran's GI and Liver Disease, p. 2272–2274; Sherris & Ryan's Medical Microbiology, p. 1870
Pathogenesis
- Enteral phase: Adult worms embed in intestinal epithelium → enteritis (eosinophils, lymphocytes, neutrophils)
- Parenteral phase: Migratory larvae invade striated muscle, heart, CNS → inflammatory responses
- Invaded muscle cells enlarge, lose cross-striations, undergo basophilic degeneration
- Specific IgG and IgM antibodies trigger eosinophil-mediated destruction of circulating larvae
- A vasculitis from circulating immune complex deposition may occur
- Th2 cytokines (IL-4, IL-5), T lymphocytes, eosinophils, and mast cells mediate worm expulsion
Clinical Features
Clinical trichinosis has two sequential phases:
Phase 1 — Intestinal (Enteral, Days 2–14)
- Nausea, abdominal pain, diarrhea, vomiting, low-grade fever
- Often mistaken for viral gastroenteritis or food poisoning
- Diarrhea can persist for weeks (T. nativa from walrus meat in Inuit populations)
Phase 2 — Systemic/Parenteral (Week 1–6+)
- Fever, myalgia, muscle weakness, muscle tenderness (cardinal features)
- Periorbital/eyelid edema (characteristic)
- Maculopapular skin rash
- Subconjunctival and subungual (splinter) hemorrhages
- Dysphagia, headache, paresthesias
Severe Disease
- Myocarditis → ECG abnormalities, tachycardia, congestive heart failure
- CNS involvement → encephalitis, meningitis, polyneuritis, delirium, psychosis, paresis, coma
- Pulmonary → hemoptysis, consolidation
- Vasculitis / venous thrombosis
- Death (larvae burden >1,000–5,000/g of tissue)
Larval burden determines severity: <10 larvae/g → asymptomatic; 100/g → significant disease; 1,000–5,000/g → potentially fatal.
— Sherris & Ryan's Medical Microbiology, p. 1871; Sleisenger and Fordtran, p. 2275
Diagnosis
| Test | Finding |
|---|---|
| Eosinophilia | 15–50% eosinophils; begins week 2, persists throughout illness; may disappear in fatal cases |
| Serum IgE | Elevated in most clinically ill patients |
| CPK (creatine phosphokinase) | Elevated (myositis) |
| Serology (IFA, ELISA) | Significant titers appear after week 3; may persist indefinitely |
| Muscle biopsy (deltoid or gastrocnemius) | Reveals encysted larvae during week 3 onward |
Serologic tests are usually negative before week 3, so early diagnosis relies on clinical picture + epidemiologic exposure history.
Treatment
| Indication | Treatment |
|---|---|
| Mild/moderate | Albendazole 400 mg twice daily × 10–15 days OR Mebendazole 5 mg/kg/day × 10–15 days |
| Severe (edema, pulmonary, cardiac, CNS) | Add glucocorticoids (corticosteroids first, then anthelmintics — to prevent hypersensitivity reaction from mass larval destruction) |
- Anthelmintics kill adult worms in the gut and halt larval production; they are less effective against already-encysted larvae
- Glucocorticoids given without anthelmintics may prolong the intestinal phase, increasing larval release — so combination is preferred in severe cases
- Mortality: ~1% in symptomatic cases; rises to ~10% with CNS involvement
— Sleisenger and Fordtran, p. 2276; Sherris & Ryan's, p. 1872
Prevention
- Thoroughly cook all meat (especially pork, wild game, bear) to ≥71°C (160°F) internally
- Freezing pork at −15°C for ≥3 weeks kills most strains (but not T. nativa, the arctic strain)
- Federal/agricultural regulations: grain-only feed for commercial pigs; meat inspection
- Control rodent contact with domestic pigs
Sources: Sleisenger and Fordtran's Gastrointestinal and Liver Disease | Sherris & Ryan's Medical Microbiology, 8th ed. | Bradley and Daroff's Neurology in Clinical Practice | Fuster and Hurst's The Heart, 15th ed.
This is a shared conversation. Sign in to Orris to start your own chat.