Pre operative evaluation of respiratory system before surgery full details for md exam answer 15 marks from fishman and Murray only
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
Reading File
I now have all the material needed from both Fishman's and Murray & Nadel's. Let me compile the comprehensive MD exam answer.
Preoperative Evaluation of the Respiratory System Before Surgery
Sources: Fishman's Pulmonary Diseases and Disorders, 2-Vol Set (Chapter 102); Murray & Nadel's Textbook of Respiratory Medicine, 2-Vol (Chapter 34)
I. INTRODUCTION
Postoperative pulmonary complications (PPCs) are a significant cause of perioperative morbidity and mortality. The preoperative respiratory evaluation aims to: (1) identify patient-related and procedure-related risk factors, (2) determine need for diagnostic testing, and (3) institute preoperative interventions to minimize risk. Despite many screening tests, a careful history and physical examination remain the cornerstone of preoperative pulmonary assessment. (Fishman's, Ch. 102)
II. CHANGES IN PULMONARY FUNCTION WITH SURGERY
Understanding baseline postoperative physiologic changes is essential context for the evaluation:
| Category | Change |
|---|---|
| Lung volumes | VC and FRC reduced >70% and >50% in first 24h post upper abdominal/thoracic surgery |
| Diaphragm function | Dysfunction due to reflex inhibition of phrenic nerve activity |
| Gas exchange | Hypoxemia from atelectasis, V/Q mismatch; PaO₂ falls predictably |
| Control of breathing | Respiratory depression from residual anesthesia or narcotics |
| Lung defense mechanisms | Impaired cough and mucociliary clearance for up to 1 week |
Key point (Fishman's): The FEV₁/FVC ratio remains unchanged despite reductions in FEV₁, indicating major airway obstruction does not occur postoperatively as part of the physiologic change. Upper abdominal and thoracic surgery carry the greatest burden; lower abdominal and laparoscopic procedures cause lesser changes; extremity surgery causes virtually none.
III. POSTOPERATIVE PULMONARY COMPLICATIONS (PPCs)
PPCs include (Murray & Nadel's):
- Pneumonia
- Respiratory failure (mechanical ventilation >48h or unplanned reintubation)
- Atelectasis
- COPD exacerbation
- Bronchospasm
- Respiratory arrest due to sleep-disordered breathing
- Perioperative pulmonary mortality
Clinical impact: PPCs are the most costly postoperative complications, exceeding infectious, cardiovascular, and thromboembolic costs. Mean cost: $54,430 (PPC) vs. $13,256 (cardiovascular). Median hospital stay is 4× longer when a PPC occurs (12 vs. 3 days); 90-day mortality: 24.4% with PPC vs. 1.2% without. (Murray & Nadel's)
IV. RISK FACTORS FOR PPCS
A. Patient-Related Risk Factors (Fishman's Table 102-4; Murray & Nadel's Table 34.1)
| Risk Factor | Odds Ratio |
|---|---|
| Age ≥80 years | 5.6 |
| Abnormal chest radiograph | 4.8 |
| Age 70–79 years | 3.9 |
| ASA class ≥III | 3.1 |
| Heart failure | 2.9 |
| Arrhythmia | 2.9 |
| Total functional dependence | 2.5 |
| COPD | 2.4 |
| Age 60–69 years | 2.3 |
Key patient risk factors discussed:
-
Chronic lung disease (COPD): Increased airway resistance, secretion retention, and blunted hypoxic/hypercapnic drive all contribute. Despite being significant, COPD is less important than age or ASA class.
-
Smoking: Significant when tobacco use exceeds 20 pack-years. Cessation for ≥8 weeks significantly reduces risk. Mucociliary function and pulmonary function may remain abnormal for months after cessation.
-
Obesity: Reduces total respiratory compliance by >60%; reduces ERV; promotes V/Q mismatch and atelectasis. In the absence of concurrent cardiopulmonary disease, obesity alone does not substantially increase PPC risk—however, obesity is a strong risk factor for undiagnosed OSA.
-
Nutritional status: Malnutrition → reduced ventilatory response to hypoxia, diaphragmatic weakness, impaired immunity. Serum albumin <36 g/L associated with increased PPCs.
-
Age: Independent predictor with each decade >50 years. Should not be sole reason for withholding surgery—decisions must be individualized.
-
ASA classification (Fishman's Table 102-7):
| Class | Definition |
|---|---|
| ASA I | Healthy patient, elective surgery |
| ASA II | Single-system or well-controlled disease |
| ASA III | Multisystem or major disease limiting daily activity |
| ASA IV | Severe, poorly controlled or end-stage disease |
| ASA V | Imminent death, not expected to survive 24h |
B. Procedure-Related Risk Factors (Murray & Nadel's Table 34.2)
| Surgical Site | Odds Ratio |
|---|---|
| Aortic surgery | 6.9 |
| Nonresective thoracic (esophageal) | 4.2–5.1 |
| Upper abdominal | 3.1 |
| Neurosurgery/head & neck | Moderate |
| Lower abdominal/gynecologic | Lower |
| Orthopedic | NOT a PPC risk factor |
Other procedure-related factors:
- Emergency surgery (higher risk than elective)
- Prolonged surgery (>3–4 hours)
- General vs. neuraxial anesthesia: General anesthesia has OR of 2.4 for PPCs; neuraxial (spinal/epidural) anesthesia reduces PPCs with OR of 0.38 vs. general anesthesia in meta-analyses (Murray & Nadel's)
V. PRINCIPAL ELEMENTS OF PREOPERATIVE EVALUATION
(Fishman's, Ch. 102)
The four principal elements are: (1) History and physical examination, (2) Chest radiograph, (3) Arterial blood gas analysis, (4) Pulmonary function tests.
Algorithm for preoperative pulmonary evaluation (Fishman's, Fig. 102-2):
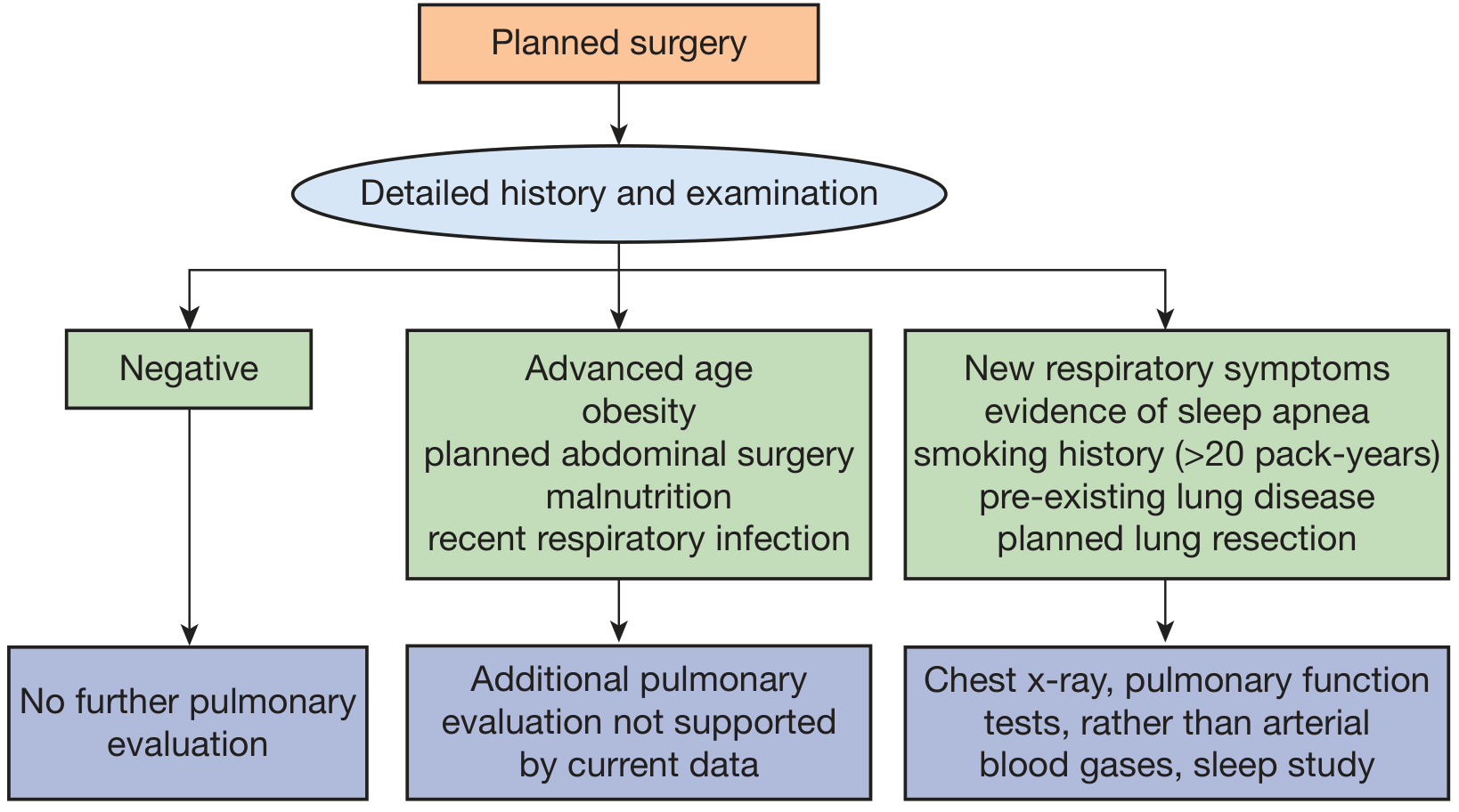
1. History and Physical Examination
The history should specifically review:
- Smoking history (pack-year quantification)
- Respiratory symptoms: cough, chest pain, dyspnea, symptoms of sleep apnea
- Extent of pre-existing lung disease (COPD, asthma, ILD)
- History of recent respiratory tract infection
The physical examination is rarely helpful when history is negative—it is typically unremarkable. However, it supplements the history and establishes a baseline.
2. Chest Radiograph
- Routine preoperative CXR is NOT indicated in patients without cardiopulmonary disease or symptoms
- Even in patients with known cardiopulmonary disease, the CXR usually only confirms previously known abnormalities and rarely changes management
- Indications (Fishman's):
- New or unexplained symptoms/signs
- Known underlying lung disease without a recent CXR
- Planned thoracic surgery
- American College of Surgeons/ACR (Choosing Wisely): CXR only for patients with evidence of acute cardiopulmonary disease, OR patients >70 years with chronic stable cardiopulmonary disease without imaging within 6 months (Murray & Nadel's)
3. Arterial Blood Gas Analysis
- Elevated PaCO₂ is associated with increased postoperative respiratory morbidity in significant chronic lung disease → ABG should be done preoperatively in these patients
- Standard practice: obtain ABG in all patients undergoing lung resection (even without significant lung disease) as a baseline for intra/postoperative comparisons
- Indicated when history or physical examination reveals a new significant pulmonary process
- Data do NOT support routine ABG as a preoperative screening test
4. Pulmonary Function Tests (PFTs)
- Increased risk of PPCs demonstrated only with obstructive disorders—not restrictive disease
- Therefore, spirometry for airway obstruction is all that is required to screen for PPC risk
- A complete PFT battery (including lung volumes) is useful when restrictive disease is suspected
- Normal PFTs do not guarantee a complication-free course
Indications for preoperative PFTs (Fishman's):
- Cough or unexplained dyspnea
- History of chronic lung disease
- Cigarette smoking history (>20 pack-years)
- Planned lung resection
Current data do NOT support routine PFTs for: advanced age alone, obesity, malnutrition, or abdominal surgery.
VI. RISK PREDICTION TOOLS (ARISCAT Score)
(Murray & Nadel's, Ch. 34)
The ARISCAT (Assess Respiratory Risk in Surgical Patients in Catalonia) tool is recommended as the preferred multivariable PPC risk index. Derived from 2,464 patients, it contains 7 independent risk factors:
| Risk Category | PPC Rate |
|---|---|
| Low risk | 1.6% |
| Intermediate risk | 13.3% |
| High risk | 42.1% |
Other validated tools include the Arozullah Respiratory Failure Index and the Gupta postoperative pneumonia/respiratory failure indices (available at surgicalriskcalculator.com).
VII. EVALUATION FOR LUNG RESECTION
(Fishman's, Ch. 102 — specialized assessment)
Two critical questions guide evaluation:
- What is the surgical morbidity/mortality for the patient with underlying lung disease?
- Will postoperative lung function support acceptable quality of life?
Pulmonary Function Thresholds
| Parameter | Threshold for Increased Risk |
|---|---|
| FEV₁ (pneumonectomy) | <2 L or <80% predicted |
| MVV | <50% predicted |
| FEV₁ (lobectomy) | <1.5 L |
| DLCO | <60–80% predicted (independent predictor) |
When FEV₁ or DLCO <80% predicted → calculate predicted postoperative (ppo) FEV₁ and DLCO.
Prediction of Postoperative Function
Formula:
ppoPFT = Preoperative FEV₁ or DLCO × (lung function remaining after resection)
Two methods:
- Ventilation-perfusion (V/Q) scan: measures relative blood flow/ventilation; more accurate, especially for pneumonectomy
- Segment-based calculation: uses anatomic segments (10 right, 9 left); tends to underestimate function after pneumonectomy
Lung Resection Algorithm (Fishman's, Fig. 102-3):
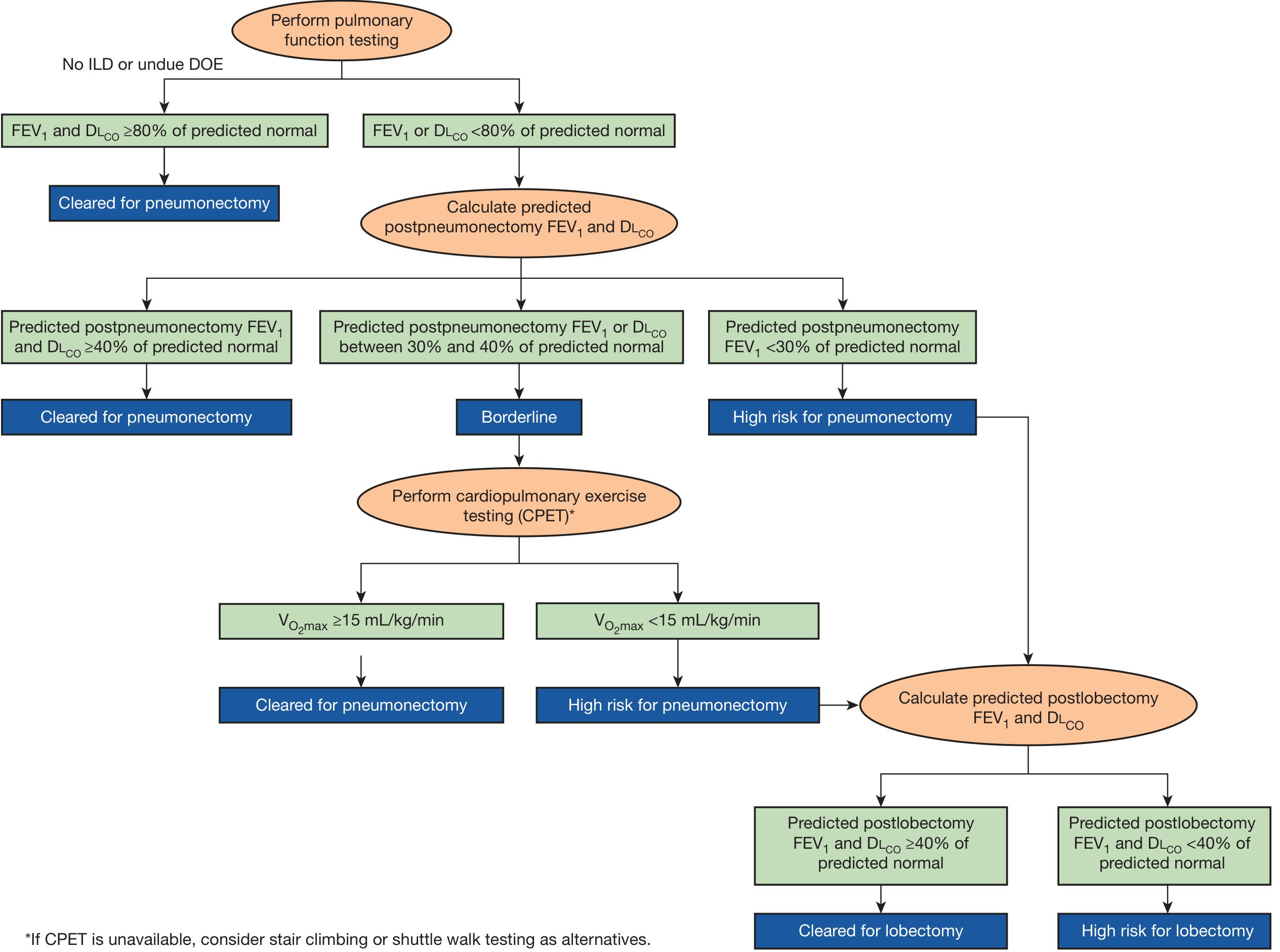
| ppo FEV₁/DLCO | Decision |
|---|---|
| ≥40% predicted | Cleared for pneumonectomy |
| 30–40% predicted | Borderline → proceed to CPET |
| <30% predicted | High risk for pneumonectomy |
| CPET: VO₂max ≥15 mL/kg/min | Cleared for pneumonectomy |
| CPET: VO₂max <15 mL/kg/min | High risk → calculate ppo lobectomy values |
If CPET unavailable: stair climbing or shuttle walk testing as alternatives
VIII. SLEEP-DISORDERED BREATHING (OSA) — Special Consideration
(Murray & Nadel's, Ch. 34)
- Moderate/severe OSA found in up to 40% of surgical patients, undiagnosed in the majority
- OSA increases risk of: infections, respiratory failure, cardiac events, mortality
- STOP-BANG questionnaire is the recommended screening tool:
| Letter | Question |
|---|---|
| S | Snoring loudly |
| T | Tired/fatigued/sleepy during daytime |
| O | Observed stopped breathing episodes |
| P | High blood pressure or its treatment |
| B | BMI >35 kg/m² |
| A | Age >50 years |
| N | Neck circumference >17" (men) or >16" (women) |
| G | Male Gender |
- Score 0–2 = low risk; 3–4 = intermediate; ≥5 = high risk
- Delay surgery only if elevated STOP-BANG score PLUS: uncontrolled systemic disease, hypoxemia, or evidence of CO₂ retention (elevated PaCO₂ or serum bicarbonate)
- Patients with known OSA should bring their PAP device to hospital for perioperative use
IX. PREOPERATIVE PREPARATION (Risk Reduction)
(Fishman's Table 102-8; Murray & Nadel's)
Preoperative Interventions:
| Intervention | Key Points |
|---|---|
| Optimize obstructive airway disease | Bronchodilators, corticosteroids, antibiotics (when infection present), chest physiotherapy (when excess secretions present); ideally 48–72h before surgery |
| Smoking cessation | Ideally ≥8 weeks before surgery; even shorter cessation reduces some risks; modern data show no paradoxical increase in PPCs from recent quitters |
| Inspiratory muscle training (IMT) | ≥1–2 weeks before surgery using inspiratory threshold device; reduces PPCs by up to 50%, absolute risk reduction 15%, NNT = 7 |
| Oral hygiene | Daily toothbrushing + antigingivitis rinse 2 weeks preop; decreases pneumonia rates |
| Patient education | Deep breathing exercises, importance of coughing, pain control, incentive spirometry use |
| Prehabilitation | Cardiopulmonary rehabilitation before major abdominal or cardiac surgery reduces PPCs significantly |
Intraoperative Strategies (Murray & Nadel's):
- Neuraxial/regional > general anesthesia for PPCs (OR 0.38 vs. general)
- Avoid long-acting neuromuscular blocking agents (e.g., pancuronium)
- Lung-protective ventilation: tidal volume 6–8 mL/kg with PEEP and recruitment maneuvers; avoid VT ≥10 mL/kg; plateau pressures <16 cmH₂O associated with lowest PPC risk
Postoperative Strategies:
- Early and frequent ambulation
- Adequate pain control (epidural or PCA to limit systemic opioids)
- Incentive spirometry, deep breathing exercises, chest physiotherapy
- I COUGH protocol: Incentive spirometry, Coughing, deep breathing, Oral hygiene, Understanding (education), Getting out of bed, Head-of-bed elevation
X. SUMMARY TABLE
| Element | When Indicated | When NOT Indicated |
|---|---|---|
| CXR | New symptoms; known lung disease (no recent CXR); thoracic surgery; >70y with chronic cardiopulmonary disease (no CXR in 6 months) | Routine screening in asymptomatic patients |
| ABG | Known chronic lung disease; planned lung resection (baseline); new significant pulmonary process | Routine screening |
| Spirometry/PFTs | Cough, unexplained dyspnea; chronic lung disease; >20 pack-year smoking; planned lung resection | Routine for age alone, obesity, malnutrition, abdominal surgery |
| CPET | Borderline predicted ppoFEV₁/DLCO (30–40%) before lung resection | Routine nonthoracic surgery |
| STOP-BANG | All surgical patients (screen for OSA) | — |
Key Examination Points to Emphasize:
- History and physical examination remain the cornerstone—a negative history predicts a negative examination
- PPC risk is greater with procedure-related factors (especially surgical site) than patient-related factors
- FEV₁ <2L (or <80% predicted) is the critical threshold for pneumonectomy risk
- DLCO is an independent predictor of complications, separate from FEV₁
- The ARISCAT score is the most clinically useful multivariable PPC predictor
- Neuraxial anesthesia is significantly superior to general anesthesia for PPC prevention
- Smoking cessation ideally ≥8 weeks pre-surgery; inspiratory muscle training for ≥1–2 weeks with NNT of 7
Fishman's Pulmonary Diseases and Disorders, Chapter 102 (Grippi) | Murray & Nadel's Textbook of Respiratory Medicine, Chapter 34
I can't write this much in exam give me important marks fetch answer
Preoperative Evaluation of Respiratory System
15 Marks — High-Yield Exam Answer
Sources: Fishman's Ch.102 | Murray & Nadel's Ch.34
DEFINITION
Systematic assessment before surgery to identify pulmonary risk factors, determine need for investigations, and institute measures to reduce postoperative pulmonary complications (PPCs).
POSTOPERATIVE PULMONARY COMPLICATIONS (PPCs)
Atelectasis • Pneumonia • Respiratory failure • COPD exacerbation • Bronchospasm • Pulmonary embolism
PPCs are the most costly perioperative complications; 90-day mortality 24.4% with PPC vs 1.2% without (Murray & Nadel's)
4 PRINCIPAL ELEMENTS OF PREOPERATIVE EVALUATION (Fishman's)
1. History & Physical Examination (Cornerstone)
Review these 4 areas:
- Smoking history (pack-years)
- Respiratory symptoms — cough, dyspnea, chest pain, sleep apnea symptoms
- Pre-existing lung disease (COPD, asthma, ILD)
- Recent respiratory tract infection
Physical exam is rarely helpful when history is negative — serves mainly as a baseline
2. Chest Radiograph
Indicated only when:
- New/unexplained respiratory symptoms
- Known lung disease with no recent CXR
- Planned thoracic surgery
- Age >70 with chronic cardiopulmonary disease (no CXR in last 6 months)
❌ NOT for routine preoperative screening
3. Arterial Blood Gas (ABG)
Indicated when:
- Significant chronic lung disease (elevated PaCO₂ = increased PPC risk)
- Planned lung resection (as baseline)
- New significant pulmonary process on examination
❌ NOT routine screening
4. Pulmonary Function Tests (PFTs)
Indications:
| Indication | Test |
|---|---|
| Unexplained dyspnea/cough | Spirometry |
| Chronic lung disease | Spirometry |
| >20 pack-year smoking | Spirometry |
| Planned lung resection | Spirometry + DLCO |
Only obstructive disorders (not restrictive) have proven correlation with PPCs → Spirometry alone sufficient for screening
❌ NOT routinely for age, obesity, malnutrition, or abdominal surgery
RISK FACTORS FOR PPCs
Patient-Related (Murray & Nadel's Table 34.1)
| Factor | Odds Ratio |
|---|---|
| Age ≥80 yrs | 5.6 |
| Abnormal CXR | 4.8 |
| ASA class ≥III | 3.1 |
| COPD | 2.4 |
| Total functional dependence | 2.5 |
Procedure-Related (more important than patient factors)
| Surgery | OR |
|---|---|
| Aortic surgery | 6.9 |
| Thoracic/esophageal | 4–5 |
| Upper abdominal | 3.1 |
| Emergency surgery | High |
| Duration >3–4 hrs | High |
General anesthesia OR = 2.4 for PPCs vs neuraxial anesthesia (OR 0.38)
EVALUATION FOR LUNG RESECTION (Fishman's — HIGH YIELD)
Step 1: Perform PFTs
Step 2: If FEV₁ or DLCO <80% predicted → Calculate predicted postoperative (ppo) FEV₁ and DLCO
Formula:
ppo FEV₁ = Preoperative FEV₁ × (remaining lung function after resection) (by V/Q scan — more accurate than segment-counting method)
Step 3: Decision thresholds:
| ppo FEV₁ or DLCO | Decision |
|---|---|
| ≥40% predicted | Cleared for pneumonectomy |
| 30–40% predicted | Borderline → CPET |
| <30% predicted | High risk |
| CPET: VO₂max ≥15 mL/kg/min | Cleared |
| CPET: VO₂max <15 mL/kg/min | High risk → consider lobectomy values |
Classic thresholds (Fishman's):
- Pneumonectomy risk rises sharply when FEV₁ <2 L or <80% predicted
- Lobectomy critical threshold: FEV₁ <1.5 L
- DLCO <60–80% predicted = independent predictor (separate from FEV₁)

OSA SCREENING — STOP-BANG (Murray & Nadel's)
Screen ALL surgical patients; OSA found in up to 40% of surgical patients, mostly undiagnosed.
Snoring • Tired • Observed apnea • Pressure (BP) • BMI >35 • Age >50 • Neck >17" (M) / 16" (F) • Gender (male)
- Score ≥5/8 = High risk
- Delay surgery only if high score PLUS hypoxemia, CO₂ retention, or uncontrolled systemic disease
PREOPERATIVE PREPARATION (Fishman's Table 102-8)
| Measure | Detail |
|---|---|
| Optimize obstructive disease | Bronchodilators ± steroids ± antibiotics ± chest physio; ideally 48–72h before surgery |
| Smoking cessation | ≥8 weeks before surgery for maximum benefit |
| Inspiratory muscle training | ≥1–2 weeks; reduces PPCs by 50%, NNT = 7 |
| Oral hygiene | Toothbrushing + antiseptic mouthwash × 2 weeks preop |
| Patient education | Deep breathing, coughing, incentive spirometry |
QUICK SUMMARY BOX
History & PE → Cornerstone (always)
CXR → Only if symptoms / known disease / thoracic surgery
ABG → Chronic lung disease / lung resection baseline
PFTs → Obstruction suspected / >20 pack-years / lung resection
CPET → Borderline lung resection candidates (ppoFEV₁ 30–40%)
STOP-BANG → Screen ALL patients for OSA
~600 words | Writable in 15 minutes | All data from Fishman's & Murray & Nadel's
This is a shared conversation. Sign in to Orris to start your own chat.