explain Neurocysticercosis from basic to advanced medicine in structured way
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching PubMed
neurocysticercosis diagnosis treatment
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Searching Images
neurocysticercosis MRI scolex stages brain cyst
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive data from multiple authoritative sources. Let me compile the complete structured response.
Neurocysticercosis (NCC) — Basic to Advanced Medicine
1. DEFINITION & OVERVIEW
Neurocysticercosis (NCC) is CNS infection by the larval (cysticercus) stage of Taenia solium, the pork tapeworm. It is the most common parasitic disease of the human CNS worldwide and the leading cause of symptomatic/provoked epilepsy globally.
"NCC is the most common cause of symptomatic epilepsy worldwide." — Bradley and Daroff's Neurology in Clinical Practice
The cysticercus consists of a thin translucent bladder containing an invaginated scolex. NCC encompasses two major anatomical categories:
- Parenchymal NCC — cysts within brain parenchyma (most common)
- Extraparenchymal NCC — cysts in ventricles, subarachnoid space, spine, or eye
2. EPIDEMIOLOGY
| Region | Burden |
|---|---|
| Latin America | 0.45–1.35 million with epilepsy from NCC |
| India | ~1 million |
| Africa | 0.31–4.6 million |
| United States | ~1,320–5,050 new cases/year (driven by immigration) |
- Endemic in Central/South America, sub-Saharan Africa, India, Eastern Europe, and parts of Asia
- An emerging infection in developed countries due to immigration and international travel
- In the USA, 33,060 cysticercosis-related hospitalizations recorded between 1998–2011 (rate: 8.03 per million population)
— Bradley and Daroff's Neurology in Clinical Practice; Goldman-Cecil Medicine
3. LIFE CYCLE & PATHOGENESIS
The Dual Role of the Human Host
| Role | Mechanism | Disease |
|---|---|---|
| Definitive host | Ingests undercooked pork containing cysticerci → adult tapeworm in gut | Taeniasis (intestinal) |
| Accidental intermediate host | Ingests T. solium eggs via fecal-oral contamination → cysticercosis | NCC |
Step-by-Step:
- A human tapeworm carrier sheds proglottids loaded with eggs in stool
- Eggs contaminate food/water/hands via fecal-oral route (auto-infection possible in carrier)
- After ingestion, oncospheres hatch in intestine, penetrate the mucosa
- Haematogenous dissemination to brain, muscle, eye, subcutaneous tissue
- Larvae mature into cysticerci in CNS tissue — initially immunologically tolerated ("phase of tolerance")
- As the parasite begins to die, the host immune response is triggered → perilesional inflammation → seizures, edema, neurological deficits
Key point: You do not need to eat pork to get NCC. A strict vegetarian can develop NCC through fecal-oral transmission of eggs from a tapeworm carrier. — Sherris & Ryan's Medical Microbiology
4. ANATOMICAL SUBTYPES
A. Parenchymal NCC (most common)
- Cysts lodge in the grey-white junction and cortex
- Progress through 4 stages (see Section 7 — Neuroimaging)
- Main presentation: focal seizures ± secondary generalization
B. Intraventricular NCC (second most common)
- Most often in the fourth and third ventricles, less commonly lateral ventricles
- Cysts are mobile (can migrate, documented on sequential imaging)
- Can cause acute obstructive hydrocephalus — a neurosurgical emergency
C. Subarachnoid / Racemose NCC
- Involves basal cisterns, sylvian fissures, cerebellopontine angle
- Racemose form: multiple clustered cysts separated by septa → classic "bunch of grapes" appearance
- Can cause large cystic lesions with meningeal nodular enhancement
- Higher morbidity; associated with basilar arachnoiditis and communicating hydrocephalus
D. Spinal NCC
- Rare; mimics intraspinal tumors
- Can be intra- or extramedullary
E. Ocular Cysticercosis
- Cyst in vitreous, subretinal space
- Can cause vision loss; surgical removal required
5. PATHOLOGY & IMMUNOPATHOLOGY
| Phase | Pathological State | Host Response |
|---|---|---|
| Vesicular (viable) | Thin-walled cyst, intact scolex | Minimal inflammation — "immunological tolerance" |
| Colloidal (early degeneration) | Cyst wall thickens, fluid becomes turbid | Robust inflammatory response — perilesional edema, BBB disruption |
| Granular-nodular | Cyst collapses, thick enhancing wall | Progressive fibrosis, decreasing edema |
| Calcified nodular | Dystrophic calcification, no viable parasite | Minimal — but calcifications can cause perilesional edema during seizures |
The degenerating parasite releases antigens that activate complement, eosinophils, and T-helper cells, producing localized cerebral edema and BBB disruption — this is when most clinical symptoms emerge, not when the parasite first arrives.
6. CLINICAL PRESENTATIONS
A. Seizures (most common — 50–70% of cases)
- New-onset focal seizures with or without secondary generalization
- Symptoms typically emerge ~5 years after initial infection
- In 80–90% of single-lesion cases, seizures resolve within 3–6 months as lesion calcifies
- Up to 20% develop refractory epilepsy requiring long-term AEDs
B. Raised Intracranial Pressure
- Due to intraventricular/subarachnoid cysts blocking CSF outflow → hydrocephalus
- Presents with headache, vomiting, papilledema
- Intraventricular cysts may cause sudden death from acute obstructive hydrocephalus
C. Focal Neurological Deficits
- Hemiparesis, hemisensory loss, cerebellar ataxia depending on cyst location
D. Meningitis / Encephalitis
- Basal arachnoiditis from subarachnoid NCC → cranial nerve palsies, communicating hydrocephalus
- Diffuse cysticercal encephalitis (rare) — mostly in children/young women with massive cyst burden → severe cerebral edema
E. Psychiatric Manifestations
- Cognitive impairment, personality change — recognized in Kaplan & Sadock's Comprehensive Textbook of Psychiatry
F. Spinal Cord Syndrome
- Cord compression or myelopathy
7. NEUROIMAGING — THE FOUR STAGES
MRI (preferred) vs CT
| Stage | MRI Appearance | CT Appearance | Clinical Correlation |
|---|---|---|---|
| Vesicular (viable) | Thin-walled cyst, CSF-isointense fluid, scolex as hyperintense dot on T1/FLAIR/DWI — "cyst with dot sign" | Hypodense cyst, scolex as hyperdense nodule | Minimal symptoms; scolex = pathognomonic |
| Colloidal vesicular (degenerating) | Proteinaceous fluid → T1/FLAIR hyperintense; ring enhancement + perilesional edema | Ring-enhancing lesion with edema | Seizures, headache — most symptomatic stage |
| Granular-nodular (dying) | Partially collapsed cyst, thick enhancing wall, decreasing edema | Thick-walled nodule | Resolving symptoms |
| Calcified nodular (dead) | Small calcified foci 2–10 mm; no enhancement typically, no edema | Hyperdense calcifications | Usually asymptomatic unless peri-ictal edema flares |
"Cyst with a dot" sign — a CSF-signal cyst containing a small eccentric hyperintense scolex — is pathognomonic for viable NCC on MRI.
Special Forms on Imaging
- Racemose NCC: "bunch of grapes" multilobulated cystic mass in subarachnoid cisterns — no scolex visible
- Intraventricular NCC: Mobile cysts — FLAIR shows cyst as slightly hyperintense vs CSF; loss of flow void within ventricle
- Mixed stages: Multiple cysts at different stages simultaneously are common
Figure: MRI stages of NCC
(A/B) Vesicular stage — CSF-signal cysts with hyperintense scolex dot (arrows); (C/D) Colloidal vesicular stage — ring-enhancing lesion with prominent FLAIR edema; (E/F) Granular-nodular stage — partial collapse with rim enhancement
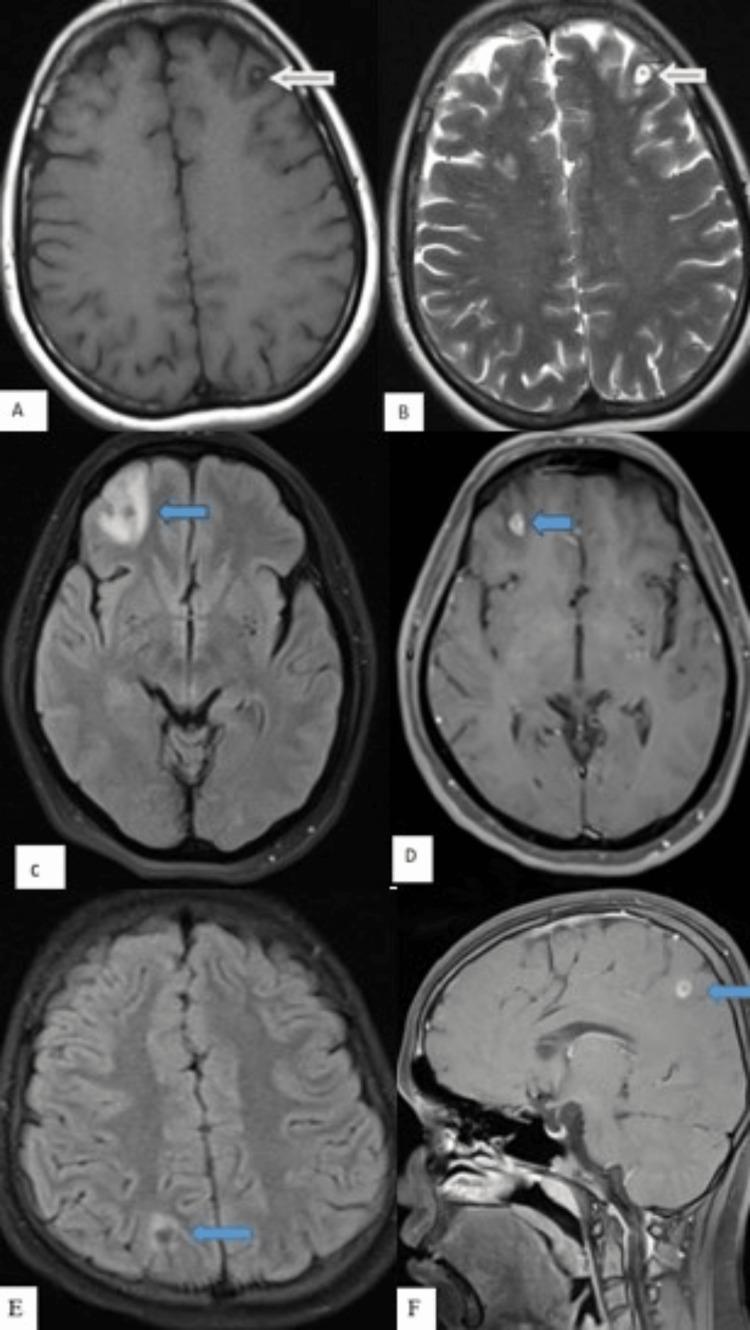
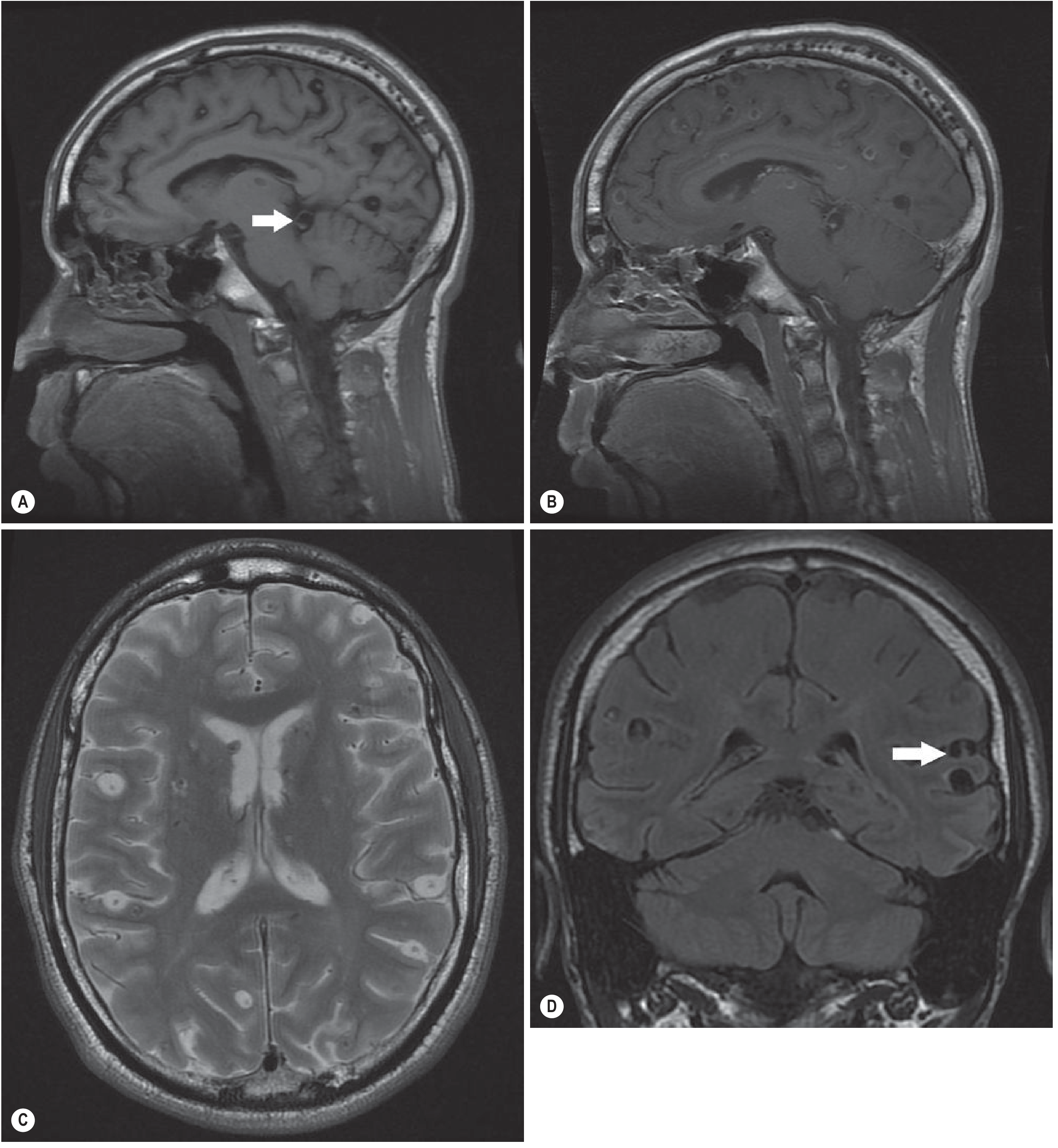
Figure: Extensive parenchymal and subarachnoid NCC (textbook case)
(Sagittal T1, contrast T1, axial T2, coronal FLAIR) — multiple thin-walled vesicular-stage cysts with scolex, with subarachnoid extension (arrow)
8. DIAGNOSIS — Del Brutto Revised Criteria (2017)
The diagnosis integrates neuroimaging, serology, clinical findings, and exposure history.
(Source: Del Brutto OH et al., J Neurol Sci 372:202, 2017 — reproduced in Harrison's Principles of Internal Medicine 22E)
Absolute Criteria (definitive diagnosis if any one met):
- Histologic demonstration of parasite from brain/spinal cord biopsy
- Visualization of subretinal cysticercus on ophthalmoscopy
- Conclusive demonstration of scolex within a cystic lesion on neuroimaging
Neuroimaging Criteria:
| Level | Finding |
|---|---|
| Major | Cystic lesion without scolex; typical small enhancing lesion; multilobulated subarachnoid cyst; parenchymal calcifications |
| Confirmative | Resolution of cysts after cysticidal therapy; documented migration of ventricular cysts |
| Minor | Obstructive hydrocephalus; abnormal basal leptomeningeal enhancement |
Clinical/Exposure Criteria:
| Level | Finding |
|---|---|
| Major | Positive EITB (enzyme-linked immunoelectrotransfer blot) antibody; cysticercosis outside CNS; household contact with T. solium |
| Minor | Clinical features suggestive of NCC; coming from/living in endemic area |
Diagnostic Certainty:
- Confirmed: 1 absolute criterion OR 2 major criteria OR 1 major + confirmative neuroimaging + any clinical/exposure criterion
- Probable: 1 major neuroimaging + ≥2 clinical/exposure criteria (including ≥1 major)
9. LABORATORY & SEROLOGICAL DIAGNOSIS
| Test | Characteristics |
|---|---|
| EITB (lentil lectin-purified glycoproteins) | >99% specific and sensitive in multiple-cyst disease; serum preferred over CSF; may be negative with single cyst or calcifications |
| ELISA (crude antigen) | High false-positive and false-negative rates — not recommended |
| Antigen detection (blood/CSF/urine) | Available commercially in Europe; useful for extraparenchymal disease monitoring |
| CSF analysis | Pleocytosis (eosinophils), elevated protein, low glucose — in subarachnoid/meningitic forms |
| Real-time PCR | Increasingly used for diagnosis and follow-up, especially in extraparenchymal disease |
10. TREATMENT
Guiding Principles
Treatment must be individualized based on:
- Number and viability of cysts
- Location (parenchymal vs extraparenchymal)
- Inflammatory state (edema, encephalitis)
(Adapted from IDSA/ASTMH 2017 Clinical Practice Guidelines — White et al., Clin Infect Dis 2018)
Treatment Table by Form
| Form | Subgroup | Recommendation |
|---|---|---|
| Parenchymal — viable/enhancing (1–2 cysts) | — | Albendazole monotherapy + corticosteroids |
| Parenchymal — viable/enhancing (>2 cysts) | — | Albendazole + praziquantel combination + corticosteroids |
| Parenchymal — calcified | Any number | No antiparasitic treatment |
| Parenchymal — encephalitis (diffuse edema) | — | No antiparasitic; steroids only |
| Intraventricular — resectable | Lateral/3rd ventricle | Neuroendoscopic removal (no medical therapy if successful) |
| Intraventricular — 4th ventricle | — | Neuroendoscopic or microsurgical removal |
| Intraventricular — not resectable | — | Ventricular shunt → then antiparasitic + steroids |
| Subarachnoid (with hydrocephalus) | — | Shunt first → prolonged albendazole (± praziquantel) + steroids |
| Hydrocephalus, no visible cysts | — | Ventricular shunt; no antiparasitic |
| Spinal NCC | Intra/extramedullary | Surgical removal or antiparasitic + steroids (individualized) |
| Ocular cysticercosis | — | Surgical resection — antiparasitics contraindicated (risk of vision loss from inflammation) |
— Bradley and Daroff's Neurology in Clinical Practice, Table 79.7
Drug Details
| Drug | Dose | Mechanism |
|---|---|---|
| Albendazole | 15 mg/kg/day in 2 divided doses × 8–30 days | Inhibits tubulin polymerization in the parasite; better CNS penetration than praziquantel; enhanced by fatty meal and concurrent steroids |
| Praziquantel | 50–100 mg/kg/day in 3 divided doses | Disrupts tegumental integrity; increased calcium permeability → spastic paralysis of parasite |
| Dexamethasone/prednisone | Variable | Reduces perilesional edema, prevents steroid-induced crisis from larval lysis |
Note: Dexamethasone reduces praziquantel levels by 50% via CYP3A4 induction — dose adjustment or preference for albendazole is recommended when combination therapy is used.
Seizure Management
- Antiepileptic monotherapy controls most seizures
- AEDs can be withdrawn after cyst resolution if no seizure recurrence
- Calcified lesions may cause chronic epilepsy requiring indefinite AED therapy
11. INTRAVENTRICULAR NCC — ADVANCED SURGICAL CONSIDERATIONS
The 2025 systematic review and meta-analysis (Mendieta-Barrera et al., Clin Neurol Neurosurg, 2025 — PMID: 40381508) supports neuroendoscopic management as the primary modality:
- Minimally invasive; direct cyst visualization and removal
- Avoids the need for open craniotomy
- For 4th ventricle cysts, approach choice depends on surgeon experience
- Antiparasitic therapy after surgical removal (not before — to avoid precipitating inflammatory hydrocephalus)
12. COMPLICATIONS
| Complication | Mechanism |
|---|---|
| Chronic epilepsy | Perilesional gliosis and calcifications |
| Obstructive hydrocephalus | Intraventricular cyst obstruction — can be acute and fatal |
| Communicating hydrocephalus | Basal arachnoiditis from subarachnoid NCC |
| Cerebral venous thrombosis | Rare; inflammatory |
| Stroke/TIA | Vasculitis from basal cysts inflaming perforating arteries |
| Cognitive decline | Chronic inflammation, multiple calcifications |
| Sudden death | Acute hydrocephalus from mobile intraventricular cyst (especially 4th ventricle) |
| Movement disorders | Rare; chorea, tremor — per 2025 systematic review (PMID: 40822429) |
13. DIFFERENTIAL DIAGNOSIS
| Condition | Distinguishing Features |
|---|---|
| Tuberculoma | No scolex; may have central calcification ("target sign"); AFB history/exposure |
| Brain abscess | Fever, elevated CRP/ESR; restricted diffusion center on DWI |
| Primary CNS lymphoma | Immunocompromised host; periventricular location; EBV PCR+ in CSF |
| Toxoplasmosis | HIV/immunosuppressed; multiple ring-enhancing at grey-white junction; responds to empiric treatment |
| Pyogenic abscess | Fever, systemic sepsis; DWI restricted; surrounding vasogenic edema |
| Metastatic tumors | Known primary; older age; multiple nodular enhancements |
| Low-grade glioma | No ring enhancement; mass effect; progressive on imaging |
14. PREVENTION
- Access to clean water and sanitation
- Handwashing before food preparation
- Thorough cooking of pork (kills cysticerci)
- Treatment of tapeworm carriers (praziquantel) to break the transmission cycle
- Mass drug administration programs in endemic communities
- Pig vaccination and treatment in endemic areas (veterinary public health)
- Improved pig husbandry — preventing pigs from accessing human feces
15. RECENT EVIDENCE (2023–2025)
| Evidence | Key Finding | PMID |
|---|---|---|
| Systematic review — ring-enhancing lesions (2024) | NCC is a leading etiology of multiple ring-enhancing brain lesions globally | 37768475 |
| Systematic review — movement disorders in NCC (2025) | NCC is an under-recognized cause of chorea, tremor, and other hyperkinetic movement disorders | 40822429 |
| Systematic review + meta-analysis — intraventricular NCC (2025) | Neuroendoscopic removal is effective and safe; supports its use as first-line for accessible intraventricular cysts | 40381508 |
SUMMARY FRAMEWORK
INGESTION OF T. solium EGGS (fecal-oral)
↓
Oncospheres hatch → gut wall penetration
↓
Haematogenous spread to CNS
↓
Cysticercus forms (viable, silent)
↓
Parasite begins to die → immune activation
↓
Perilesional inflammation / edema
↓
CLINICAL DISEASE (seizures, ICP, deficits)
↓
Cyst dies → calcification
↓
Chronic epilepsy or resolution
Key diagnostic tool: MRI showing a "cyst with a dot" (scolex) = pathognomonic
Key treatment principle: Antiparasitic therapy is tailored to stage and location; never give antiparasitics in cysticercal encephalitis or ocular NCC; always add corticosteroids when treating viable cysts
Sources: Bradley and Daroff's Neurology in Clinical Practice; Harrison's Principles of Internal Medicine 22E; Goldman-Cecil Medicine; Tintinalli's Emergency Medicine; Grainger & Allison's Diagnostic Radiology; Katzung's Basic and Clinical Pharmacology 16E; Washington Manual of Medical Therapeutics; IDSA/ASTMH 2017 NCC Guidelines (White et al., Clin Infect Dis)
This is a shared conversation. Sign in to Orris to start your own chat.