Physiology of sleep
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive material from Harrison's, Adams & Victor's, and Miller's Anesthesia. Here is a thorough synthesis:
Physiology of Sleep
1. Definition and Overview
Sleep is an active, reversible state of unconsciousness in which the brain is relatively more responsive to internal than external stimuli. It is not a passive phenomenon — the brain is highly active during sleep, particularly during REM sleep. Modern understanding is based on polysomnography (PSG), which continuously records the EEG, electrooculogram (EOG), and surface EMG to define sleep and wakefulness.
— Miller's Anesthesia, p. 781; Harrison's, p. 253
2. Sleep Stages
Sleep is divided into two fundamentally distinct states:
NREM Sleep (Non-Rapid Eye Movement)
NREM sleep is further subdivided into three stages (N1–N3), characterized by progressive slowing of cortical EEG and increasing arousal threshold:
| Stage | EEG Features | Features |
|---|---|---|
| N1 (Stage 1) | Low-voltage, mixed frequency; loss of alpha waves | Drowsiness; slow, roving eye movements; muscle relaxation |
| N2 (Stage 2) | Sleep spindles (12–14 Hz bursts); K-complexes (vertex waves) | True sleep; higher arousal threshold |
| N3 (Stage 3–4 / SWS) | Predominantly delta (slow-wave) activity | Deepest sleep; very high arousal threshold; growth hormone released |
REM Sleep (Rapid Eye Movement)
- Low-amplitude, mixed-frequency EEG (similar to N1)
- Rapid eye movements in bursts (EOG)
- Muscle atonia — near-complete abolition of skeletal muscle EMG activity (except respiratory muscles), mediated by brainstem
- Vivid dreaming occurs predominantly here
- Cerebral blood flow and oxygen consumption increase during REM (vs. decrease in NREM)
— Harrison's, p. 253; Adams & Victor's, p. 411
3. Sleep Architecture
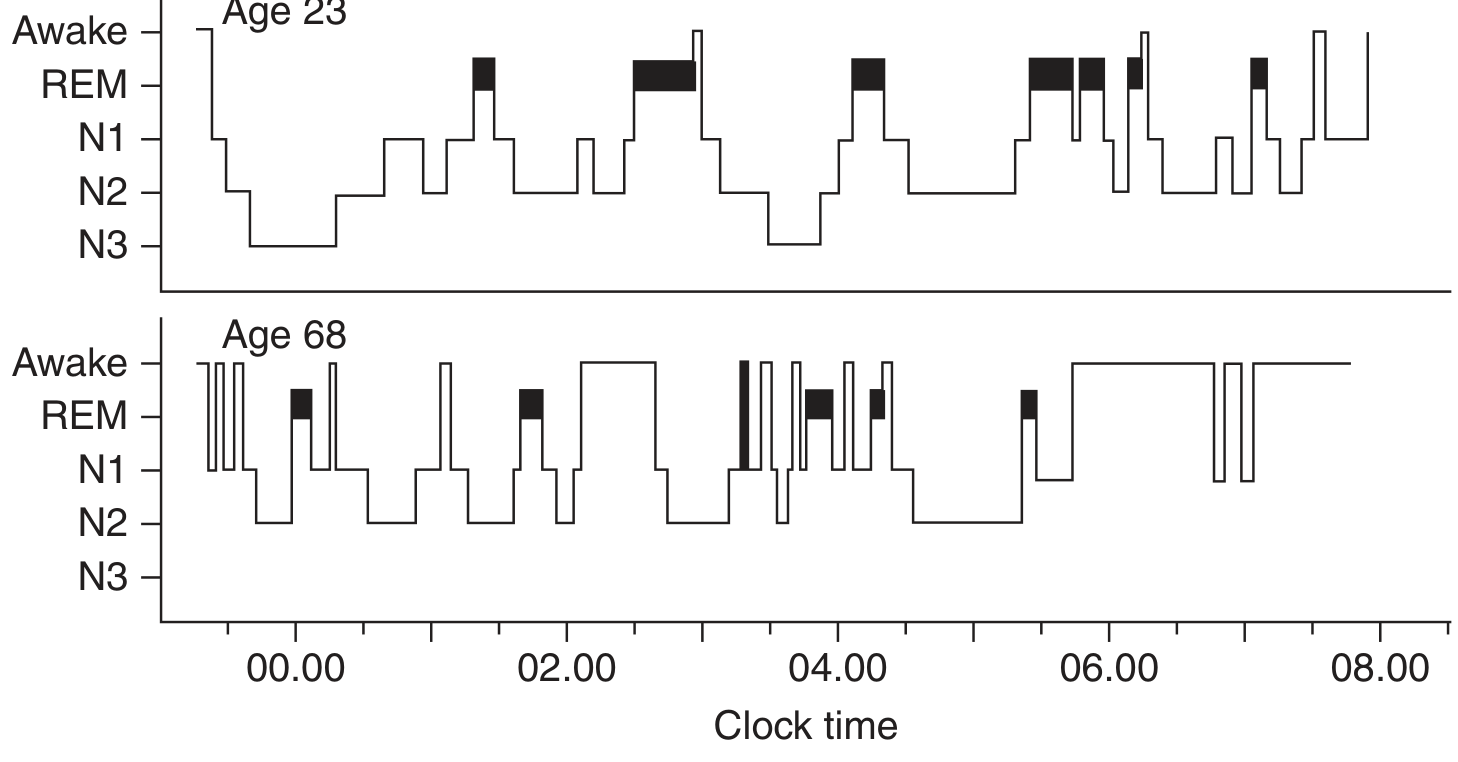
Figure: Sleep hypnogram showing NREM (N1–N3) and REM cycles across the night. Young adults show prominent N3 (slow-wave) sleep early in the night; REM sleep dominates the second half. Older adults show markedly reduced N3 and frequent awakenings.
Normal nocturnal sleep in a healthy young adult:
- Sleep onset → progresses through N1→N2→N3 within 45–60 min
- N3 (slow-wave sleep) predominates in the first third of the night (15–25% of total sleep)
- First REM episode appears after ~90 min
- NREM and REM alternate in ~60–160 min ultradian cycles (4–6 cycles per night)
- REM sleep constitutes 20–25% of total sleep time
- N1 + N2 constitute 50–60%
After sleep deprivation, NREM (especially SWS) recovers first; REM recovers second.
— Harrison's, p. 253
4. Regulation of Sleep: Two-Process Model
Sleep timing and duration are governed by two interacting systems:
Process C — Circadian Drive
- Generated by the suprachiasmatic nucleus (SCN) of the hypothalamus — the "master clock"
- The intrinsic period is ~25 hours; light-dark cycles entrain it to 24 h via retinal inputs
- Governs the timing of sleep; produces a circadian wake-promoting signal in the evening
- SCN integrates melatonin (from the pineal gland) as a signal of darkness
- Lesions of the SCN disorganize sleep-wake cycles and other cyclic rhythms (temperature, feeding, activity)
Process S — Homeostatic Drive
- "Sleep pressure" accumulates continuously during wakefulness
- It can be partially offset by the circadian wake-promoting signal
- Once sleep pressure becomes overwhelming, sleep is initiated
- SWS (N3) reflects the primary homeostatic discharge — slow-wave activity is proportional to prior waking duration
— Miller's Anesthesia, p. 782; Adams & Victor's, p. 410
5. Neural Circuits of Sleep–Wake Control: The Flip-Flop Switch
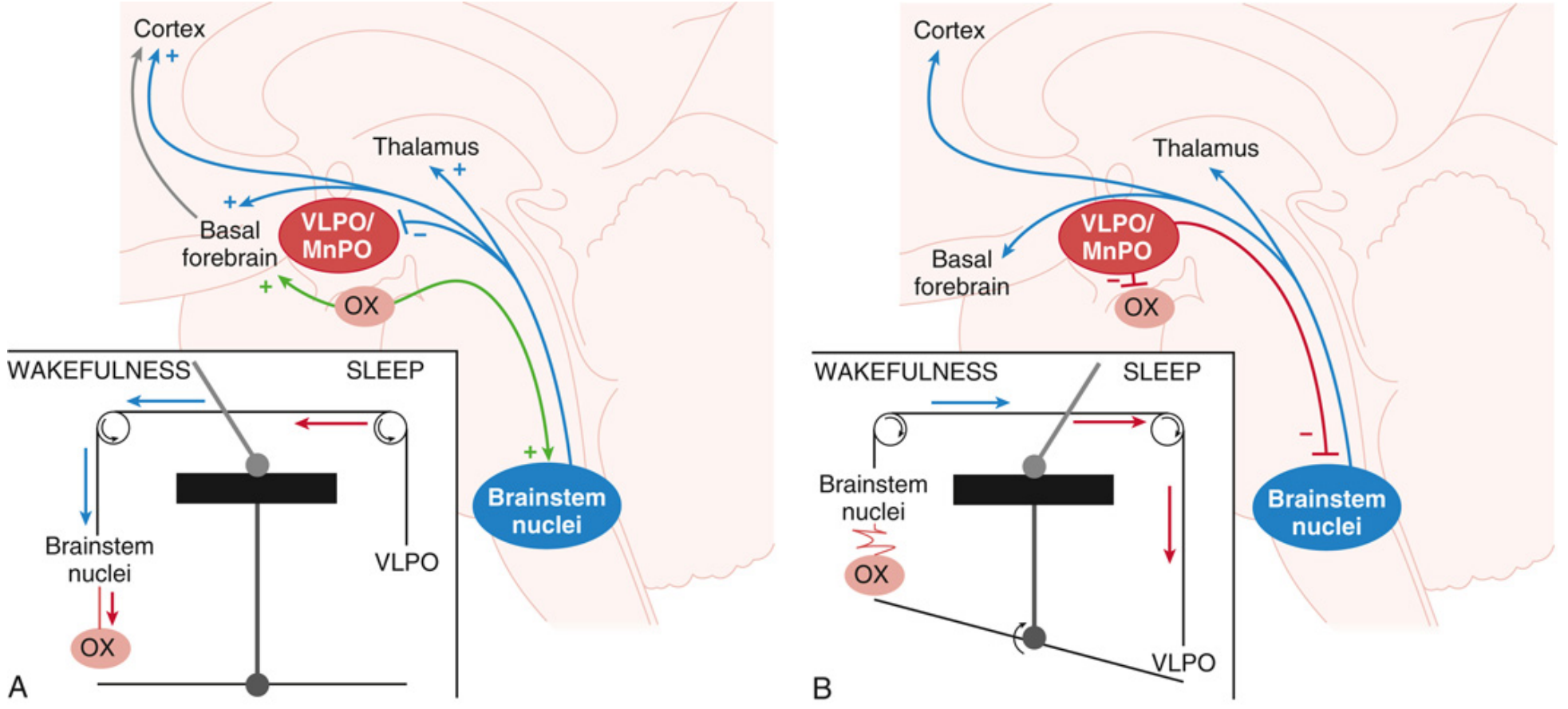
Figure: (A) During wakefulness, brainstem arousal nuclei activate the thalamus, basal forebrain, and cortex; orexin (OX) reinforces this. (B) During sleep, VLPO/MnPO neurons inhibit the arousal system and orexinergic neurons, flipping the switch to sleep.
Ascending Arousal System (Wake-ON)
Clusters of neurons extending from the upper pons to the hypothalamus and basal forebrain, using:
- Norepinephrine (locus coeruleus)
- Serotonin (dorsal raphe nuclei)
- Histamine (tuberomammillary nucleus, TMN)
- Dopamine
- Acetylcholine (pedunculopontine and laterodorsal tegmental nuclei)
- Glutamate
- Some GABAergic basal forebrain neurons disinhibit the cortex
These neurons activate the cortex directly and via the thalamus.
Orexin (Hypocretin) — The Stabilizer
- Wake-promoting peptide from neurons in the lateral hypothalamus
- Reinforces activity of all arousal-promoting cell groups (especially TMN via OX2R)
- Stabilizes the flip-flop switch — preventing inappropriate transitions into sleep
- Loss of orexin neurons → narcolepsy
Sleep-Promoting System (Wake-OFF)
- Ventrolateral preoptic nucleus (VLPO) and median preoptic nucleus (MnPO) — key sleep centers
- Use GABA (and galanin) to inhibit arousal neurons
- VLPO neurons fire at higher frequency during sleep; they are themselves inhibited by arousal neurons
- This mutual inhibition = the flip-flop switch → promotes rapid, complete state transitions (no intermediate "fuzzy" states)
- Melanin-concentrating hormone (MCH) neurons in the lateral hypothalamus also promote REM sleep
NREM–REM Switch
A second flip-flop switch in the brainstem:
- REM-ON (sublaterodorsal nucleus): GABAergic + glutamatergic neurons promote REM; drive atonia via descending projections to spinal motor neurons
- REM-OFF (ventrolateral periaqueductal gray + lateral pontine tegmentum): inhibit the REM-ON neurons
- Mutual inhibition generates sharp NREM↔REM transitions
— Harrison's, p. 254; Miller's Anesthesia, p. 793–796; Adams & Victor's, p. 412
6. Physiological Changes During Sleep
Cardiovascular
- Blood pressure and heart rate fall during NREM/SWS ("nocturnal dipping") — loss of this dip (e.g., in sleep apnea) is associated with daytime hypertension and cardiovascular risk
- During REM: sympathetic tone increases, causing blood pressure and heart rate fluctuations
Respiratory
- Breathing becomes more regular during NREM; more irregular during REM
- Thermoregulation is maintained during NREM but suspended during REM — shivering, panting, and sweating are impaired in REM
Renal
- Urine output decreases; urine osmolality increases (due to elevated ADH secretion)
Endocrine
- Growth hormone (GH): large surge in the first 1–2 h of sleep, tied to slow-wave sleep
- Cortisol: lowest at sleep onset; peaks near awakening
- TSH: peaks at sleep onset
- Melatonin: secreted in darkness by the pineal gland; suppressed by light
- Prolactin: rises during sleep; highest shortly after sleep onset
- LH/Testosterone: sleep-associated LH surges occur during puberty
Neurological
- Cerebral blood flow and metabolism are reduced across the brain during NREM, and increase during REM
- Parasympathetic outflow is periodically activated in REM
- Penile/clitoral erections occur periodically during REM
— Adams & Victor's, p. 412
7. Effects of Age on Sleep
| Age | Sleep Pattern |
|---|---|
| Newborn | 16–20 h/day; ~50% REM |
| Child | 10–12 h; gradual consolidation |
| Young adult | 7–7.5 h; N3 prominent; REM ~25% |
| Older adult (>65) | 6–6.5 h; N3 markedly reduced or absent; frequent awakenings; fragmented sleep |
- VLPO neurons are progressively lost during normal aging, correlating with sleep fragmentation
- Also lost in Alzheimer's disease, contributing to severely disrupted sleep
— Harrison's, p. 254; Adams & Victor's, p. 411
8. Neurotransmitter Summary
| Neurotransmitter | Role in Sleep-Wake |
|---|---|
| Orexin/Hypocretin | Wake-promoting, stabilizes arousal |
| Histamine | Wake-promoting (TMN → cortex) |
| Norepinephrine | Wake-promoting (locus coeruleus) |
| Serotonin | Wake-promoting, modulates REM |
| Acetylcholine | Wake and REM-promoting |
| GABA | Sleep-promoting (VLPO → arousal nuclei) |
| Galanin | Sleep-promoting (co-released with GABA at VLPO) |
| Melanin-concentrating hormone | REM sleep promotion |
| Adenosine | Homeostatic sleep pressure accumulation |
| Melatonin | Circadian phase signal (darkness indicator) |
During wakefulness, monoaminergic (inhibitory) neurons are dominant; during NREM, aminergic inhibition declines and cholinergic excitation rises; REM sleep occurs when this shift is complete. — Adams & Victor's, p. 412
Key Clinical Points
- Sleep deprivation → impaired vigilance, slowed reaction time, glucose intolerance, immune dysfunction, increased cardiovascular risk, accelerated neurodegeneration
- Narcolepsy → loss of orexin neurons; inappropriate transitions into REM sleep
- Insomnia → often reflects dysfunction of VLPO or homeostatic pressure
- Sleep apnea → fragments sleep; abolishes nocturnal BP dip; elevates cardiovascular risk
Sources: Harrison's Principles of Internal Medicine 22E, p. 253–255; Adams & Victor's Principles of Neurology 12E, p. 410–414; Miller's Anesthesia 10E, p. 781–796
This is a shared conversation. Sign in to Orris to start your own chat.