Is intraductal calcification is noted in plain abdominal x ray in chronic pancreatitis
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Searching Images
intraductal calcification chronic pancreatitis plain abdominal x-ray
Yes — intraductal calcification is indeed visible on plain abdominal X-ray in chronic pancreatitis.
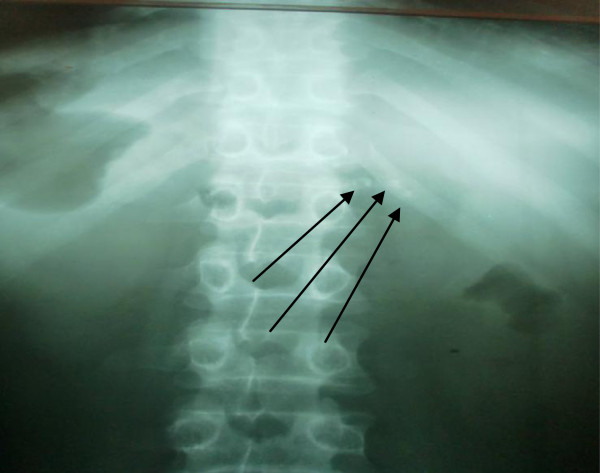
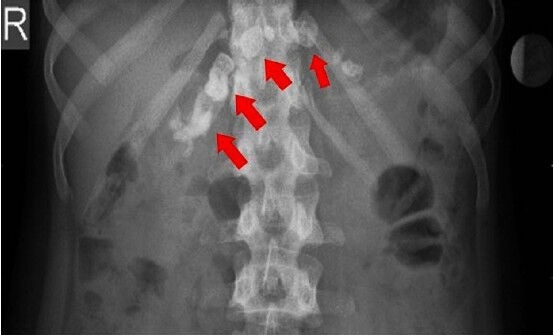
Pancreatic Calcification on Plain Abdominal X-ray in Chronic Pancreatitis
What is seen?
Plain abdominal radiographs can reveal pancreatic calcification in chronic pancreatitis. These appear as:
- Multiple discrete, punctate or coarse radiopaque densities located across the upper abdomen
- Distributed transversely across the midline, following the anatomical course of the pancreatic head, body, and tail
- Often described as a "chain-like" or stippled pattern at the L1–L2 vertebral level
These opacities represent either:
- Intraductal calculi (stones within the main pancreatic duct — true intraductal calcification)
- Parenchymal calcification (calcium deposits in the pancreatic parenchyma)
Both may co-exist and can be visible on plain film.
Clinical Significance
- The presence of pancreatic calcification on plain X-ray is highly suggestive of chronic pancreatitis, particularly chronic alcohol-related pancreatitis
- It indicates advanced/late-stage disease with long-standing inflammation and fibrosis
- It is considered one of the most specific plain radiographic findings for chronic pancreatitis
- In the CT era, a combination of parenchymal calcifications + intraductal calcification + parenchymal atrophy + cystic lesions (≥3 of 4) allows diagnosis with high specificity
Differentiating from Other Calcifications
| Feature | Pancreatic calcification | Renal calculi | Gallstones |
|---|---|---|---|
| Location | Midline, L1–L2 level, transverse | Lateral, along renal outline | Right upper quadrant |
| Pattern | Chain-like, stippled | Discrete, unilateral | Faceted/laminated |
| Crossing midline | Yes | No | No |
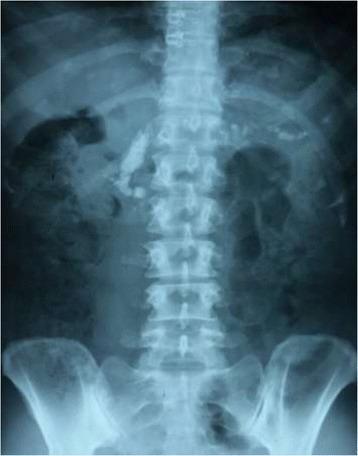
Radiographic Images
Here are actual plain abdominal X-rays demonstrating intraductal/parenchymal calcification in chronic pancreatitis:
Summary
Yes, intraductal calcification is visible on plain abdominal X-ray in chronic pancreatitis. While plain X-ray has lower sensitivity than CT (which can detect smaller calcifications), visible pancreatic calcification on a plain film is a highly specific finding for chronic pancreatitis, particularly the alcohol-related type, and indicates advanced disease.
Sources: Grainger & Allison's Diagnostic Radiology; Sleisenger and Fordtran's Gastrointestinal and Liver Disease; Schwartz's Principles of Surgery, 11th Edition
Staging of pancreatic carcinoma
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Here is a comprehensive account of pancreatic carcinoma staging, covering both the AJCC TNM system and the clinically critical resectability-based classification.
Staging of Pancreatic Carcinoma
There are two complementary staging systems used in practice:
- AJCC TNM Staging — provides prognostic grouping
- NCCN Resectability-based Staging — guides surgical decision-making
1. AJCC TNM Staging (8th Edition)
T — Primary Tumour
| Category | Description |
|---|---|
| TX | Primary tumour cannot be assessed |
| T0 | No evidence of primary tumour |
| Tis | Carcinoma in situ |
| T1 | Tumour ≤ 2 cm in greatest dimension (limited to pancreas) |
| T2 | Tumour > 2 cm but ≤ 4 cm in greatest dimension |
| T3 | Tumour > 4 cm; OR invading the duodenum or common bile duct |
| T4 | Tumour invading the celiac axis, SMA, and/or common hepatic artery; OR adjacent organs (stomach, spleen, colon, adrenal gland) |
Key change in 8th edition: T staging is now purely size-based for T1–T3. T4 is defined by arterial involvement (celiac, SMA, common hepatic artery), making it inherently unresectable.
N — Regional Lymph Nodes
| Category | Description |
|---|---|
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Regional (peripancreatic) lymph node metastasis |
Note: Nodal staging has limitations on imaging, as nodes can be involved without being enlarged.
M — Distant Metastasis
| Category | Description |
|---|---|
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| M1a | Metastasis confined to liver |
| M1b | Metastases in ≥1 extrahepatic site (lung, ovary, non-regional nodes, peritoneum, bone) |
| M1c | Both hepatic and extrahepatic metastases |
Stage Grouping
| Tumour | Node | Metastasis | Stage |
|---|---|---|---|
| T1 | N0 | M0 | Stage I |
| T2 | N0 | M0 | Stage II |
| T3 | N0 | M0 | Stage II |
| T4 | N0 | M0 | Stage III |
| Any T | N1 | M0 | Stage III |
| Any T | Any N | M1 | Stage IV |
2. NCCN Resectability-based Classification
The AJCC TNM system alone does not indicate whether a tumour is surgically resectable. The NCCN classification based on CT vascular anatomy is essential for clinical management. Patients are stratified into four groups:
Resectable
| Relationship | Criteria |
|---|---|
| Tumour–artery | No contact with CA, SMA, or CHA |
| Tumour–vein | No SMV/PV contact, OR contact ≤ 180° without contour irregularity |
| Metastatic disease | Absent |
Borderline Resectable
| Relationship | Criteria |
|---|---|
| Tumour–artery | Contact ≤ 180° of SMA or CA; OR contact with CHA without extension to CA or hepatic bifurcation; OR contact with variant arterial anatomy |
| Tumour–vein | SMV/PV contact > 180°; OR ≤ 180° with contour irregularity; OR contact with IVC; OR short-segment SMV/PV thrombosis amenable to reconstruction |
| Metastatic disease | Absent |
Locally Advanced (Unresectable)
| Relationship | Criteria |
|---|---|
| Tumour–artery | Contact > 180° of SMA or celiac axis; OR extensive SMV/PV involvement without proximal/distal targets for vascular reconstruction |
| Metastatic disease | Absent |
SMA resection is not performed due to poor survival and high morbidity — hence >180° SMA involvement = unresectable.
Metastatic
- Evidence of spread to liver, peritoneum, or lung (most common sites)
- No surgical resection of primary tumour
Key Clinical Points
- Only ~15–20% of patients present with resectable disease at diagnosis
- Diagnostic laparoscopy identifies occult metastases in ~17% of patients who appear resectable on imaging — these are typically 1–2 mm liver/peritoneal lesions invisible on CT
- The "tear-drop deformity" of the SMV/PV on CT is a specific sign of venous infiltration
- Tumour–vessel contact > 180° circumference indicates vascular involvement; < 90° indicates low probability
- Splenic artery involvement is not T4 (it is resected with the pancreas in distal pancreatectomy)
- Pancreatic calcification with focal mass on CT should raise concern for coexisting cancer in chronic pancreatitis (up to 16-fold increased risk)
Sources: Schwartz's Principles of Surgery, 11th Ed. | Grainger & Allison's Diagnostic Radiology | Mulholland & Greenfield's Surgery, 7th Ed. | Current Surgical Therapy, 14th Ed.
Ix used in pancreatic carcinoma staging
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Here is a comprehensive breakdown of the investigations used in staging pancreatic carcinoma:
Investigations Used in Staging Pancreatic Carcinoma
1. Multidetector CT (MDCT) — Gold Standard
The pancreatic protocol MDCT (contrast-enhanced, dual-phase) is the most widely used and most important investigation for staging.
Technique:
- Contrast-enhanced CT with pancreatic parenchymal + venous phase acquisition
- Thin-collimation, high-resolution slices
- Individualised bolus-tracking delay (aortic time + 25 seconds) is superior to a fixed 40-second delay
- 3D reconstructions using MPR, MIP, and VR to assess peripancreatic vasculature
What it assesses:
| Parameter | CT Finding |
|---|---|
| Primary tumour | Poorly enhancing hypodense mass in densely enhancing pancreas |
| Ductal dilatation | Upstream MPD/CBD dilation ("double duct sign" in head tumours) |
| Vascular involvement | Tumour–vessel interface, % circumference of contact with SMA/SMV/CA/CHA/PV |
| Lymph nodes | Nodes ≥10 mm short axis (but sensitivity is only ~14% for nodal metastases) |
| Liver metastases | Low-density poorly marginated lesions on portal venous phase |
| Peritoneal spread | Detected but small implants often missed |
Performance:
- Predictive value for unresectability: 90–100%
- Predictive value for resectability: 76–90%
- Sensitivity for masses < 2 cm: ~77% (limitation)
Secondary signs of isoattenuating tumour:
- Focal MPD dilation with abrupt cut-off
- Upstream pancreatic atrophy
- Biliary obstruction
- Unusual pancreatic contour
2. Transabdominal Ultrasound (US)
- Usually the first investigation ordered (inexpensive, widely available)
- Highly accurate in differentiating obstructive vs. non-obstructive jaundice
- Can detect small tumours in the head with expertise
- NOT sufficient for staging — cannot assess extrapancreatic spread, vascular encasement, or peritoneal disease reliably
- Negative US does not rule out pancreatic cancer (especially body/tail tumours)
3. Endoscopic Ultrasound (EUS)
- Accuracy of 97.6% for diagnosing malignancy
- Key roles:
- Biopsy of equivocal or non-obstructing lesions (EUS-guided FNA/FNB)
- Defines anatomical relationship between tumour and peripancreatic vessels
- Detects small tumours missed on CT (especially < 2 cm)
- Assesses local nodal disease
- Particularly useful when CT is non-diagnostic or when tissue confirmation is needed before neoadjuvant therapy
4. MRI / MRCP
- Theoretical advantages over CT for small tumour detection and characterisation
- However, most studies show no significant advantage over MDCT in practice; less accessible and more costly
- MRCP is valuable for:
- Detailed ductal anatomy (strictures, filling defects)
- Excluding obstructing tumour when CT is ambiguous
- Gadolinium-enhanced MRI + DWI helps exclude malignancy as cause of ductal dilation when CT is equivocal
- If ERCP has been done (causing artefact/cholangitis), MRI can still assess the biliary system
- Uses identical vascular criteria to CT for resectability assessment
5. ERCP
- Still used for cytological sampling and biliary stent insertion in obstructive jaundice
- Limitation for staging: prior ERCP + stent insertion + cholangitis degrades the quality of subsequent MDCT/MRI, making detection of small tumours and assessment of longitudinal spread more difficult
- Therefore, "exploratory" ERCP without prior cross-sectional imaging should be avoided
6. Contrast-Enhanced Ultrasound (CEUS)
- High value for characterisation and differentiation of ductal adenocarcinoma from other pancreatic diseases
- Useful for cystic pancreatic lesions
- A recent meta-analysis confirmed its utility for distinguishing adenocarcinoma from chronic pancreatitis/focal masses
7. FDG PET/CT
- Limited role in initial staging
- Cannot reliably differentiate malignant from inflammatory pancreatic masses
- Low sensitivity for liver metastases
- Main current use: detection of local recurrence after treatment (high accuracy)
8. Diagnostic / Staging Laparoscopy
- Identifies occult metastases (liver/peritoneal) in ~17% of patients who appear resectable on imaging
- These are typically 1–2 mm implants below CT resolution
- Patients who avoid unnecessary laparotomy start palliative chemotherapy significantly earlier (17.9 vs. 39.9 days)
- Recommended in treatment-naïve patients before planned resection
Summary Table
| Investigation | Primary Role in Staging | Key Limitation |
|---|---|---|
| MDCT (pancreatic protocol) | Gold standard — resectability, vascular involvement, metastases | Misses isoattenuating tumours, tiny liver mets |
| Transabdominal US | First-line, detects jaundice aetiology | Not adequate for staging |
| EUS ± FNA | Tissue diagnosis, small tumours, local vascular anatomy | Operator-dependent |
| MRI/MRCP | Ductal anatomy, equivocal CT cases | No clear advantage over MDCT in most studies |
| ERCP | Cytology, biliary stenting | Degrades subsequent CT/MRI quality |
| CEUS | Characterisation, differentiation from pancreatitis | Limited availability |
| FDG PET/CT | Recurrence detection | Poor at distinguishing Ca from inflammation |
| Staging laparoscopy | Detects occult peritoneal/liver metastases | Invasive |
Sources: Grainger & Allison's Diagnostic Radiology | Yamada's Textbook of Gastroenterology, 7th Ed. | Current Surgical Therapy, 14th Ed.
. A 70-year-old man with diabetes mellitus,ischemic heart disease and a history of heavy smoking presents with episodes of severe central abdominal pain soon after meals. He has lost 5kg over last 3 months despite a normal appetite.Abdominal examination is unremarkable. What is the most likely diagnosis? A) Gallstone disease B) Acute gastritis C) Acute relapsing pancreatitis D) Gastric carcinoma E) Mesenteric ischaemia
Answer: E) Mesenteric Ischaemia
Why Mesenteric Ischaemia?
This clinical vignette is a classic presentation of chronic mesenteric ischaemia (intestinal angina). Let's analyse each clue:
Risk Factor Profile — Points Strongly to Vascular Disease
| Risk Factor | Relevance |
|---|---|
| Age 70 | Advanced atherosclerosis |
| Diabetes mellitus | Accelerated atherosclerosis |
| Ischaemic heart disease | Confirms established atherosclerotic disease |
| Heavy smoking | Major risk factor for mesenteric artery stenosis |
This is a patient with widespread atherosclerosis — the same disease process affecting his coronary arteries is affecting his mesenteric vessels.
Classic Triad of Chronic Mesenteric Ischaemia
| Feature | This Patient |
|---|---|
| Postprandial abdominal pain ("intestinal angina") | ✅ Severe central pain soon after meals |
| Weight loss despite normal appetite | ✅ 5 kg over 3 months — patient fears eating ("sitophobia") |
| Unremarkable abdominal examination | ✅ Classic — "pain out of proportion to examination" |
The mechanism is identical to cardiac angina: increased metabolic demand (gut digestion requires increased blood flow) in the setting of fixed stenosis of mesenteric vessels → ischaemic pain. At rest, collateral supply is adequate, hence the normal examination.
Why NOT the Other Options?
| Option | Against |
|---|---|
| A) Gallstone disease | Pain is typically right upper quadrant/colicky, not central; not consistently postprandial; no vascular risk factor link |
| B) Acute gastritis | Would not cause weight loss of 5 kg; not consistently postprandial; unremarkable exam expected but episodic pattern doesn't fit |
| C) Acute relapsing pancreatitis | Pain radiates to back, associated with nausea/vomiting; would expect tenderness on examination; no history of alcohol or gallstones mentioned |
| D) Gastric carcinoma | Weight loss fits, but pain is not characteristically immediately postprandial; appetite is described as normal (gastric Ca causes anorexia); examination would likely reveal a mass or epigastric tenderness |
Key Distinguishing Feature
Weight loss WITH normal appetite is the hallmark of mesenteric ischaemia — the patient is hungry but afraid to eat because eating triggers pain (sitophobia). In gastric carcinoma, weight loss is accompanied by loss of appetite.
Diagnosis & Investigation
- Duplex Doppler USS — first-line screening (assesses celiac/SMA flow)
- CT angiography — investigation of choice to confirm stenosis/occlusion of celiac axis and SMA
- Digital subtraction angiography (DSA) — gold standard, also allows intervention
Treatment
- Endovascular stenting of the stenosed mesenteric vessels (preferred)
- Surgical revascularisation (bypass) in suitable candidates
- Optimise vascular risk factors (antiplatelet, statin, smoking cessation)
This is a shared conversation. Sign in to Orris to start your own chat.