Guidlines and treatment of osteoporosis
Reading File
Finding Sources
Searching PubMed
"osteoporosis"[MeSH Terms] AND "treatment guidelines"
Searching the Web
osteoporosis treatment guidelines 2024 2025 pharmacological management
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Searching Images
osteoporosis treatment algorithm pharmacological management flowchart
I have two excellent management flowchart images. I now have comprehensive content from Harrison's 22E (2025), Goldman-Cecil, web guidelines (NOGG 2024, ACP 2023, AACE), and relevant diagrams to synthesize a thorough answer.
Osteoporosis: Guidelines and Treatment
Definition & Diagnosis
Osteoporosis is a skeletal disorder characterized by compromised bone strength that predisposes to an increased risk of fracture. The WHO defines it as bone mineral density (BMD) T-score ≤ −2.5 standard deviations below young-adult mean. Osteopenia is T-score between −1.1 and −2.4.
Dual-energy X-ray absorptiometry (DXA) at the femoral neck and lumbar spine (L1–L4) is the gold-standard diagnostic tool. T-scores use NHANES reference values for women aged 20–29, applied equally to men.
Risk Assessment: FRAX
FRAX (Fracture Risk Assessment Tool) estimates the 10-year probability of a major osteoporotic fracture (hip, vertebral, forearm, humerus) and is central to all current guidelines.
| FRAX Threshold (USA) | Action |
|---|---|
| 10-year major fracture risk >20% | Treat |
| 10-year hip fracture risk >3% | Treat |
| Low risk | Lifestyle measures, reassess in 2–4 years |
Key clinical risk factors entered into FRAX include: age, sex, BMI, prior fragility fracture, parental hip fracture, glucocorticoid use, rheumatoid arthritis, secondary osteoporosis, smoking, and alcohol intake (≥3 units/day).
Indications for DXA Scanning
- Women ≥65 years and men ≥70 years (routine screening)
- Postmenopausal women and men ≥50 with clinical risk factors
- Individuals on long-term glucocorticoids (≥3 months)
- Any fragility fracture after age 50
- Vertebral imaging in women ≥70 or men ≥80 if T-score <−1.0
Non-Pharmacological Management (All Patients)
These apply universally across all risk categories:
- Calcium: 1,000 mg/day (age 19–50); 1,200 mg/day (women >51, men >70) — dietary preferred, supplement if deficient
- Vitamin D: 800–1,000 IU/day (target serum 25(OH)D >50 nmol/L); up to 2,000 IU/day in deficient patients
- Weight-bearing and resistance exercise (reduces fall risk and improves bone density)
- Fall prevention: balance training, home hazard assessment, vision correction
- Smoking cessation and alcohol reduction (<2 units/day)
- Avoid immobility and optimize nutrition (adequate protein)
Pharmacological Treatment
Overview
Pharmacologic therapies are classified as antiresorptive or anabolic:
| Class | Agents |
|---|---|
| Antiresorptive | Bisphosphonates, Denosumab, SERMs (Raloxifene, Bazedoxifene), Estrogen therapy, Calcitonin |
| Anabolic | Teriparatide, Abaloparatide, Romosozumab (dual action) |
1. Bisphosphonates — First-Line Therapy
The 2023 American College of Physicians (ACP) guidelines reinforce bisphosphonates as the clear first-line treatment for postmenopausal women and men with osteoporosis (updated from earlier guidance that had treated denosumab as equivalent).
| Agent | Route | Dose | Frequency |
|---|---|---|---|
| Alendronate | Oral | 70 mg | Weekly |
| Risedronate | Oral | 35 mg or 150 mg | Weekly or monthly |
| Ibandronate | Oral / IV | 150 mg PO; 3 mg IV | Monthly / every 3 months |
| Zoledronic acid | IV | 5 mg | Annually |
Mechanism: Bind hydroxyapatite, inhibit osteoclast farnesyl pyrophosphate synthase → reduced bone resorption.
Fracture reduction:
- Vertebral fractures: ~50–70% relative risk reduction
- Hip fractures: ~40% (alendronate, zoledronic acid)
- Non-vertebral fractures: ~20–40%
Duration (NOGG 2024):
- Oral bisphosphonates: at least 5 years, then reassess
- IV bisphosphonates (zoledronate): at least 3 years, then reassess
- Extended therapy (beyond 5 years oral / 3 years IV) for: age ≥70, prior hip/vertebral fracture, high-dose glucocorticoids (≥7.5 mg prednisolone/day)
Drug holiday: After 5 years oral or 3 years IV in lower-risk patients, a temporary pause of 18–36 months can be considered, with reassessment.
Side effects:
- GI irritation (oral); esophageal ulceration if not taken properly
- Acute-phase reaction (flu-like) with first IV dose
- Rare: osteonecrosis of the jaw (ONJ), atypical femoral fractures (with prolonged use >5 years)
Contraindications: eGFR <35 mL/min (most bisphosphonates), esophageal dysmotility (oral forms), hypocalcemia.
2. Denosumab (Prolia) — Second-Line / High-Risk
Mechanism: Fully humanized monoclonal antibody against RANKL → inhibits osteoclastogenesis → ↓ bone resorption while allowing continued bone formation.
Dose: 60 mg SC every 6 months
Approved for: Postmenopausal osteoporosis, male osteoporosis, glucocorticoid-induced osteoporosis, and oncology (bone loss from hormonal therapies in breast/prostate cancer).
Advantages: Usable in renal impairment (unlike bisphosphonates); larger BMD gains than bisphosphonates; no fixed treatment duration.
Critical warnings:
- Do NOT stop denosumab abruptly — rebound increase in bone turnover occurs, leading to multiple vertebral fractures. Always transition to a bisphosphonate when stopping (NOGG 2024 recommendation #19).
- Before starting, ensure a long-term plan is in place.
- Risk of hypocalcemia, especially in patients with low GFR.
3. Anabolic Agents — For Very High-Risk Patients
Used when fracture risk is very high (T-score ≤ −3.0, multiple prior fractures, or failure of antiresorptives).
Teriparatide (PTH 1-34)
- Dose: 20 µg SC daily for up to 24 months
- Stimulates osteoblast activity → new bone formation
- Reduces vertebral fractures by ~65%, non-vertebral ~35%
- Must be followed by antiresorptive therapy to maintain gains
Abaloparatide (PTHrP analogue)
- Dose: 80 µg SC daily for up to 18 months
- Similar mechanism to teriparatide; may have a more cortical bone effect
- Followed by antiresorptive consolidation therapy
Romosozumab (Evenity) — Dual Action
- Mechanism: Anti-sclerostin monoclonal antibody → simultaneously stimulates bone formation AND inhibits resorption (dual action)
- Dose: 210 mg SC monthly for 12 months only
- Produces the largest 12-month BMD gains of any agent
- Followed by antiresorptive therapy (denosumab or bisphosphonate)
- Contraindicated in patients with prior MI or stroke within 12 months (cardiovascular signal)
4. Selective Estrogen Receptor Modulators (SERMs)
| Agent | Notes |
|---|---|
| Raloxifene | 60 mg/day; reduces vertebral fractures ~40%; no hip fracture benefit; used in women <60 or <10 years post-menopause without vasomotor symptoms; ↑ VTE risk |
| Bazedoxifene | Combined with conjugated estrogens (Duavee); bone protection + treats vasomotor symptoms |
5. Hormone Therapy (HT/MHT)
- Estrogen ± progestin reduces hip and spine fractures by 34% and all clinical fractures by 24% (Women's Health Initiative)
- Now used primarily in women <60 years or <10 years post-menopause with vasomotor symptoms where bone protection is an additional benefit
- Not recommended as first-line for osteoporosis treatment alone due to breast cancer, cardiovascular, and VTE risks
- Standard doses: conjugated equine estrogen 0.625 mg/day (oral) or estradiol 50 µg/day (transdermal)
6. Calcitonin
- Salmon calcitonin 200 IU nasal spray daily
- Modest antifracture benefit (vertebral only); analgesic benefit in acute vertebral fracture
- Now rarely used as a primary agent; largely superseded
Sequential / Combination Therapy Strategy
Current guidelines recommend a "treat-to-target" sequential approach for high and very high-risk patients:
- Very high risk: Start with anabolic agent (teriparatide/abaloparatide/romosozumab) first → follow with antiresorptive (bisphosphonate or denosumab) to consolidate gains
- High risk: Bisphosphonate or denosumab first-line; switch to anabolic if inadequate response
- Moderate risk: Oral bisphosphonate; consider drug holiday after 5 years
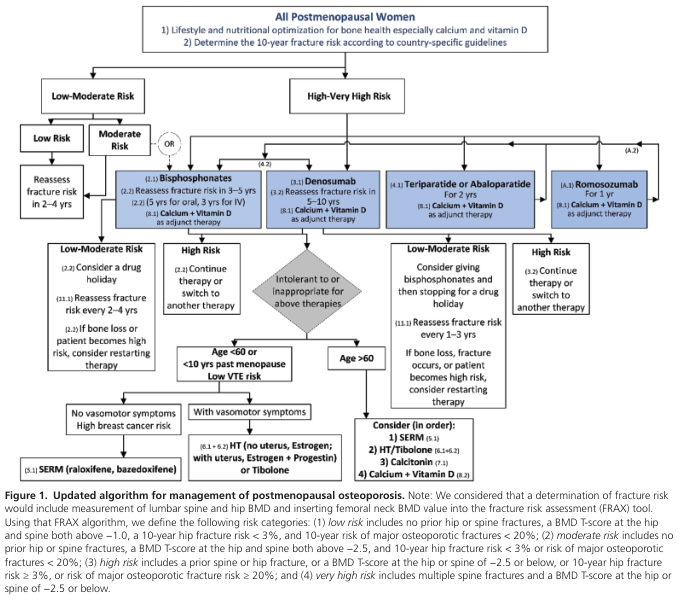
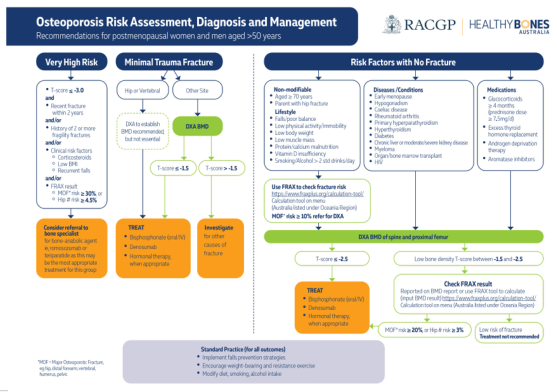
Special Populations
Glucocorticoid-Induced Osteoporosis (GIOP)
- Affects all patients on systemic glucocorticoids ≥7.5 mg prednisolone/day for ≥3 months
- Begin calcium and vitamin D supplementation for all
- Start bisphosphonate (alendronate or risedronate oral; zoledronate IV) based on FRAX-adjusted risk
- Denosumab and teriparatide are alternatives
- Glucocorticoids cause rapid bone loss — early intervention is key
Male Osteoporosis
- First-line: Oral bisphosphonates (alendronate or risedronate) — 2024 IOF/NOS guideline
- Second-line: Denosumab or zoledronate
- Very high risk: Sequential anabolic followed by antiresorptive
- Check testosterone, calcium, 25(OH)D, 24-hour urine calcium to rule out secondary causes
Renal Impairment
- Bisphosphonates generally avoid if eGFR <35 mL/min
- Denosumab is the preferred antiresorptive in CKD; monitor closely for hypocalcemia
- Teriparatide can be used
Monitoring
| Marker | Purpose | Timing |
|---|---|---|
| BMD (DXA) | Assess treatment response | 1–2 years after starting therapy |
| CTX-I (bone resorption) | Monitor antiresorptive adherence/response | Baseline; 3 months after starting |
| P1NP (bone formation) | Monitor anabolic agent response | Baseline; 1, 3, 6, 12 months |
| Serum calcium, vitamin D | Safety monitoring | Periodically |
For antiresorptive agents, a >56% decrease in CTX-I or >38% decrease in P1NP at 3 months defines treatment response.
Key Guideline Recommendations Summary
| Guideline | Key Points |
|---|---|
| ACP 2023 | Bisphosphonates are clear first-line for postmenopausal osteoporosis (de-emphasized denosumab equivalence) |
| NOGG 2024 (UK) | Oral bisphosphonates ≥5 years; IV ≥3 years; no abrupt denosumab discontinuation; FRAX-based thresholds |
| AACE 2020 | Risk-stratified approach; anabolics first for very high-risk; sequential therapy |
| IOF/NOS 2024 (Men) | Alendronate/risedronate first-line; denosumab/zoledronate second-line; anabolics for very high-risk men |
| Korean Society of Menopause 2024 | Treatment response monitoring with BTMs at 3 months; calcium 1,200 mg/day for women >51 |
Sources: Harrison's Principles of Internal Medicine, 22nd Ed. (2025); Goldman-Cecil Medicine, International Ed.; Lippincott Pharmacology; Rockwood & Green's Fractures in Adults, 10th Ed. (2025); NOGG 2024 Guidelines; ACP 2023; IOF/NOS Men Guidelines 2024. PubMed: PMID 40652262 (network meta-analysis of antiresorptive/anabolic agents, 2025).
This is a shared conversation. Sign in to Orris to start your own chat.