Give me a notes about irritable bowel syndrome
Reading File
Finding Sources
Searching PubMed
"irritable bowel syndrome"[MeSH Terms] AND management
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information to write detailed notes on IBS. Let me compile everything:
Irritable Bowel Syndrome (IBS)
Source: Goldman-Cecil Medicine, Sleisenger & Fordtran's GI and Liver Disease
Definition
IBS is a functional bowel disorder characterized by chronic or recurrent abdominal pain associated with alterations in stool form and/or frequency (diarrhea, constipation, or both). It is attributable to the middle or lower GI tract and exists on a spectrum with related disorders: functional constipation, functional diarrhea, and functional abdominal bloating/distention. Subtypes can transition over time in the same patient.
Epidemiology
| Parameter | Data |
|---|---|
| Global prevalence (Rome IV) | ~4.1% |
| US/UK/Canada prevalence | ~4.6% |
| Female vs male | 5.2% vs 2.9% |
| Incidence | ~38 per 10,000 person-years |
| Most common age | Under 50 years |
| Annual physician visits (US) | ~4.4 million |
Subtypes (Rome IV distribution):
- IBS-D (diarrhea-predominant): 35–40%
- IBS-M (mixed bowel habits): 35–40%
- IBS-C (constipation-predominant): ~25%
- IBS-U (unclassified): <5%
Up to 50% of sufferers never seek medical care.
Pathobiology
IBS is a multifactorial disorder of dysregulated gut-brain interaction (GBI), resulting in alterations across multiple domains:
1. Familial & Genetic Factors
- Family clustering: 1.75–2.75× higher odds in relatives of affected persons
- Susceptibility loci on chromosome 9q31.2 (especially in women)
- Six independent GWAS loci identified — four overlap with mood/anxiety disorders, reinforcing brain-gut axis importance
- Rare genetic variants (~2–3%): SCN5A channelopathy, sucrase-isomaltase variants, 5-HTTLPR (serotonin transporter), 5-HT3 receptor polymorphisms
2. Post-Infectious IBS
- Acute GI infection (bacterial, viral, or protozoal) increases IBS risk ~4-fold at 12 months
- Strongest risk factor for IBS-D
- Risk especially elevated in: younger patients, females, those with prior GERD, dyspepsia, anxiety, or depression
3. Stressful Life Events
- Physical, sexual, or emotional/verbal abuse
- Severe illness, parental death or mental illness, wartime exposure
4. Increased Visceral Perception
- Visceral stimuli are processed via primary afferents → dorsal horn → thalamus → cortical regions
- Peripheral sensitization: mediators (histamine, substance P) from mast cells/epithelial cells activate sensory nerves
- Central sensitization: central amplification of afferent input
- Patients show lowered thresholds to rectal/colonic balloon distension (visceral hypersensitivity) — seen in a subset of IBS patients
5. Altered CNS Processing
- Greater sensorimotor cortex volume and thickness (correlates with symptom severity)
- Alterations in resting-state insula/amygdala connectivity
- Decreased corticolimbic inhibitory feedback
- Greater engagement of salience detection and emotional arousal networks
6. Gut Transit & Motility
- Increased colonic motility during fasting, postprandially, and with stress
- ~50% of IBS-D patients have accelerated colon transit
7. Peripheral GI Factors
- Increased colonic mucosal nerves expressing substance P, TRPV1, protease-activated receptors
- Altered mucosal barrier: decreased tight junction protein expression → increased permeability → greater abdominal pain
- Mast cell activation: histamine release → afferent nerve activation
- Immune activation: mast cells, T lymphocytes
8. Gut Microbiota Dysbiosis
- Increased: Enterobacteriaceae, Lactobacillaceae, Bacteroides
- Decreased: Clostridiales I, Faecalibacterium, Bifidobacterium
- ~25% of IBS patients have bile acid diarrhea
9. Dysregulated Stress Responsiveness
- HPA axis dysregulation in IBS patients (vs. healthy controls)
- Stress increases visceral sensitivity, motility, permeability, and immune responses
Clinical Manifestations
Core symptom: Chronic or recurrent abdominal pain associated with diarrhea, constipation, or both.
Postprandial symptoms (in ~2/3 patients): pain, bloating, flatulence — triggered by large meals, high-fat/carbohydrate foods, coffee, alcohol, spicy foods.
Gender differences: Women more commonly have constipation; symptoms worsen premenstrually (declining estrogen/progesterone).
Extraintestinal associations:
- Temporomandibular joint disorder
- Interstitial cystitis/painful bladder syndrome
- Dysmenorrhea
- Migraine headaches
- Fibromyalgia
- Chronic fatigue syndrome
- Anxiety and depression
- Somatization
- Sleep disturbances
~1/3 of patients have coexisting functional dyspepsia or functional heartburn.
Diagnosis
Rome IV Diagnostic Criteria
Criterion A — Recurrent abdominal pain, ≥1 day/week on average in the last 3 months, with symptoms onset ≥6 months, plus two or more of:
- Related to defecation
- Associated with change in stool frequency
- Associated with change in stool form/appearance
Criterion B — Supportive symptoms:
- Stool frequency: ≤3/week or >3/day
- Abnormal stool form (lumpy/hard or loose/watery — Bristol Scale)
- Straining, urgency, incomplete evacuation, mucus, bloating/distention
Alarm Features ("Red Flags") — prompt targeted investigation:
- New-onset symptoms at age ≥50 years
- Unintentional weight loss
- Hematochezia or melena (not from hemorrhoids)
- Nocturnal diarrhea
- Anemia
- Palpable abdominal mass or lymphadenopathy
- Family history of colorectal cancer, IBD, or celiac disease
Diagnostic Testing
| Recommended | Not Recommended |
|---|---|
| CBC, CRP, fecal calprotectin/lactoferrin | Routine stool testing |
| Celiac serologies (IgA anti-tTG + total IgA) | Routine colonoscopy <45–50 yrs without alarm features |
| Colonoscopy if new onset ≥50 years | Food allergy/intolerance testing |
| Giardia stool antigen (endemic areas) | Lactose or glucose hydrogen breath tests |
| Bile acid diarrhea testing (IBS-D if suspected) | |
| Anorectal physiology (if pelvic floor dysfunction suspected) |
Differential diagnosis: Celiac disease, IBD (Crohn's, UC), microscopic/collagenous colitis, colorectal cancer, bile acid diarrhea, food intolerances.
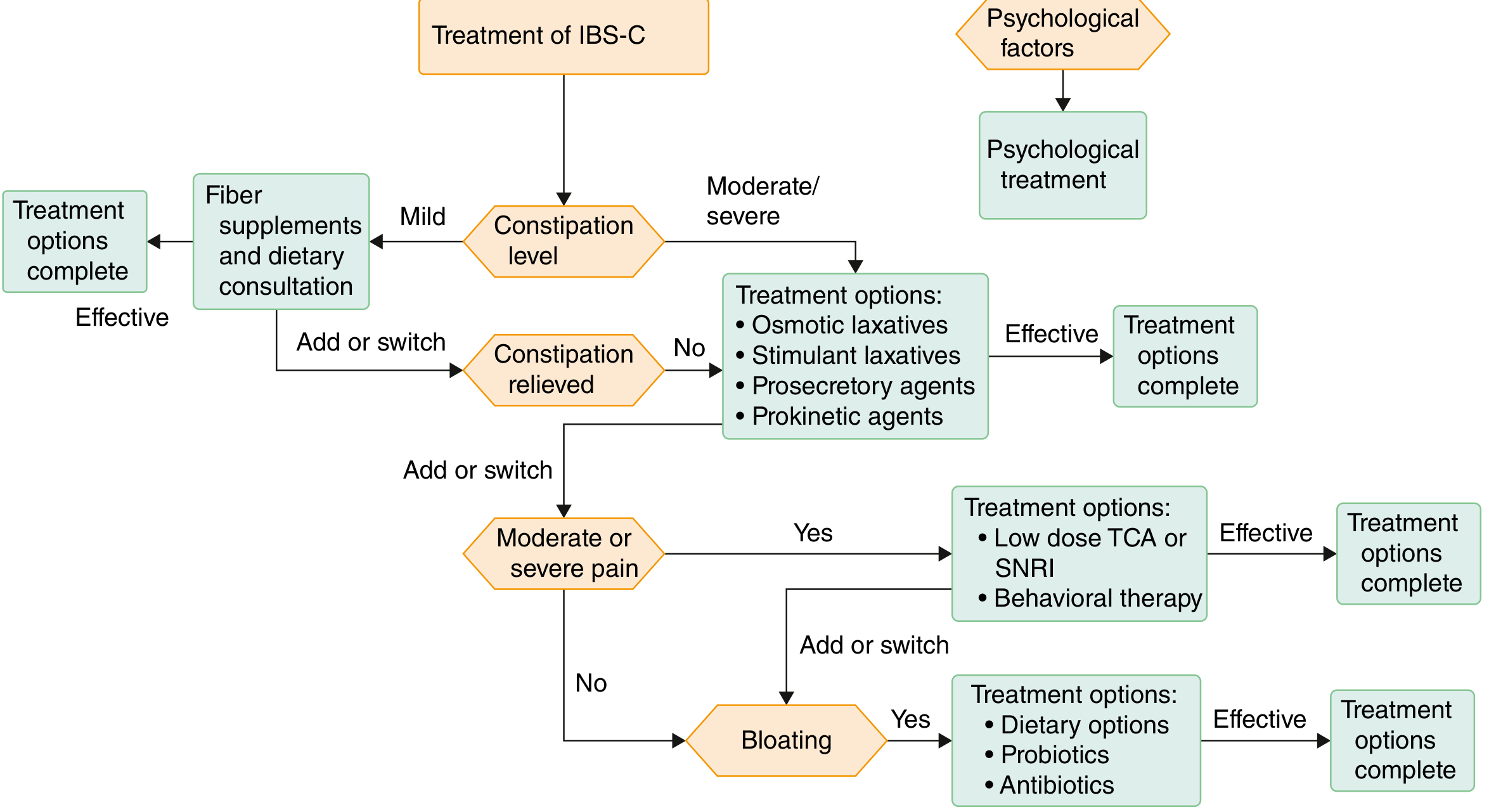
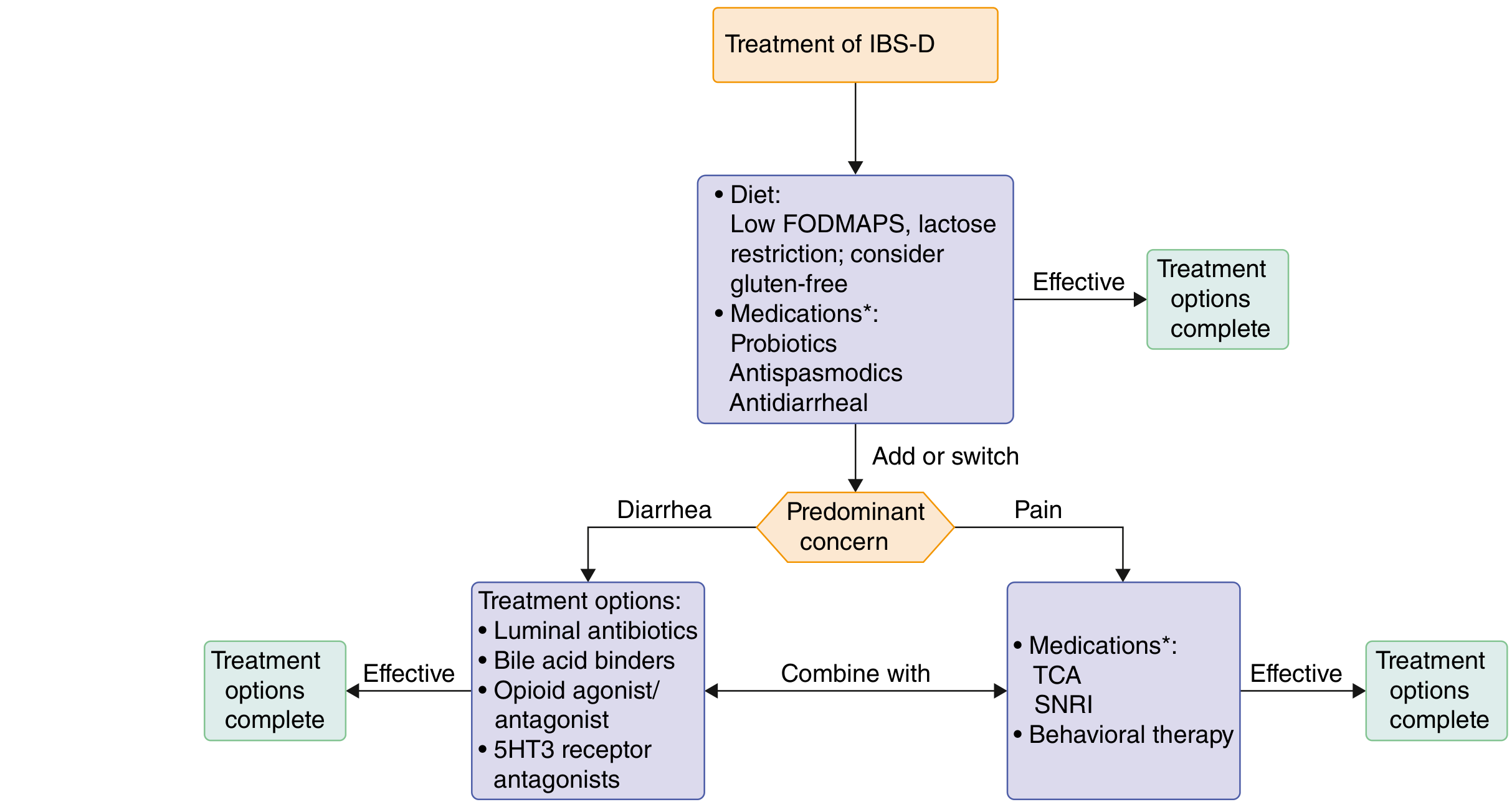
Treatment
Treatment is stratified by symptom severity and subtype:
- Mild: education, reassurance, dietary advice, OTC medications
- Moderate: add pharmacotherapy
- Severe: integrated pharmacologic + behavioral approach
Diet (First-Line)
- Low-FODMAP diet (fermentable Oligosaccharides, Disaccharides, Monosaccharides, and Polyols): improves global symptoms, especially in IBS-D — best supervised by a dietitian
- Soluble fiber (psyllium, up to 25–35 g/day): especially for IBS-C; start low, titrate up
- Insoluble fiber (wheat bran) is not recommended
- Keep a 1–2 week food/symptom diary to identify triggers
Pharmacotherapy — by symptom
For Pain/Spasm:
| Drug | Dose | Side Effects |
|---|---|---|
| Peppermint oil | 1–2 capsules TID | Heartburn, nausea |
| Hyoscyamine (antispasmodic) | 0.125 mg TID–QID | Dry mouth, blurred vision, constipation |
| Dicyclomine (antispasmodic) | 10 mg TID–QID | Dry mouth, blurred vision |
Central Brain-Gut Neuromodulators (used for pain, not necessarily for depression):
| Drug | Dose |
|---|---|
| Amitriptyline (TCA) | 10–25 mg QHS (up to 100 mg) |
| Desipramine / Nortriptyline (TCA) | 10–25 mg QHS (up to 100 mg) |
| SSRIs (citalopram, fluoxetine, paroxetine, sertraline) | Standard low doses |
For IBS-C:
| Drug | Mechanism | Dose |
|---|---|---|
| Polyethylene glycol (osmotic laxative) | Osmotic | 17 g daily |
| Lubiprostone | Cl⁻ channel activator | 8 μg BID with food |
| Linaclotide | Guanylate cyclase-C agonist | 290 μg daily |
| Plecanatide | Guanylate cyclase-C agonist | 3 mg daily |
| Tenapanor | NHE3 inhibitor (↓ Na/H₂O reabsorption) | 50 mg BID |
For IBS-D:
| Drug | Mechanism | Notes |
|---|---|---|
| Loperamide | μ-opioid agonist | Reduces stool frequency; does NOT improve global symptoms or pain |
| Eluxadoline | μ/κ-opioid agonist + δ-antagonist | 100 mg BID; CI: no gallbladder, hepatic impairment, pancreatic disease, alcohol use disorder |
| Rifaximin | Minimally absorbed antibiotic | 550 mg TID × 14 days; up to 3 retreatment courses |
| Alosetron | 5-HT3 antagonist | Reserved for severe IBS-D in women only; risk of ischemic colitis — REMS program |
| Bile acid sequestrants | Bind bile acids | If bile acid diarrhea confirmed/suspected |
Probiotics: Combination products may reduce global symptoms by ~20%; evidence is mixed — most useful for bloating/flatulence.
Psychological Interventions
Indicated for severe/refractory symptoms or significant psychological comorbidity. Associated with ~30% symptom reduction:
- Cognitive-behavioral therapy (CBT) — most evidence-based
- Gut-directed hypnotherapy
- Relaxation training
- Mindfulness meditation
- Psychotherapy, stress management
Integrated care models (physician + dietitian + psychologist + pelvic floor therapist) are beneficial for complex cases.
Prognosis
- 30–45% of patients have persistent symptoms for up to 12 years
- Predictors of persistent symptoms: anxiety, negative perception of stressful life events, female gender, younger age, prior anxiety/depression
- IBS subtypes can transition over time
- Significant impact on quality of life, work productivity, and healthcare utilization
📊 Treatment Algorithm Diagrams
Recent Evidence (2024–2025)
- A 2025 network meta-analysis in Lancet Gastroenterology & Hepatology (PMID 40258374) evaluated dietary interventions in IBS, providing updated comparative evidence on low-FODMAP and other diets.
- A 2024 systematic review in Nutrients (PMID 38999862) found that combination probiotic products and synbiotics outperformed single-strain probiotics and FMT for global IBS symptom reduction.
- A 2025 meta-analysis in European Journal of Gastroenterology & Hepatology (PMID 40359286) reported global IBS prevalence of ~3.8% using Rome IV criteria across 2006–2024 studies, with female sex and younger age as key risk factors.
Goldman-Cecil Medicine, 27th Ed. (Ch. 123) | Sleisenger & Fordtran's Gastrointestinal and Liver Disease
This is a shared conversation. Sign in to Orris to start your own chat.