Here is a comprehensive clinical reference on both topics, drawn from major medical textbooks and supported by diagnostic imaging.
I. CHOLECYSTITIS & BILIARY TRACT INFLAMMATION
Normal Physiology
The liver produces approximately 1,500 mL of bile per day, composed of bile acids, phospholipids, cholesterol, bilirubin, water, and electrolytes. The gallbladder concentrates bile 5–10-fold and stores it between meals. Cholecystokinin (CCK), released from duodenal I-cells in response to fat and protein, stimulates gallbladder contraction and relaxation of the sphincter of Oddi. Secretin stimulates bicarbonate-rich bile from cholangiocytes. The enterohepatic circulation recycles ~90% of bile acids via ileal reabsorption.
Bile emulsifies dietary fats, enabling lipase activity and micelle formation for fat-soluble vitamin absorption (A, D, E, K).
Etiology
| Type | Cause |
|---|
| Calculous cholecystitis (>90%) | Gallstone obstructing the cystic duct — cholesterol stones (80%), pigment stones (20%) |
| Acalculous cholecystitis (~5–10%) | Biliary stasis + ischemia: prolonged fasting, TPN, major trauma/burns, sepsis, DM, mechanical ventilation |
| Cholangitis | Biliary obstruction (stones, stricture, tumor) + bacterial infection |
| Primary sclerosing cholangitis | Immune-mediated fibro-inflammatory stricturing of bile ducts; associated with IBD (70%) |
| Choledocholithiasis | Common bile duct stones — primary (pigment) or secondary (migrated from gallbladder) |
Risk factors for gallstones: "5 F's" — Female, Fat, Fertile, Forty, Fair-skinned; also rapid weight loss, hemolytic anemia (pigment stones), cirrhosis, Crohn's disease.
Pathophysiology / Mechanisms
Three overlapping mechanisms drive acute cholecystitis:
- Mechanical obstruction — Cystic duct obstruction causes gallbladder distension, increased intraluminal pressure, wall ischemia, and venous/lymphatic congestion.
- Chemical inflammation — Concentrated bile components (lysolecithin from phospholipase A action, prostaglandins PGE₁/E₂) directly damage the gallbladder mucosa and amplify inflammation.
- Bacterial superinfection — Secondary infection by E. coli, Klebsiella, Enterococcus, Bacteroides, Clostridium (common in older patients, diabetics, and biliary obstruction).
In cholangitis, biliary obstruction raises ductal pressure, allowing bacterial reflux into the hepatic sinusoids → bacteremia and sepsis.
Physical Examination
| Sign | Description | Clinical Significance |
|---|
| Murphy's sign | Patient inspires deeply while examiner palpates the RUQ; patient arrests inspiration due to pain as inflamed GB contacts the fingers | Highly specific for acute cholecystitis (sensitivity ~65%, specificity ~87%) |
| Sonographic Murphy's sign | Same maneuver performed under ultrasound guidance | More reliable; positive = maximal tenderness directly over the visualized GB |
| Courvoisier's sign | Palpable, non-tender gallbladder with jaundice | Suggests malignant bile duct obstruction (pancreatic head cancer, cholangiocarcinoma), NOT stones (stones cause scarring that prevents GB dilation) |
| Charcot's triad | Fever + RUQ pain + jaundice | Classic for acute cholangitis |
| Reynolds' pentad | Charcot's triad + altered mental status + hypotension | Suppurative (severe) cholangitis — septic shock |
| RUQ tenderness | Involuntary guarding/rigidity | Peritoneal irritation (perforation or gangrene) |
Laboratory Findings
| Test | Finding | Significance |
|---|
| WBC | 10,000–15,000/μL (leukocytosis) | Inflammation/infection; >20,000 suggests empyema or perforation |
| ALT/AST | Mildly elevated | Hepatic involvement |
| ALP, GGT | Elevated | Biliary obstruction (choledocholithiasis, cholangitis) |
| Bilirubin | >2.5 mg/dL (mild elevation in cholecystitis); >5 mg/dL strongly suggests CBD stones | Biliary obstruction |
| Serum amylase/lipase | May be mildly elevated | Biliary pancreatitis if co-existing |
| Blood cultures | Positive in 50–70% of cholangitis | Guide antibiotic therapy |
| CRP/ESR | Elevated | Confirms acute inflammatory phase |
Diagnostic Imaging & Tests
Ultrasound (First-line)
- Sensitivity 85%, Specificity 95% for acute cholecystitis
- Findings: gallstones (hyperechoic foci with acoustic shadowing), GB wall thickening (>4 mm), pericholecystic fluid, sonographic Murphy's sign, biliary sludge
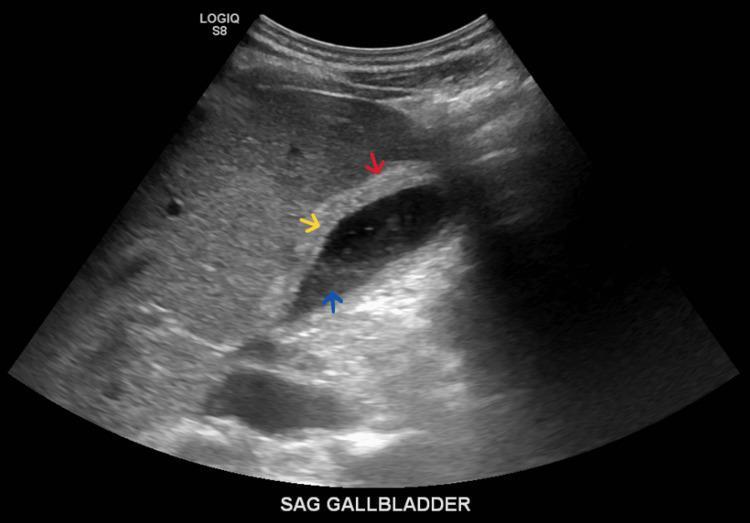
Ultrasound showing gallbladder wall thickening (red arrow), biliary sludge (blue arrow), and pericholecystic fluid (yellow arrow) — classic findings in acute cholecystitis
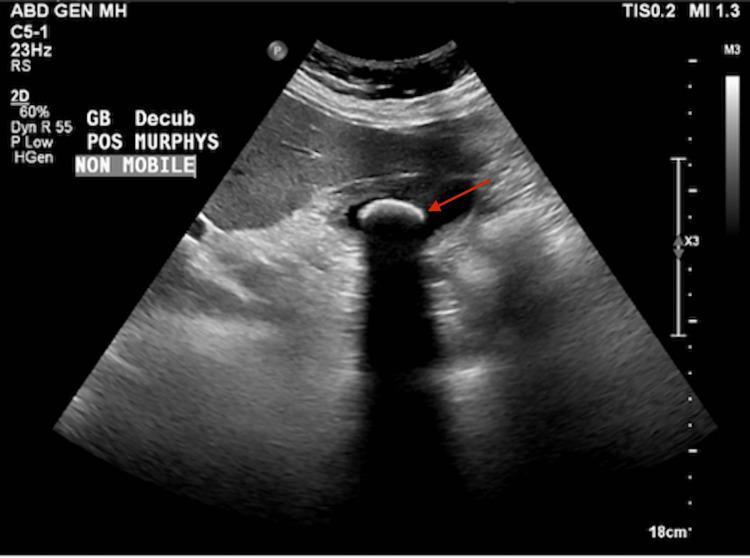
Ultrasound showing a non-mobile echogenic gallstone with posterior acoustic shadowing and a positive sonographic Murphy's sign
Scleroatrophic (shrunken) gallbladder in chronic cholecystitis
HIDA Scan (Hepatobiliary Iminodiacetic Acid / Cholescintigraphy)
- Gold standard for confirming cystic duct obstruction
- Non-visualization of the gallbladder at 4 hours = cystic duct obstruction = acute cholecystitis (sensitivity 97%, specificity 90%)
- Used when ultrasound is equivocal
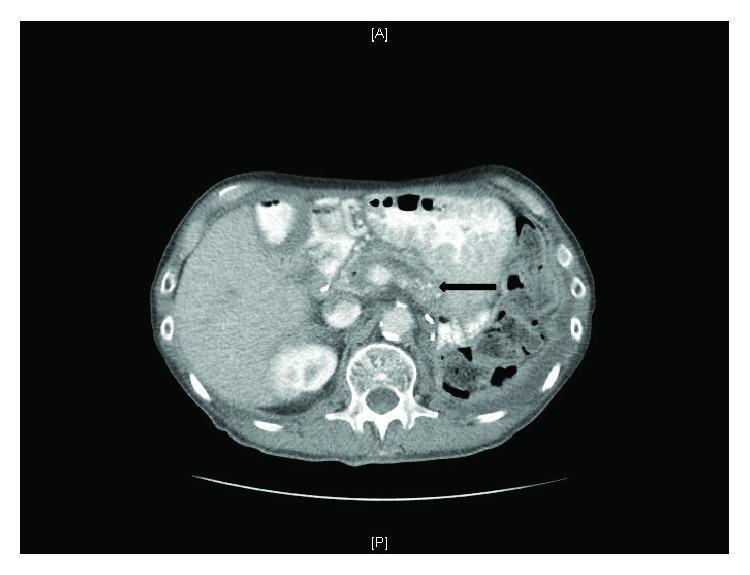
CT Abdomen
- Not first-line, but better for complications: perforation, pericholecystic abscess, empyema, fistula
- Can identify choledocholithiasis and cholangitis
MRCP (Magnetic Resonance Cholangiopancreatography)
- Non-invasive; >90% sensitivity/specificity for CBD stones
- Preferred for evaluating biliary tree anatomy before ERCP
ERCP (Endoscopic Retrograde Cholangiopancreatography)
- Diagnostic AND therapeutic — gold standard for CBD stone removal
- Allows sphincterotomy, stone extraction, biliary stenting
- Complications: post-ERCP pancreatitis (~5%), bleeding, perforation
EUS (Endoscopic Ultrasound)
- High sensitivity for small CBD stones, biliary strictures, early cholangiocarcinoma
- Preferred over MRCP for equivocal cases
Diagnostic Criteria (Tokyo Guidelines 2018 — Acute Cholecystitis)
Suspected (1 local sign + 1 systemic sign):
- Local: Murphy's sign, RUQ mass/pain/tenderness
- Systemic: fever, elevated CRP, leukocytosis
- Imaging confirmation required
Severity grading:
- Grade I (mild): No organ dysfunction
- Grade II (moderate): WBC >18,000, symptom duration >72h, pericholecystic abscess
- Grade III (severe): Organ dysfunction (cardiovascular, renal, hepatic, hematologic, neurologic, respiratory)
Therapy & Medications
Acute Cholecystitis
| Intervention | Details |
|---|
| Early laparoscopic cholecystectomy | Within 48–72 hours of symptom onset — gold standard (reduces complications vs. delayed surgery) |
| IV Antibiotics | Cover gram-negatives and anaerobes: Piperacillin-tazobactam, or Cefazolin + Metronidazole; escalate to carbapenems for severe/hospital-acquired |
| IV Fluids & NPO | Fluid resuscitation, bowel rest |
| Analgesia | NSAIDs (ketorolac) preferred; opioids (morphine, fentanyl) as needed |
| Percutaneous cholecystostomy | For Grade III or surgical high-risk patients — temporizing drainage |
| UDCA (Ursodeoxycholic acid) | Dissolves small cholesterol stones in non-surgical candidates; 8–12 months |
Cholangitis
| Intervention | Details |
|---|
| Emergency biliary decompression | ERCP + sphincterotomy/stone extraction — within 24–48h for moderate; emergency for severe/Reynolds' pentad |
| IV Antibiotics | Broad-spectrum (Pip-Tazo or carbapenem + metronidazole); blood cultures before starting |
| Biliary stenting | For malignant obstruction or strictures |
Primary Sclerosing Cholangitis (PSC)
- No proven disease-modifying therapy
- UDCA controversial (may be harmful at high doses)
- Biliary dilation + stenting for dominant strictures
- Liver transplantation for end-stage disease
Related Syndromes
| Syndrome | Description |
|---|
| Biliary colic | Transient cystic duct obstruction — severe RUQ pain, resolves within 6h, no fever/WBC elevation |
| Choledocholithiasis | CBD stones — jaundice, elevated bilirubin/ALP |
| Mirizzi syndrome | Gallstone compresses common hepatic duct externally from within the cystic duct — obstructive jaundice |
| Gallstone ileus | Fistula between GB and bowel → large stone enters intestine → bowel obstruction (air in biliary tree on X-ray = Rigler's triad) |
| Bouveret syndrome | Gallstone obstructs the duodenum (duodenal ileus) |
| Biliary pancreatitis | Gallstone passes through CBD → obstructs ampulla of Vater → acute pancreatitis |
| Cholangiocarcinoma | Biliary malignancy; associated with PSC, choledochal cysts, liver flukes |
| Choledochal cysts | Congenital biliary dilation — risk of cholangiocarcinoma |
Complications
- Empyema — pus-filled gallbladder; surgical emergency
- Gangrenous cholecystitis — necrosis of GB wall; perforation risk
- Perforation — free bile spillage → bile peritonitis (mortality >30%)
- Pericholecystic abscess — contained perforation
- Fistula formation — cholecystoenteric fistula
- Gallstone ileus — mechanical small bowel obstruction from large stone
- Suppurative cholangitis — life-threatening biliary sepsis (Reynolds' pentad)
- Secondary biliary cirrhosis — chronic biliary obstruction → hepatic fibrosis
II. EXOCRINE PANCREATIC INSUFFICIENCY (EPI)
Normal Exocrine Pancreatic Physiology
The exocrine pancreas consists of acinar cells (95% of mass) and ductal cells:
- Acinar cells produce digestive enzyme precursors (zymogens): trypsinogen, chymotrypsinogen, proelastase, prolipase, proamylase, prophospholipase A₂
- Ductal cells produce bicarbonate-rich fluid (pH 7.5–8.5) that neutralizes gastric acid
- CCK (from duodenal I-cells, triggered by fat/protein) → stimulates acinar enzyme secretion
- Secretin (from duodenal S-cells, triggered by acid) → stimulates bicarbonate fluid secretion
- Trypsinogen is activated to trypsin in the duodenum by enterokinase (from intestinal brush border); trypsin then activates all other zymogens
Critical threshold: Steatorrhea (fat malabsorption) only appears when lipase secretion drops below 10% of normal — the pancreas has enormous functional reserve.
Etiology
| Cause | Details |
|---|
| Chronic pancreatitis | Most common cause (~80%); alcohol (~50%), smoking (independent risk factor), idiopathic, tropical (nutritional) |
| Genetic mutations | CFTR (cystic fibrosis), PRSS1 (hereditary pancreatitis), SPINK1, CTRC mutations |
| Cystic fibrosis | CFTR mutations → abnormally thick secretions → ductal plugging → acinar destruction |
| Pancreatic surgery | Pancreatectomy, Whipple procedure, drainage procedures |
| Autoimmune pancreatitis | IgG4-associated; type 1 (systemic IgG4 disease) or type 2; responsive to steroids |
| Pancreatic cancer | Ductal obstruction |
| Celiac disease | Reduced CCK stimulation (mucosal damage) → reduced pancreatic stimulation |
| Diabetes mellitus | Type 3c "pancreatogenic" diabetes — loss of both endocrine and exocrine tissue |
| Shwachman-Diamond syndrome | Rare autosomal recessive; EPI + bone marrow failure in children |
Pathophysiology / Mechanisms
In chronic pancreatitis (the archetypal cause):
- Repeated acinar injury (alcohol metabolites, oxidative stress, ductal hypertension) → necrosis-fibrosis sequence
- Pancreatic stellate cell activation → TGF-β and PDGF-driven fibrogenesis → progressive parenchymal replacement by fibrous tissue
- Ductal obstruction → protein plug formation → calcification → intraductal calculi → ductal hypertension → further acinar destruction
- Enzyme deficiency cascade: Lipase most sensitive (lost earliest) → proteases → amylase (most preserved)
- Bicarbonate deficiency: Low duodenal pH inactivates remaining lipase, worsens malabsorption
Steatorrhea requires >90% loss of exocrine function (enormous pancreatic reserve). Fat malabsorption → fat-soluble vitamin deficiencies (A, D, E, K) → osteoporosis, coagulopathy, night blindness.
Clinical Features & Physical Examination
Symptoms:
- Epigastric pain radiating to the back — constant or episodic, worse after meals and alcohol
- Steatorrhea — pale, bulky, greasy, malodorous, floating stools difficult to flush; oily film in toilet water
- Weight loss — malabsorption + reduced food intake due to pain
- Fat-soluble vitamin deficiencies: Night blindness (Vit A), osteoporosis/tetany (Vit D/Ca), bleeding tendency (Vit K)
- Type 3c diabetes — late-stage, when islets are also destroyed
Physical Examination:
- Epigastric tenderness, often without rebound or guarding
- Signs of malnutrition: muscle wasting, temporal wasting, peripheral edema (hypoalbuminemia)
- Jaundice (if biliary obstruction from pancreatic head fibrosis)
- Ascites (pancreatic pseudocyst or pancreatic duct disruption)
- Cullen's sign / Grey Turner's sign — only in acute pancreatitis, not EPI per se
Diagnostic Tests
Functional Tests (Direct)
| Test | Method | Normal/Abnormal | Notes |
|---|
| Secretin stimulation test | IV secretin → duodenal intubation → aspirate fluid; measure HCO₃⁻ and volume | Peak HCO₃⁻ <80 mEq/L = abnormal | Gold standard for early EPI; not widely available |
| Secretin-CCK (pancreozymin) test | Same + CCK stimulation → measure enzyme output | Reduced amylase, lipase, trypsin output | Most sensitive direct test |
Functional Tests (Indirect)
| Test | Details | Cutoffs | Notes |
|---|
| Fecal elastase-1 (FE-1) | ELISA on stool sample; elastase-1 is pancreas-specific, not degraded in transit | <200 μg/g = mild/moderate EPI; <100 μg/g = severe EPI | First-line non-invasive test; false negatives with diarrhea |
| 72-hour fecal fat | Quantitative fat on 100 g/day fat diet | >7 g/day (normal <7) | Sensitivity ~95% for severe EPI; laborious |
| Coefficient of fat absorption | Fecal fat / dietary fat intake | <93% abnormal | Reference standard for steatorrhea |
| Serum trypsinogen | Radioimmunoassay | <20 ng/mL = severe EPI | Highly specific for advanced EPI |
| NBT-PABA test | Oral synthetic substrate; urinary PABA measured | Low PABA = EPI | Rarely used now |
Structural Imaging
| Modality | Findings in EPI/Chronic Pancreatitis |
|---|
| Abdominal X-ray | Pancreatic calcifications (highly specific for chronic pancreatitis) |
| CT Abdomen | Parenchymal atrophy, calcifications, main pancreatic duct dilation, pseudocysts |
| MRI/MRCP | Chain-of-lakes ductal morphology; best for ductal anatomy; MRCP with secretin injection improves sensitivity |
| EUS (Endoscopic Ultrasound) | Most sensitive for early chronic pancreatitis (Rosemont criteria: ≥5 EUS features = consistent with CP) |
| ERCP | Cambridge classification of ductal changes; now largely replaced by MRCP for diagnosis |
CT abdomen: Pancreatic atrophy, multifocal calcifications throughout head/body/tail, and main pancreatic duct dilation — advanced chronic pancreatitis with EPI
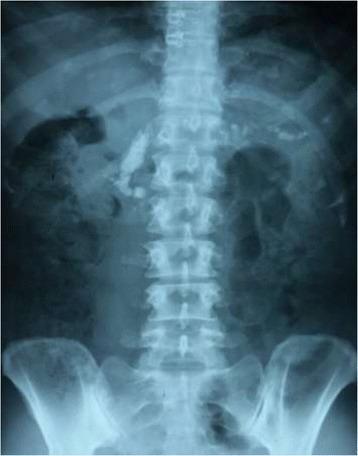
Plain abdominal X-ray showing diffuse pancreatic calcifications spanning head, body, and tail — highly specific for chronic calcific pancreatitis
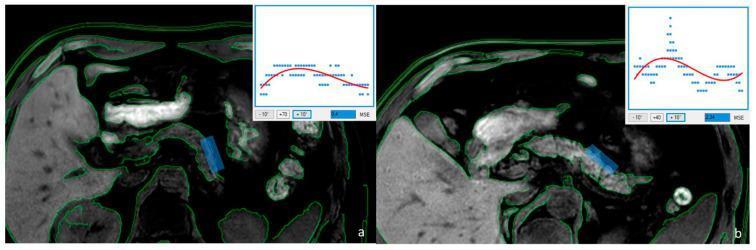
T1 MRI: (a) Irregular pancreatic margins + heterogeneous texture = severe EPI (FE-1 = 45 μg/g); (b) Smoother borders = preserved function (FE-1 = 320 μg/g)
Diagnostic Criteria for Chronic Pancreatitis
Diagnostic classification: Definite (calcifications, severe duct changes, histology), Probable (mild duct changes, pseudocysts, abnormal function tests), Borderline (clinical history only)
Therapy & Medications
Pancreatic Enzyme Replacement Therapy (PERT)
The cornerstone of EPI treatment:
| Preparation | Dosing | Notes |
|---|
| Pancrelipase (Creon, Zenpep, Pertzye) | 40,000–90,000 USP lipase units/main meal; 20,000–40,000 units/snack | Enteric-coated microspheres — protect enzymes from acid; take WITH meals (not before or after) |
| High-dose PERT | Up to 90,000 units/meal | For persisting steatorrhea despite standard dosing |
Adjuncts to PERT:
- PPI (Proton pump inhibitor) — Omeprazole 20–40 mg — raises duodenal pH to optimize lipase activity; critical when bicarbonate secretion is impaired
- H₂ blockers — Alternative to PPI
Nutritional Support
| Intervention | Rationale |
|---|
| Fat-soluble vitamins (A, D, E, K) | Replace malabsorbed vitamins; Vitamin D + calcium for osteoporosis prevention |
| Vitamin B₁₂ | Monitor levels (ileal absorption may be impaired) |
| Low-fat diet | May reduce steatorrhea, but can worsen malnutrition — individualize |
| Medium-chain triglycerides (MCT) | Absorbed without lipase/bile acids; useful supplement |
| Nutritional support | Enteral feeding (nasojejunal) for severe malnutrition; parenteral as last resort |
Pain Management
| Agent | Notes |
|---|
| Non-opioid analgesics first | Paracetamol, NSAIDs |
| Tramadol, opioids | For refractory pain; risk of dependence |
| Antioxidants | Selenium, Vitamin C, E, methionine — benefit in idiopathic chronic pancreatitis |
| Celiac plexus block | EUS-guided; for refractory pain |
| Pregabalin / duloxetine | Neuropathic pain component |
Lifestyle Modifications
- Strict alcohol cessation — Slows disease progression, may partially recover function
- Tobacco cessation — Smoking is independent risk factor for disease progression and pancreatic cancer
Surgical & Endoscopic Interventions
| Procedure | Indication |
|---|
| ERCP + stone extraction | Pancreatic duct stones causing obstruction |
| ESWL (Extracorporeal shockwave lithotripsy) | Fragmentation of large pancreatic duct calculi before ERCP |
| Lateral pancreaticojejunostomy (Puestow procedure) | Dilated main duct (>6 mm) + pain |
| Whipple (pancreaticoduodenectomy) | Pancreatic head inflammatory mass causing obstruction |
| Total pancreatectomy + islet autotransplantation (TPIAT) | Painful small-duct disease; preserves glycemic control |
Management of Type 3c Diabetes
- Avoid aggressive insulin therapy (risk of hypoglycemia — lost glucagon from islet destruction)
- Metformin cautiously; insulin required as disease progresses
Related Syndromes
| Syndrome | Relationship |
|---|
| Steatorrhea syndrome | Clinical hallmark of EPI — fat malabsorption |
| Type 3c diabetes (pancreatogenic) | Late complication; combined exocrine + endocrine destruction |
| Cystic fibrosis | CFTR mutations → EPI from birth; most severe form of inherited EPI |
| Hereditary pancreatitis | PRSS1 mutations; early onset, 40% lifetime pancreatic cancer risk in smokers |
| Autoimmune pancreatitis (AIP) | IgG4-related; "mass-forming" mimics pancreatic cancer; steroid-responsive |
| Tropical calcific pancreatitis | Nutritional (cassava-related?); endemic in southern India/Africa; young patients, severe calcifications |
| SIBO (Small intestinal bacterial overgrowth) | Secondary to reduced pancreatic secretions/altered motility; worsens malabsorption |
| Pancreatic pseudocyst | Loculated fluid collection; may cause biliary or gastric outlet obstruction |
| Groove pancreatitis | Fibrous inflammation in pancreatic head groove; mimics malignancy |
Complications
| Complication | Details |
|---|
| Malnutrition / cachexia | Fat + protein malabsorption; fat-soluble vitamin deficiencies |
| Osteoporosis | Vit D/Ca malabsorption; screen with DEXA scan |
| Type 3c diabetes mellitus | Brittle, hypoglycemia-prone; insulin-requiring |
| Pancreatic pseudocysts | May rupture, bleed, become infected, or cause compression |
| Biliary obstruction | Fibrotic pancreatic head compresses CBD → obstructive jaundice |
| Duodenal obstruction | Rare, from fibrotic mass |
| Portal/splenic vein thrombosis | Regional hypertension; varices |
| Pancreatic cancer | 4% lifetime risk in chronic pancreatitis; up to 40% in hereditary pancreatitis + smokers |
| Pancreatic ascites | Ductal disruption → internal pancreatic fistula |
| SIBO | Functional malabsorption superimposed on EPI |
Summary Comparison Table
| Feature | Cholecystitis / Biliary Disease | Exocrine Pancreatic Insufficiency |
|---|
| Key mechanism | Obstruction → inflammation → bacterial infection | Acinar loss → enzyme deficiency → malabsorption |
| Classic symptom | RUQ pain, fever, jaundice | Epigastric pain, steatorrhea, weight loss |
| Key physical sign | Murphy's sign, Courvoisier's sign | Epigastric tenderness, signs of malnutrition |
| First-line imaging | Ultrasound | CT abdomen / EUS |
| Gold standard test | HIDA scan (acute cholecystitis); ERCP (CBD stones) | Secretin stimulation test; Fecal elastase-1 |
| Key lab finding | Leukocytosis, elevated ALP/bilirubin | Low fecal elastase-1 (<100 μg/g severe); fecal fat >7 g/day |
| Primary treatment | Cholecystectomy; IV antibiotics; ERCP | PERT + PPI + vitamins + lifestyle modification |
| Major complications | Empyema, perforation, cholangitis, biliary cirrhosis | Malnutrition, diabetes type 3c, pancreatic cancer |