Physiological changes in pregnancy.
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
I now have comprehensive data from Morgan & Mikhail's Clinical Anesthesiology. Let me compile a thorough, well-organized response.
Physiological Changes in Pregnancy
Pregnancy induces adaptive changes in virtually every organ system to meet the increased metabolic demands of the mother and fetus. The changes below represent maximum values near term.
Summary Table of Key Changes
| Parameter | Change |
|---|---|
| MAC (anesthetic requirement) | ↓ 40% |
| Oxygen consumption | ↑ 20–50% |
| Airway resistance | ↓ 35% |
| FRC | ↓ 20% |
| Minute ventilation | ↑ 50% |
| Tidal volume | ↑ 40% |
| Respiratory rate | ↑ 15% |
| PaO₂ | ↑ 10% |
| PaCO₂ | ↓ 15% |
| HCO₃⁻ | ↓ 15% |
| Blood volume | ↑ 35% |
| Plasma volume | ↑ 55% |
| Cardiac output | ↑ 40% |
| Stroke volume | ↑ 30% |
| Heart rate | ↑ 20% |
| Systolic BP | ↓ 5% |
| Diastolic BP | ↓ 15% |
| Peripheral resistance | ↓ 15% |
| Pulmonary resistance | ↓ 30% |
| Hemoglobin | ↓ 20% |
| Platelets | ↓ 10% |
| Clotting factors | ↑ 30–250% |
| GFR | ↑ 50% |
1. Central Nervous System
- MAC decreases progressively during pregnancy — by up to 40% at term for all general anesthetic agents. It returns to normal by the third day postpartum.
- Progesterone rises up to 20× normal at term and, being inherently sedating, is largely responsible for the reduced MAC.
- A surge in β-endorphin levels during labor also contributes.
- Pregnant patients show enhanced sensitivity to local anesthetics during regional anaesthesia; neural blockade occurs at reduced drug concentrations. This may relate to progesterone-induced sensitization of nerve membranes and the dilutional effect of epidural venous engorgement (reducing epidural/subarachnoid space volume).
2. Respiratory System
Anatomical Changes
- Progesterone causes capillary engorgement of the respiratory mucosa → mucosal swelling, friability, and increased secretions throughout the nose, oropharynx, larynx, and trachea.
- The growing uterus elevates the diaphragm by ~4 cm, reducing FRC. Rib cage flaring partially compensates.
Lung Volume Changes
| Volume | Change |
|---|---|
| FRC (ERV + RV) | ↓ 20% |
| ERV | ↓ 20% |
| RV | ↓ 20% |
| TLC | ↓ 5% |
| Inspiratory capacity | ↑ 5% |
| Closing capacity | Unchanged |
- The closing volume may exceed FRC in the supine position, causing atelectasis, V/Q mismatch, and shunting — especially relevant during induction of anaesthesia.
Ventilation Changes
- Minute ventilation ↑ 50%: primarily driven by ↑ tidal volume (40%), with a smaller ↑ in respiratory rate (15%).
- Progesterone is the main stimulus, increasing sensitivity of the respiratory centre to CO₂.
- Result: chronic respiratory alkalosis — PaCO₂ falls to ~28–32 mmHg; renal compensation lowers HCO₃⁻ to ~20 mEq/L, maintaining pH near normal (7.44).
- PaO₂ rises to ~103–107 mmHg (due to hyperventilation).
- Oxygen consumption ↑ 20–50% (fetal/placental metabolism + increased cardiac/respiratory work).
- Apnoeic desaturation is rapid in pregnant patients because of the combination of ↓ FRC and ↑ O₂ consumption — critical consideration during airway management.
3. Cardiovascular System
Cardiac Output
- CO ↑ 40% by the end of the first trimester and stays elevated through term.
- Driven by ↑ stroke volume (30%) and ↑ heart rate (20%).
- In labour, CO increases further (15–50%) with each uterine contraction (autotransfusion of ~300–500 mL blood from the uterus).
- Immediately postpartum, CO rises sharply as the uterus involutes, then gradually falls over 2 weeks.
Blood Pressure
- Systolic BP ↓ ~5%, diastolic BP ↓ ~15% despite the raised CO — due to reduced peripheral vascular resistance from progesterone-mediated vasodilation and arteriovenous shunting through the placenta.
Blood Volume
- Plasma volume ↑ 55%, red cell mass ↑ ~20–30% → dilutional (physiological) anaemia (Hb falls ~20%).
- Provides a buffer against haemorrhage at delivery.
Aortocaval Compression
- After ~20 weeks, the gravid uterus compresses the inferior vena cava and the aorta in the supine position.
- IVC compression reduces venous return → ↓ CO and hypotension ("supine hypotensive syndrome"); the aorta compression reduces uteroplacental flow.
- Left lateral tilt (15°) relieves this.
Other Cardiac Changes
- ECG findings: left axis deviation (diaphragm elevation), sinus tachycardia, ST-T changes, and increased ectopic beats — all normal.
- Heart sounds: S1 louder; S3 audible; soft systolic ejection murmur common due to ↑ flow.
4. Haematological System
Blood and Coagulation
- Plasma volume ↑ more than red cell mass → dilutional anaemia (physiological Hb ~11 g/dL at term).
- Clotting factors I (fibrinogen), VII, VIII, IX, X, XII ↑ 30–250% → hypercoagulable state.
- Protein S ↓, resistance to activated protein C → further prothrombotic tendency (risk of DVT/PE ↑ 5×).
- Platelets ↓ ~10% (dilutional); gestational thrombocytopaenia common but usually benign.
- Fibrinolysis is reduced.
5. Renal System
- Renal blood flow and GFR ↑ ~50% by the end of the first trimester.
- Serum creatinine and urea fall (values normal in non-pregnant women may represent renal impairment in pregnancy).
- Glycosuria is common (GFR exceeds tubular reabsorption threshold) — does not indicate diabetes alone.
- Progesterone-induced smooth muscle relaxation causes ureteral dilation and urinary stasis → ↑ risk of urinary tract infection and pyelonephritis.
- Mild proteinuria (up to 300 mg/day) is normal.
6. Gastrointestinal System
- Gastric emptying is delayed in labour (pain, opioids, anxiety).
- Progesterone reduces lower oesophageal sphincter (LES) tone and increases intragastric pressure (from the enlarged uterus) → high risk of gastro-oesophageal reflux and pulmonary aspiration (Mendelson's syndrome).
- These changes begin as early as the first trimester and are most pronounced after 20 weeks.
- Hepatic function is broadly maintained, though plasma alkaline phosphatase rises (placental isoenzyme).
7. Musculoskeletal System
- Progesterone and relaxin loosen ligaments → instability of sacroiliac, sacrococcygeal, and symphysis pubis joints.
- The lumbar lordosis increases as the uterus enlarges, shifting the centre of gravity → common low back pain.
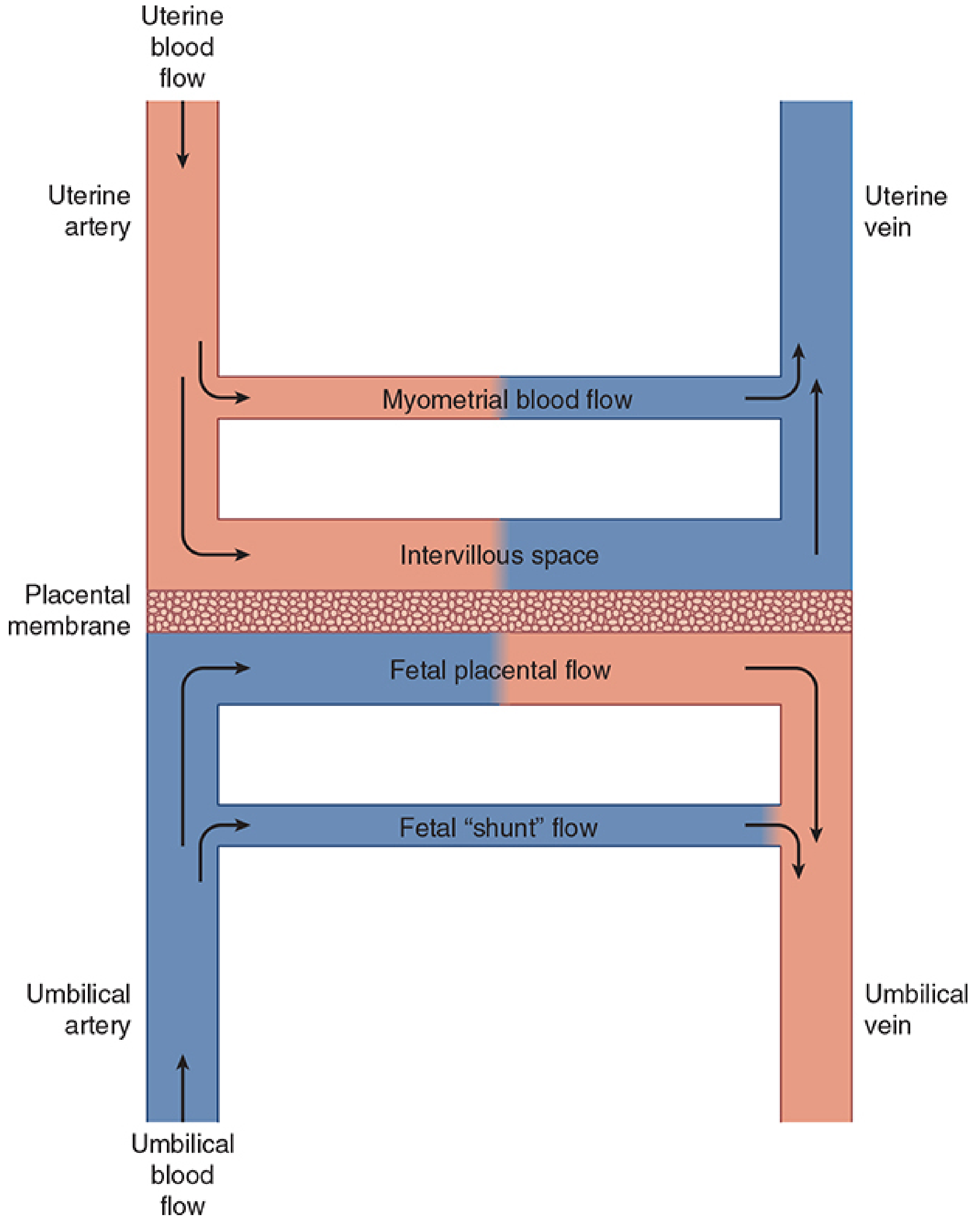
8. Uteroplacental Circulation
- At term, uterine blood flow = 600–700 mL/min (~10% of CO), compared with 50 mL/min in the non-pregnant state. About 80% supplies the placenta.
- Uterine vasculature is maximally dilated (no autoregulation) but remains sensitive to α-adrenergic agonists.
Three major factors decrease uterine blood flow:
- Systemic hypotension (aortocaval compression, haemorrhage, sympathetic block)
- Uterine vasoconstriction (catecholamines, α-agonists)
- Uterine contractions (↑ venous pressure, arterial compression)
Clinical Implications Summary
| System | Key Clinical Point |
|---|---|
| Airway | Mucosal oedema, friability → difficult intubation; rapid desaturation |
| Respiratory | ↓ FRC + ↑ O₂ demand → pre-oxygenate before intubation |
| CVS | Left lateral tilt after 20 weeks; avoid vasopressors with pure α-activity |
| GI | Assume full stomach; RSI for all general anaesthetics from 1st trimester |
| Renal | Normal non-pregnant creatinine may indicate impairment |
| Haematology | Hypercoagulable → VTE prophylaxis; anaemia is physiological |
— Morgan and Mikhail's Clinical Anesthesiology, 7e, pp. 1572–1581
Apache Critical Care
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
I now have comprehensive content from Harrison's on APACHE and ICU scoring. Let me compile a thorough answer.
APACHE Score in Critical Care
What is APACHE?
APACHE stands for Acute Physiology and Chronic Health Evaluation. It is a severity-of-illness (SOI) scoring system developed to quantify the degree of physiological derangement in critically ill patients admitted to the ICU. It has gone through four major iterations: APACHE I → II → III → IV, with APACHE II being the most widely used in clinical practice.
Purpose & Utility
SOI scoring systems like APACHE are validated for:
- Predicting population-level mortality (not reliably individual outcomes)
- Stratifying patients for clinical trials and epidemiologic research
- Guiding hospital administrative decisions — resource allocation, nursing ratios
- Benchmarking ICU quality over time
- Defining disease severity (e.g., acute pancreatitis, sepsis, ARDS)
Scoring system validations are based on the premise that age, chronic medical illness, and derangements from normal physiology are associated with increased mortality. — Harrison's Principles of Internal Medicine, 22e
APACHE II — Components
APACHE II scores are calculated from 12 acute physiological variables measured in the first 24 hours of ICU admission, plus age and chronic health status.
A. Acute Physiology Score (APS) — 12 Variables
| Variable | Scored Parameter |
|---|---|
| 1. Temperature | Rectal (°C) |
| 2. Mean arterial pressure | mmHg |
| 3. Heart rate | beats/min |
| 4. Respiratory rate | breaths/min |
| 5. Oxygenation | PaO₂ or A-aDO₂ depending on FiO₂ |
| 6. Arterial pH | |
| 7. Serum sodium | mEq/L |
| 8. Serum potassium | mEq/L |
| 9. Serum creatinine | mg/dL |
| 10. Haematocrit | % |
| 11. White blood cell count | ×10³/mm³ |
| 12. Glasgow Coma Scale | Score (15 − actual GCS) |
Each variable is scored 0–4 based on how far it deviates from normal (in either direction). The worst value in the first 24 hours is used.
B. Age Score
| Age (years) | Points |
|---|---|
| < 44 | 0 |
| 45–54 | 2 |
| 55–64 | 3 |
| 65–74 | 5 |
| ≥ 75 | 6 |
C. Chronic Health Score
Patients with severe organ system insufficiency or immunocompromise receive additional points:
- +5 points if non-operative or emergency postoperative admission
- +2 points if elective postoperative admission
Qualifying conditions include:
- Liver: cirrhosis with portal hypertension, encephalopathy, or variceal bleeding
- Cardiovascular: NYHA Class IV heart failure
- Respiratory: chronic hypoxia, hypercapnia, polycythaemia, severe pulmonary hypertension, or ventilator dependence
- Renal: chronic dialysis
- Immunocompromise: receiving immunosuppressive therapy, chemotherapy, steroids, or having haematologic malignancy/AIDS
APACHE II Total Score
APACHE II = APS + Age Score + Chronic Health Score
Maximum possible score = 71
Score Interpretation & Mortality
| APACHE II Score | Approximate ICU Mortality |
|---|---|
| 0–4 | ~4% |
| 5–9 | ~8% |
| 10–14 | ~15% |
| 15–19 | ~25% |
| 20–24 | ~40% |
| 25–29 | ~55% |
| 30–34 | ~73% |
| ≥ 35 | ~85% |
Exact mortality varies by diagnosis and institution.
Disease-Specific Thresholds
Acute Pancreatitis:
- APACHE II ≥ 8 at 24 hours → associated with ICU need, pancreatic necrosis, secondary infection, and increased mortality. — Swanson's Family Medicine Review
General Critical Care:
- Rising scores over time correlate with worsening prognosis
- Used alongside other systems (SOFA, Ranson, BISAP depending on context)
APACHE Versions
| Version | Key Features |
|---|---|
| APACHE I | Original 34-variable system (1981) |
| APACHE II | Simplified to 12 variables + age + chronic health (1985); most widely adopted |
| APACHE III | Added 5 more variables; developed proprietary prediction equations (1991) |
| APACHE IV | Updated equations using more recent data; improved discrimination (2006) |
APACHE vs. Other ICU Scoring Systems
| Score | Focus | Key Difference |
|---|---|---|
| APACHE II | Overall severity; mortality prediction | Single 24-hr snapshot; chronic health included |
| SOFA | Organ failure assessment | Repeated daily; tracks trajectory; used for sepsis diagnosis |
| qSOFA | Rapid bedside sepsis screen | 3 criteria: RR ≥22, altered mentation, SBP ≤100 |
| SAPS II | Simplified acute physiology | European equivalent; 17 variables |
| BISAP | Acute pancreatitis severity | 5 bedside criteria; simpler than APACHE |
| Ranson | Acute pancreatitis | 11 criteria over 48 hours; no repeated use |
Limitations
- Not validated for individual prediction — a high score does not mandate withdrawal of care
- Requires 24 hours of data — not useful for immediate triage
- Does not capture trajectory of illness (unlike SOFA)
- Worst-value rule may over-penalise transient derangements
- Chronic health scoring is subjective in borderline cases
- Originally validated on 1980s patient populations — may not reflect modern ICU outcomes
Key Clinical Takeaway
APACHE II remains the most commonly used critical care scoring tool worldwide. Its primary value lies in population-level risk stratification, benchmarking, and clinical research — not at-the-bedside individual prognostication. For dynamic, real-time assessment of organ failure trajectory (especially in sepsis), the SOFA score is preferred.
— Harrison's Principles of Internal Medicine, 22e; Swanson's Family Medicine Review; Sabiston Textbook of Surgery, 20e
Management of ARDS in pancreatitis patient
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content from Murray & Nadel's and Current Surgical Therapy. Let me compile a complete, well-structured answer.
Management of ARDS in a Pancreatitis Patient
Background: ARDS as a Complication of Pancreatitis
ARDS develops in approximately 15–20% of patients with acute pancreatitis and accounts for up to 60% of deaths. It typically manifests 2–7 days after onset. Pathologic changes in the lungs are identical to ARDS from any other cause.
Pathogenesis of Lung Injury in Pancreatitis
| Mechanism | Detail |
|---|---|
| Pancreatic enzymes | Elastase and lipase released systemically → ↑ lung vascular permeability |
| Phospholipase A2 | Degrades surfactant → alveolar collapse + ↑ permeability; strongly correlates with respiratory insufficiency |
| Neutrophil sequestration | Activated neutrophils accumulate in alveolar and interstitial spaces |
| Proinflammatory cytokines | TNF-α, IL-8 → augment neutrophil recruitment and activation in the lungs |
| Reactive oxygen species | Platelet-activating factor, adhesion molecules → ↑ vascular permeability |
Diagnosis: Berlin Criteria for ARDS
| Feature | Mild | Moderate | Severe |
|---|---|---|---|
| Hypoxemia (PaO₂:FiO₂) | 201–300 mmHg with PEEP ≥5 | 101–200 mmHg with PEEP ≥5 | ≤100 mmHg with PEEP ≥5 |
| Timing | Within 1 week of clinical insult or new/worsening respiratory symptoms | ||
| Chest imaging | Bilateral opacities on CXR/CT not explained by effusions, lobar collapse, or nodules | ||
| Origin of oedema | Not explained by cardiac failure or volume overload (echo to exclude hydrostatic oedema) |
Management: General Principles
"Treatment of pancreatitis-associated ARDS is supportive and does not differ from that for other forms of ARDS. Key measures include cardiovascular support and lung-protective ventilation." — Murray & Nadel's Textbook of Respiratory Medicine
1. Lung-Protective Ventilation (Standard of Care)
The cornerstone of ARDS management, regardless of aetiology.
Targets:
| Parameter | Target |
|---|---|
| Tidal volume (VT) | 6 mL/kg predicted body weight (PBW) |
| Plateau pressure (Pplat) | < 30 cm H₂O |
| Driving pressure (ΔP = Pplat − PEEP) | Minimise; ↑ ΔP independently associated with ↑ mortality |
| SpO₂ / PaO₂ target | SpO₂ 88–95% / PaO₂ 55–80 mmHg |
| pH target | 7.30–7.45 |
- The landmark ARMA trial (ARDSNet) showed 6 mL/kg PBW versus 12 mL/kg PBW reduced mortality significantly — establishing low tidal volume ventilation as standard of care.
- Volutrauma and barotrauma from overdistension drive ventilator-induced lung injury (VILI) and amplify the systemic inflammatory response.
Permissive Hypercapnia
- Deliberate hypoventilation to keep Pplat < 30 cm H₂O causes hypercapnia and respiratory acidosis.
- Managed medically with sodium bicarbonate or tromethamine (THAM).
- Use with caution if raised intracranial pressure is a concern.
2. PEEP Titration
Higher PEEP recruits collapsed alveoli, reduces V/Q mismatch, and improves oxygenation.
FiO₂/PEEP table (open lung strategy):
| FiO₂ | 0.3 | 0.3 | 0.3 | 0.4 | 0.5 | 0.5 | 0.8 | 0.9 | 1.0 |
|---|---|---|---|---|---|---|---|---|---|
| PEEP (cm H₂O) | 8 | 10 | 12 | 14 | 16 | 18 | 22 | 22 | 22–24 |
- Optimal PEEP may be individualised using transpulmonary pressure (measured by oesophageal balloon manometry) or driving pressure titration.
- Both under-distension and over-distension worsen VILI.
3. Fluid Management — Critical Nuance in Pancreatitis
This is the key tension in pancreatitis-associated ARDS:
- Aggressive fluid resuscitation is standard in acute pancreatitis (to prevent organ failure and pancreatic necrosis).
- However, overzealous resuscitation increases both pulmonary and non-pulmonary complications in this setting.
- Once ARDS is established, a conservative fluid strategy is recommended:
- Reduces duration of mechanical ventilation and ICU stay.
- Consider diuresis if CVP > 4 with persistent hypoxaemia — but only after confirming adequate perfusion and cardiac output.
"Although aggressive fluid resuscitation remains a pillar in the management of acute pancreatitis, overzealous resuscitation may increase the rate of both pulmonary and nonpulmonary complications." — Murray & Nadel's
4. Prone Positioning (Moderate–Severe ARDS)
- Indicated for PaO₂:FiO₂ < 150 with FiO₂ ≥ 0.6, PEEP ≥ 5 cm H₂O.
- PROSEVA trial: prone ≥16 hours/day → 90-day mortality 23.6% vs. 41% in controls.
- Mechanism: recruits dependent (posterior) lung zones, improves V/Q matching.
- Must be initiated within 36 hours of moderate–severe ARDS for benefit.
- Requires careful planning to avoid: unplanned extubation, pressure injuries, inadvertent line removal.
5. Neuromuscular Blockade (NMB)
- Consider for early, severe ARDS (PaO₂:FiO₂ ≤150, within 36 hours of intubation).
- ACURASYS trial: NMB improved 90-day survival and increased ventilator-free days.
- ROSE trial (follow-up): no clear mortality benefit vs. light sedation — but NMB remains a salvage option to:
- Eliminate ventilator dyssynchrony
- Reduce consumptive hypoxaemia
- Prevent patient-self-inflicted lung injury
6. Cardiovascular Support
- Maintain adequate MAP (typically ≥65 mmHg) to preserve organ perfusion.
- Vasopressors: noradrenaline first-line in vasodilatory/distributive shock pattern.
- Avoid excess fluid after initial resuscitation once ARDS is established.
7. Salvage Strategies for Refractory ARDS
Recruitment Manoeuvres (RM)
- Transiently increase transpulmonary pressure to reopen collapsed alveoli.
- Common approach: 40 cm H₂O airway pressure for 2 minutes.
- Caution: a trial using very high pressures (45 cm H₂O) showed higher 28-day mortality — routine high-pressure RMs should be avoided.
Inhaled Pulmonary Vasodilators
- Inhaled nitric oxide (iNO) and inhaled prostacyclin improve short-term oxygenation but do not reduce mortality or ventilator days.
- Reserved for life-threatening hypoxaemia despite all other measures, or as a bridge to ECMO or transfer.
Extracorporeal Membrane Oxygenation (ECMO)
- For severe refractory ARDS failing all standard + salvage therapies.
- CESAR trial: transfer to ECMO-capable centre improved outcomes.
- EOLIA trial: stopped early for futility on primary outcome, but secondary outcomes suggested benefit.
- Approximate survival ~50% in experienced centres.
- Requires veno-venous (VV) ECMO for respiratory failure.
8. Pancreatitis-Specific Adjuncts
Octreotide (Somatostatin Analogue)
- Two small studies (mid-1990s) suggest octreotide may reduce ARDS incidence and mortality in severe acute pancreatitis by inhibiting exocrine pancreatic secretions (and thus reducing systemic enzyme release).
- Evidence is preliminary — larger RCTs needed before routine recommendation.
Treatment of the Underlying Pancreatitis
- Adequate analgesia (IV opioids)
- Nil by mouth / enteral nutrition via nasojejunal tube early (within 24–48 h in severe cases)
- Treat the cause (biliary: ERCP if obstruction; alcohol: supportive)
- Antibiotics only if proven infected pancreatic necrosis
Summary Algorithm
ARDS in Pancreatitis
│
├─ Intubate + Mechanical Ventilation
│ └── VT 6 mL/kg PBW | Pplat <30 | Permissive hypercapnia
│
├─ PEEP titration (FiO2/PEEP table or transpulmonary pressure)
│
├─ Conservative fluid strategy (avoid excess resuscitation)
│
├─ PaO2:FiO2 <150 → Prone positioning (≥16 h/day)
│
├─ PaO2:FiO2 <150 → Consider NMB (cisatracurium)
│
├─ Refractory → Recruitment manoeuvres ± iNO/prostacyclin
│
└─ Failing all → VV-ECMO at specialised centre
Sources: Murray & Nadel's Textbook of Respiratory Medicine, 7e; Current Surgical Therapy, 14e; Harrison's Principles of Internal Medicine, 22e
Retry
Could you clarify what you'd like me to retry? For example:
- Regenerate the ARDS in pancreatitis answer (same topic, fresh response)
- A different topic you meant to type
- Expand or focus on a specific section of the previous answer
This is a shared conversation. Sign in to Orris to start your own chat.