Emphysema lung full details for md exam answer 15 marks
Reading File
Finding Sources
Finding Sources
Searching Images
emphysema lung pathology gross microscopy
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Reading File
Emphysema (15-mark MD exam answer)
Definition
Emphysema is abnormal permanent enlargement of airspaces distal to terminal bronchioles with destruction of alveolar walls, without significant fibrosis.
- Robbins & Kumar Basic Pathology, p. 255
- Robbins, Cotran & Kumar Pathologic Basis of Disease, p. 634
Etiology and risk factors
- Cigarette smoking (most important cause in COPD-related emphysema)
- Alpha-1 antitrypsin (A1AT) deficiency (classically panacinar type, earlier age)
- Air pollutants/biomass exposure (contributory)
- Genetic susceptibility (other loci also influence risk)
- Robbins, Cotran & Kumar Pathologic Basis of Disease, p. 636
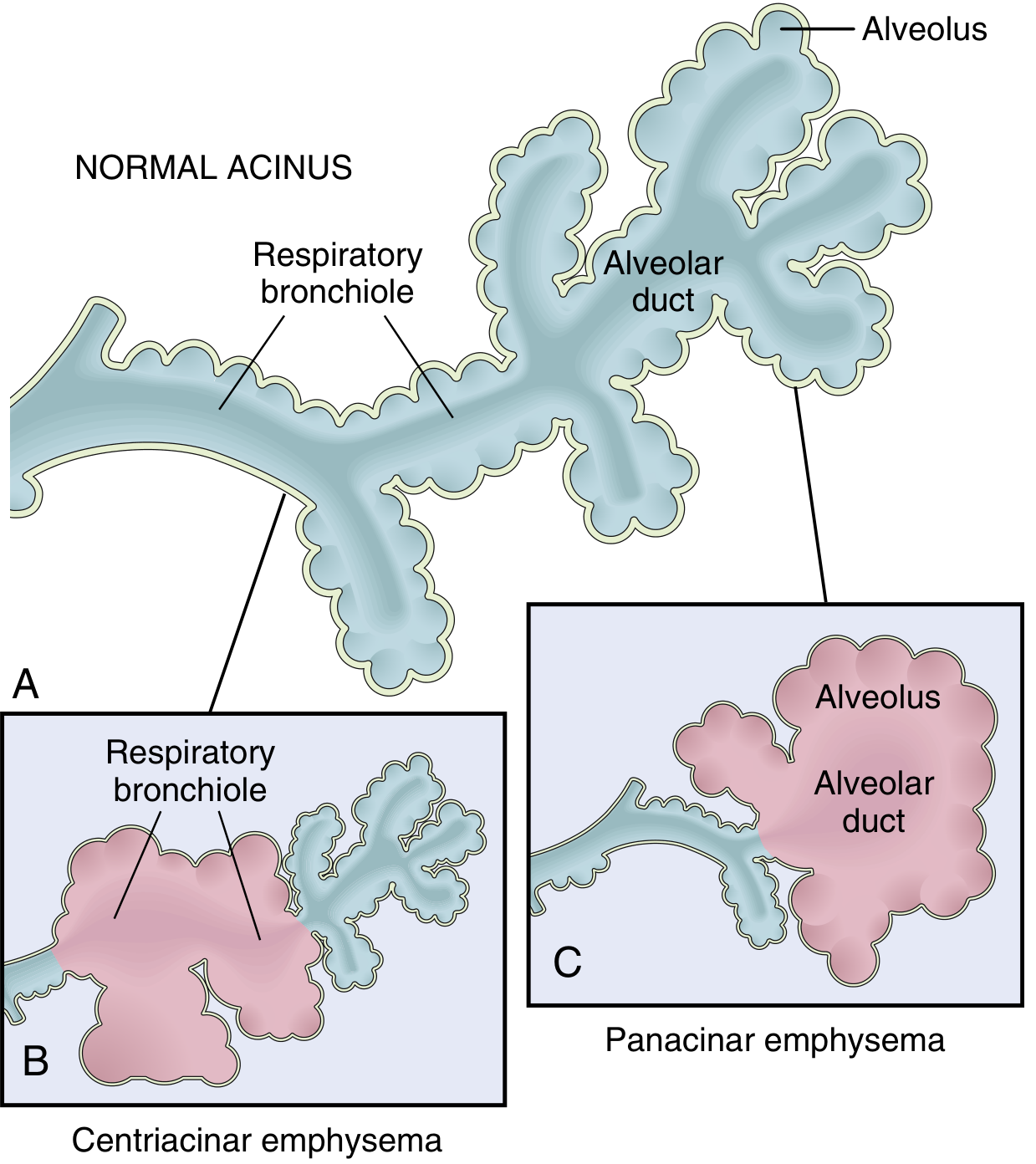
Classification (anatomic patterns)
-
Centriacinar (centrilobular)
- Proximal acinus (respiratory bronchiole) affected, distal alveoli initially spared
- Upper lobes/apices more severe
- Strongly associated with smoking
- Most common clinically significant type (>95% in some series)
-
Panacinar (panlobular)
- Uniform enlargement of entire acinus (bronchiole to terminal alveoli)
- More common in lower zones and anterior margins, severe at bases
- Associated with A1AT deficiency; worsened by smoking
-
Distal acinar (paraseptal)
- Distal acinus involved near pleura/septa
- Can form bullae; linked to spontaneous pneumothorax in young adults
-
Irregular (paracicatricial)
- Irregular acinar involvement with scarring
- Usually asymptomatic/incidental
- Robbins & Kumar Basic Pathology, pp. 255-275
- Robbins, Cotran & Kumar Pathologic Basis of Disease, pp. 634-635
Pathogenesis (core exam mechanism)
Main concept: protease-antiprotease imbalance + oxidative stress + inflammation
-
Toxic injury and inflammation
- Smoke/noxious particles injure epithelium and trigger macrophage + neutrophil-rich inflammation
- Mediators: IL-8, TNF, leukotriene B4, others
-
Protease-antiprotease imbalance
- Proteases (especially elastase) digest alveolar septal matrix
- Inadequate antiprotease protection (especially in A1AT deficiency) permits progressive destruction
-
Oxidative stress
- Oxidants from smoke and inflammatory cells directly damage tissue
- Oxidants also inactivate antiproteases and amplify inflammation
-
A1AT deficiency biology
- Severe deficiency (e.g., PiZZ) causes low anti-elastase defense
- Leads to early, severe panacinar emphysema, markedly accelerated by smoking
- Robbins, Cotran & Kumar Pathologic Basis of Disease, p. 636
Morphology
Gross
- Lungs are overdistended, pale, voluminous; may overlap heart
- Bullae/blebs may be present, especially paraseptal disease
Microscopy
-
Enlarged airspaces with thinning/rupture of alveolar septa
-
Loss of alveolar capillary bed and elastic recoil framework
-
Mild chronic inflammation may be present in bronchioles/alveolar walls
-
Robbins, Cotran & Kumar Pathologic Basis of Disease, pp. 634-635
Pathophysiology
- Loss of elastic recoil -> reduced expiratory driving pressure
- Small airway collapse during expiration -> airflow limitation, air trapping
- Hyperinflation -> increased work of breathing, flattened diaphragm
- Reduced gas-exchange surface -> impaired diffusion in advanced disease
- V/Q mismatch may cause hypoxemia (late stages)
Clinical features
- Progressive exertional dyspnea, prolonged expiration
- Minimal sputum compared with chronic bronchitis-predominant COPD
- Barrel chest, use of accessory muscles, weight loss in advanced cases
- Hyperresonant chest, diminished breath sounds, wheeze may occur
- Late: respiratory failure, pulmonary hypertension, cor pulmonale (less early than bronchitis phenotype)
Investigations
- Spirometry: persistent airflow obstruction (↓FEV1, ↓FEV1/FVC)
- Lung volumes: hyperinflation (↑TLC, ↑RV)
- DLCO: reduced (especially emphysema-predominant disease)
- CXR: hyperlucent lungs, flattened diaphragm
- HRCT: distribution/type of emphysema (centrilobular, panlobular, paraseptal)
- A1AT level/genotype in early onset, family history, basilar-predominant disease, or nonsmokers
Complications
- Recurrent infective exacerbations
- Pneumothorax (especially bullous/paraseptal)
- Respiratory failure
- Pulmonary hypertension and cor pulmonale (advanced COPD)
- Cachexia, reduced exercise tolerance, mortality increase
Management (short exam-oriented points)
- Smoking cessation (most effective disease-modifying step)
- Bronchodilators: LABA/LAMA ± SABA relief
- Inhaled corticosteroids in selected COPD phenotypes/exacerbators
- Pulmonary rehabilitation, nutrition optimization
- Vaccination (influenza, pneumococcal)
- Long-term oxygen therapy for chronic hypoxemia
- A1AT augmentation therapy in selected severe deficiency cases
- Surgical/interventional: bullectomy, lung volume reduction, endobronchial valves, lung transplantation in advanced disease
Diagram to draw in exam (high yield)
Draw normal acinus vs centriacinar vs panacinar and label:
- respiratory bronchiole
- alveolar duct
- alveoli
- proximal/distal acinar involvement
One-page conclusion line for viva/written
“Emphysema is a smoking- and A1AT deficiency-related destructive airspace disease characterized by irreversible distal airspace enlargement, loss of alveolar septa, reduced elastic recoil, expiratory airflow limitation, and progressive hyperinflation, with centriacinar (smoking-related) and panacinar (A1AT-related) patterns being most clinically significant.”
This is a shared conversation. Sign in to Orris to start your own chat.