Tirofiben in ACS
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of tirofiban in ACS:
Tirofiban in Acute Coronary Syndromes
Drug Class
Tirofiban is a GP IIb/IIIa receptor antagonist — one of three parenteral agents in the class (alongside abciximab and eptifibatide).
Mechanism of Action
GP IIb/IIIa (integrin αIIbβ3) is the most abundant receptor on platelets (~80,000 copies/platelet). On resting platelets it is inactive, but upon activation, inside-outside signalling causes conformational change that allows it to bind fibrinogen and von Willebrand factor — bridging adjacent platelets and driving aggregation.
Tirofiban is a nonpeptidic, small-molecule tyrosine derivative that acts as an RGD (Arg-Gly-Asp) mimetic, blocking the fibrinogen-binding site on αIIbβ3 and thereby preventing platelet aggregation. Unlike abciximab, tirofiban is specific for GP IIb/IIIa and does not inhibit the αvβ3 (vitronectin) or αMβ2 receptors.
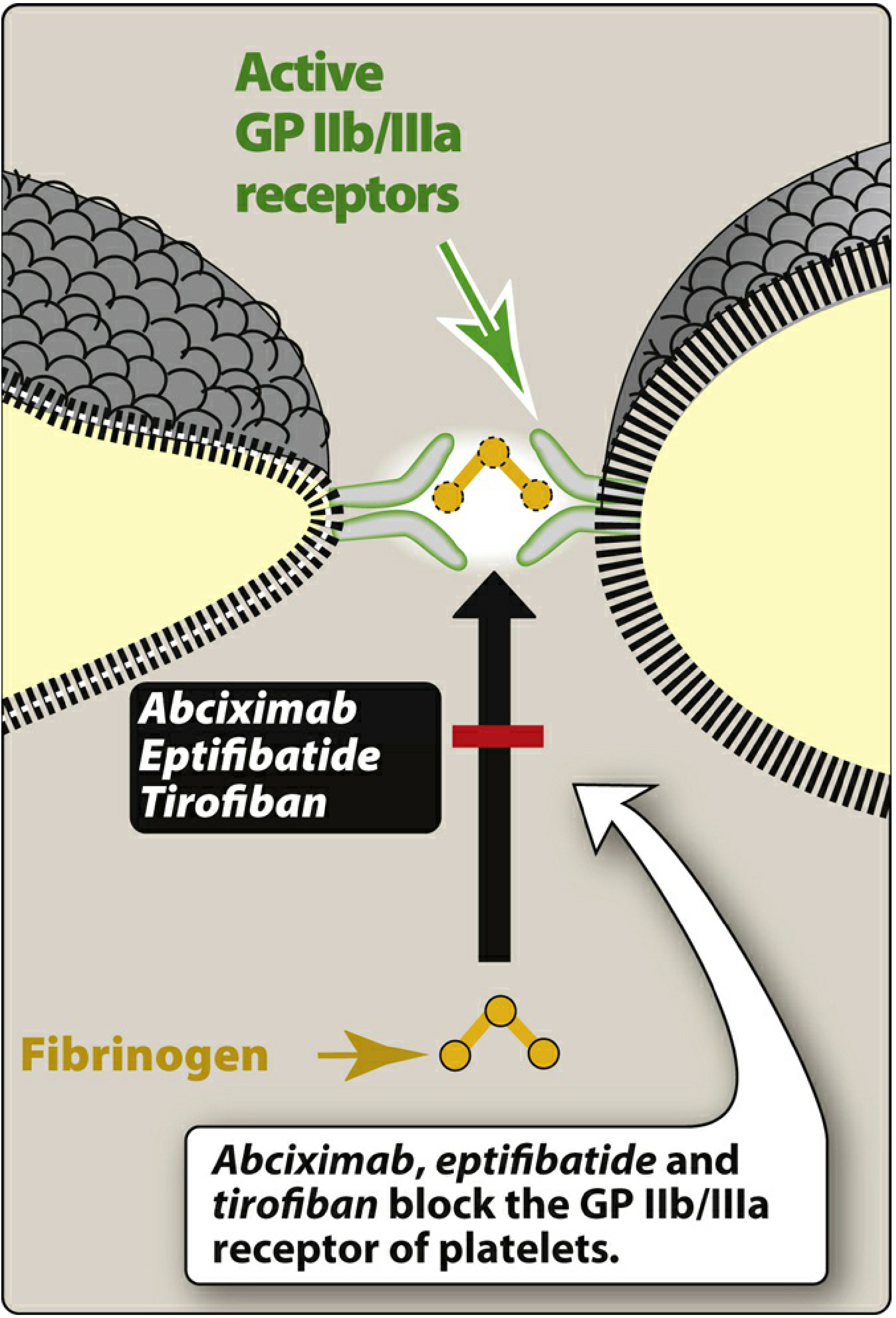
Pharmacokinetics
| Feature | Tirofiban |
|---|---|
| Nature | Nonpeptidic RGD-mimetic |
| GPIIb/IIIa specific? | Yes |
| Plasma t½ | ~2.0 hours |
| Platelet-bound t½ | Short (seconds) |
| Clearance | Renal (dose-reduce if CrCl <60 mL/min) |
| Route | IV bolus + infusion |
- Dose: 25 µg/kg IV bolus followed by 0.15 µg/kg/min infusion for up to 18 hours.
- Renal adjustment: Infusion dose reduced by 50% in renal insufficiency (CrCl <60 mL/min).
- On stopping the infusion, tirofiban is rapidly cleared from plasma; platelet function recovers within approximately 4 hours.
Indications in ACS
- NSTE-ACS (UA/NSTEMI): Tirofiban and eptifibatide are used in high-risk patients with unstable angina or NSTEMI.
- PCI: Used adjunctively in patients undergoing PCI, particularly in the setting of acute MI, no-reflow, or thrombotic complications during intervention.
- Use has declined with the availability of potent oral P2Y₁₂ inhibitors (prasugrel, ticagrelor).
Adverse Effects
| Effect | Details |
|---|---|
| Bleeding | Major side effect; risk increases with concurrent anticoagulants |
| Thrombocytopenia | ~1% of patients; immune-mediated (antibodies against neoantigens on GP IIb/IIIa) |
Thrombocytopenia with tirofiban is less common than with abciximab (which occurs in up to 5%).
Comparison with Other GP IIb/IIIa Inhibitors
| Abciximab | Eptifibatide | Tirofiban | |
|---|---|---|---|
| Type | Humanized mAb Fab fragment | Cyclic KGD heptapeptide | Nonpeptidic RGD-mimetic |
| GPIIb/IIIa specific | No | Yes | Yes |
| Plasma t½ | Short (minutes) | ~2.5 h | ~2.0 h |
| Platelet-bound t½ | Long (days) | Short (seconds) | Short (seconds) |
| Renal dose reduction | No | Yes | Yes |
| Thrombocytopenia | Up to 5% | ~0.5–1% | ~1% |
Key Clinical Points
- Tirofiban is given IV only — always in combination with aspirin and heparin.
- Its short platelet-bound half-life means platelet function recovers quickly after stopping the infusion (unlike abciximab, where inhibition persists for up to 2 weeks).
- Contraindications are similar to fibrinolytics (active bleeding, recent surgery, prior stroke, severe hypertension, etc.).
- Overall use of GP IIb/IIIa inhibitors has decreased as potent oral antiplatelet agents have become standard.
Sources: Goodman & Gilman's Pharmacological Basis of Therapeutics; Braunwald's Heart Disease; Lippincott Illustrated Reviews Pharmacology; Goldman-Cecil Medicine
This is a shared conversation. Sign in to Orris to start your own chat.