Hi
Hi! How can I help you today?
Wallerian degeneration
Reading File
Finding Sources
Searching PubMed
Wallerian degeneration mechanism axon
Finding Sources
Searching Images
Wallerian degeneration axon nerve
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive overview of Wallerian degeneration drawn from authoritative neurology texts.
Wallerian Degeneration
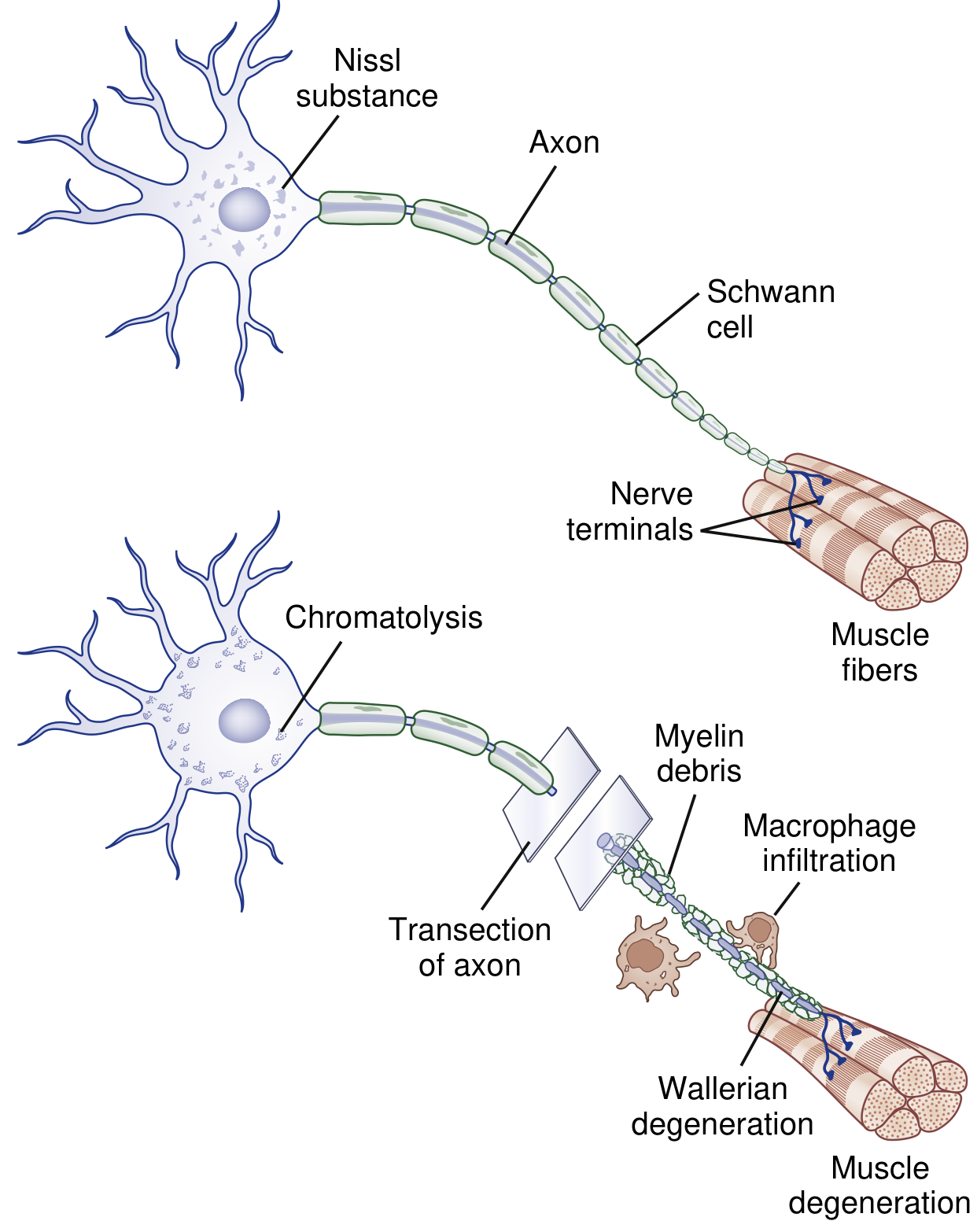
Named after Augustus Waller (1850), Wallerian degeneration is the stereotyped response of a peripheral axon and its myelin sheath distal to the site of axonal interruption. It follows grade II–V nerve injuries (i.e., any injury severe enough to disrupt axonal continuity).
Pathophysiology
Wallerian vs. axonal degeneration: Wallerian degeneration "dies forward" — the nerve degenerates from the point of axonal damage outward toward the periphery. This contrasts with axonal degeneration ("dying-back" neuropathy), which progresses proximally, typically in metabolic/toxic disorders like diabetes.
Distal segment changes
- Earliest event: disruption of anterograde and retrograde axonal transport immediately after injury.
- Ion influx: rapid inflow of Ca²⁺ and Na⁺ through the disrupted axonal membrane activates proteases and a cell-death-like cascade.
- Cytokine cascade: leukocyte recruitment and cytokine-mediated signaling trigger synthesis of neurotrophins, chemokines, extracellular matrix molecules, proteolytic enzymes, and interleukins.
- Day 3: Schwann cells retract from the node of Ranvier; activated Schwann cells and macrophages begin to digest myelin.
- ~1 week: the entire distal axonal process of Wallerian degeneration is complete.
- Myelin breakdown: myelin fragments into blocks/ovoids ("digestion chambers of Cajal") containing axon remnants; macrophages convert these into neutral fats and cholesterol esters and transport them to the bloodstream.
Proximal segment changes
- A limited degree of axon breakdown extends proximal to the injury, up to the first node of Ranvier.
- The cell body undergoes chromatolysis: breakup/dispersion of rough ER, eccentric displacement of the nucleus, and upregulation of transcription factors that switch gene expression from axon maintenance → protein synthesis.
- Very proximal injuries (e.g., proximal arm amputation) may cause apoptosis of the cell body itself.
Key Cellular Players
| Cell | Role |
|---|---|
| Schwann cells | Retract from nodes of Ranvier, dedifferentiate, upregulate c-Jun, transition to "repair cells," phagocytose myelin debris, secrete neurotrophins (cadherins, laminins, NCAM) to guide regeneration |
| Macrophages | Recruited by cytokines, phagocytose myelin/axon debris, convert lipid fragments for clearance |
| Neuronal cell body | Undergoes chromatolysis; shifts from maintenance to regenerative protein synthesis |
Comparison with Other Nerve Injury Responses
| Process | Site of damage | Axon status | Cause |
|---|---|---|---|
| Wallerian degeneration | Distal to axonal cut | Disrupted | Trauma (Grade II–V) |
| Segmental demyelination | Local internode(s) | Intact | Mild compression/traction |
| Axonal degeneration | Distal-to-proximal ("dying-back") | Diffusely affected | Metabolic/toxic (diabetes, renal failure) |
Nerve Regeneration After Wallerian Degeneration
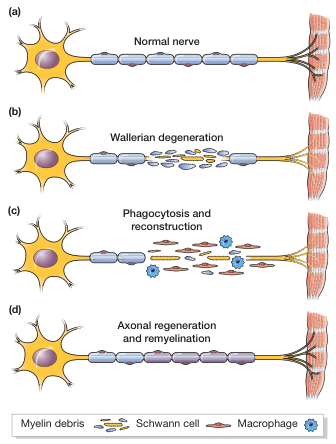
- Regeneration from the proximal stump begins only after Wallerian degeneration is complete.
- Schwann cells form bands of Büngner — longitudinal cords that guide the regenerating axon tip distally.
- Axon sprouts arise from nodes of Ranvier (nodal sprouts) or nerve terminals (terminal sprouts) within ~4 days.
- Rate of regeneration: approximately 1–3 mm/day.
- Collateral sprouting (from surviving axons) provides earlier functional recovery in partial injuries (3–6 months).
- New myelin is thinner, with more internodes per original internode.
- If regeneration fails, permanent denervation and target organ atrophy result.
Clinical Relevance
- Electrodiagnostics: after Wallerian degeneration, distal nerve stimulation fails to produce a CMAP/SNAP (both amplitudes fall), distinguishing it from conduction block (where distal stimulation still works).
- Sensory nerve special case: destruction of the dorsal root proximal to the dorsal root ganglion causes Wallerian degeneration of the posterior columns (central), but NOT of the peripheral sensory nerve — because the ganglion cell body maintains the distal axon's integrity.
- CNS Wallerian degeneration: also occurs in the CNS (e.g., descending corticospinal tract degeneration after stroke or motor neuron injury), but CNS regeneration is far more limited due to inhibitory myelin-associated proteins and absence of Schwann cell-mediated support.
Sources: Bradley and Daroff's Neurology in Clinical Practice, p. 1334–1336; Adams and Victor's Principles of Neurology 12th Ed., p. 1290.
This is a shared conversation. Sign in to Orris to start your own chat.