Tirofiban in acute coronary syndrome
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of tirofiban in acute coronary syndrome (ACS):
Tirofiban in Acute Coronary Syndrome
Class
Tirofiban belongs to the glycoprotein (GP) IIb/IIIa receptor antagonists, along with abciximab and eptifibatide. It is a nonpeptidic, small-molecule RGD mimetic (tyrosine derivative) — distinct from eptifibatide (a cyclic peptide) and abciximab (a monoclonal antibody fragment).
Mechanism of Action
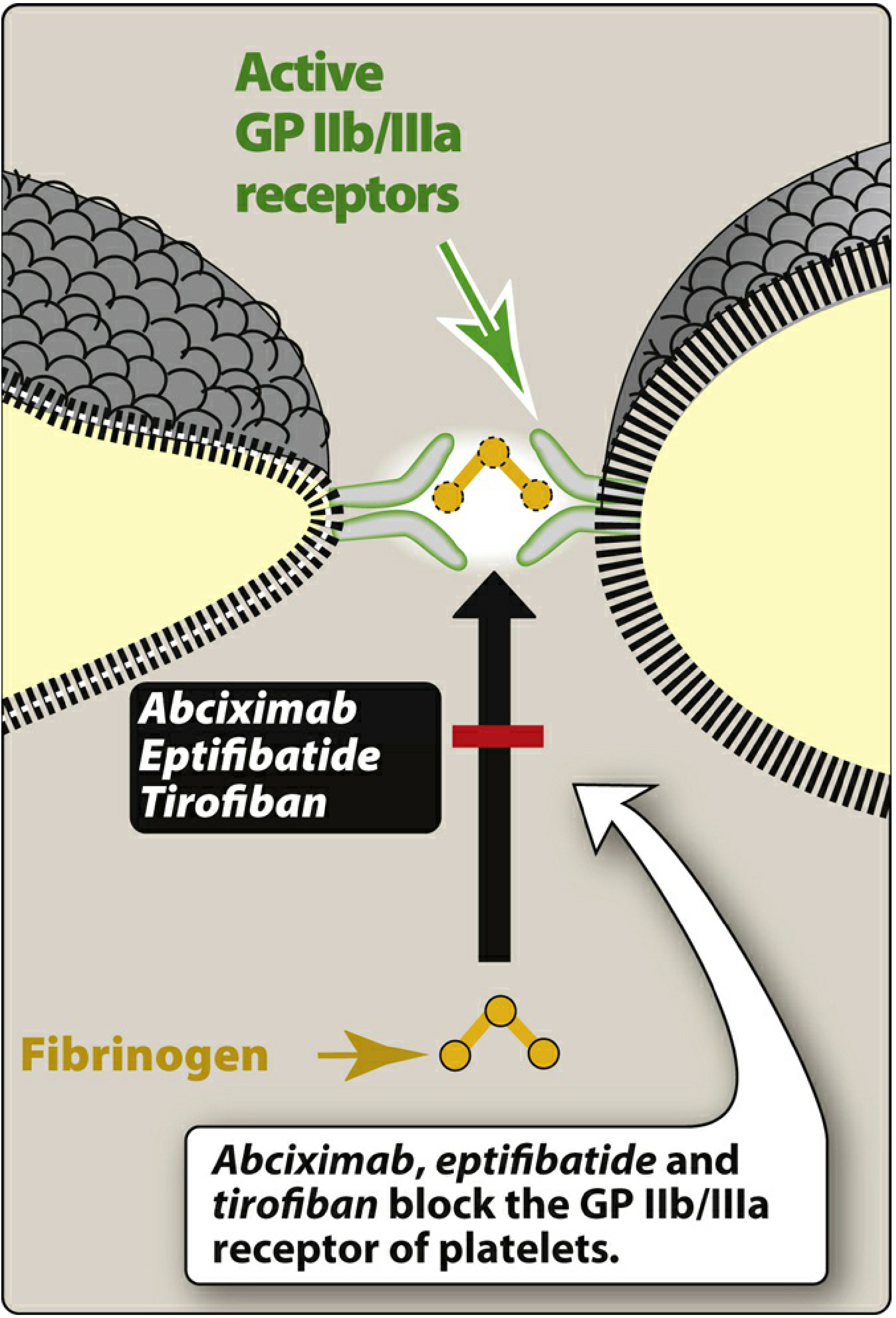
Figure: Abciximab, eptifibatide, and tirofiban block the GP IIb/IIIa receptor of platelets, preventing fibrinogen binding and platelet aggregation. — Lippincott Illustrated Reviews: Pharmacology, p. 445
GP IIb/IIIa (integrin αIIbβ3) is the most abundant receptor on platelets (~80,000 copies/platelet). On resting platelets it is inactive, but upon platelet activation, conformational changes allow it to bind fibrinogen and von Willebrand factor (vWF), bridging adjacent platelets to drive aggregation.
Tirofiban competitively blocks the fibrinogen/vWF binding site on GPIIb/IIIa by acting as an RGD (Arg-Gly-Asp) mimetic. Unlike abciximab, tirofiban is specific for GPIIb/IIIa and does not inhibit the αvβ3 (vitronectin) or αMβ2 (leukocyte) receptors. This mechanism is entirely distinct from aspirin, P2Y₁₂ inhibitors (clopidogrel, ticagrelor), or PAR-1 inhibitors. — Goodman & Gilman's Pharmacological Basis of Therapeutics
Indications in ACS
- NSTE-ACS (unstable angina / NSTEMI): Tirofiban and eptifibatide are the preferred GP IIb/IIIa agents for high-risk patients with unstable angina/NSTEMI managed medically or proceeding to PCI.
- PCI (percutaneous coronary intervention): Used adjunctively in patients undergoing PCI, particularly in acute MI settings or complex interventions (e.g., no-reflow phenomenon).
- All three GP IIb/IIIa agents are given intravenously along with heparin and aspirin.
- Overall use has declined with the advent of more potent oral P2Y₁₂ inhibitors (prasugrel, ticagrelor). — Braunwald's Heart Disease; Goldman-Cecil Medicine
Dosing
| Parameter | Tirofiban |
|---|---|
| Administration | IV bolus + infusion |
| Bolus | 25 µg/kg IV |
| Infusion | 0.15 µg/kg/min for up to 18 h |
| Renal dose adjustment | Reduce infusion by 50% if CrCl < 60 mL/min |
— Goodman & Gilman's Pharmacological Basis of Therapeutics
Pharmacokinetics
| Feature | Tirofiban |
|---|---|
| Structure | Nonpeptidic RGD mimetic |
| Plasma t½ | ~2.0 hours |
| Platelet-bound t½ | Short (seconds) — rapid dissociation |
| Clearance | Primarily renal (excreted largely unchanged), minor fecal |
| Specificity | GPIIb/IIIa specific (yes) |
| Recovery after stopping | Rapid — functional platelet recovery within hours |
The short platelet-bound half-life means that once the infusion is stopped, platelet function recovers quickly — a practical advantage over abciximab, which remains platelet-bound for up to 2 weeks. — Braunwald's Heart Disease; Goodman & Gilman's
Adverse Effects
- Bleeding — the major risk, particularly when combined with anticoagulants (heparin). Frequency varies from 1–10% depending on co-therapy intensity.
- Thrombocytopenia — immune-mediated, due to antibodies against neoantigens exposed on GPIIb/IIIa upon drug binding. Occurs in ~1% of patients (less common than with abciximab, which causes thrombocytopenia in up to 5%).
Contraindications mirror those for fibrinolytic therapy (active bleeding, recent surgery, severe hypertension, etc.).
Comparison with Other GP IIb/IIIa Agents
| Feature | Abciximab | Eptifibatide | Tirofiban |
|---|---|---|---|
| Type | Humanized mAb Fab | Cyclic KGD heptapeptide | Nonpeptidic RGD mimetic |
| GPIIb/IIIa specific | No | Yes | Yes |
| Plasma t½ | Short (min) | 2.5 h | 2.0 h |
| Platelet-bound t½ | Long (days) | Short (sec) | Short (sec) |
| Renal clearance | No | Yes | Yes |
| Thrombocytopenia | ~5% | ~0.5–1% | ~1% |
| Preferred use | STEMI/PCI | NSTEMI/PCI | NSTE-ACS/PCI |
— Goodman & Gilman's Pharmacological Basis of Therapeutics, Table 36-4; Braunwald's Heart Disease, Table 95.2
Key Clinical Points
- Tirofiban is administered only intravenously — there is no oral formulation.
- Dose reduction is mandatory in renal impairment (CrCl < 60 mL/min).
- Tirofiban is used when rapid, reversible platelet inhibition is needed — its short platelet-bound half-life facilitates faster recovery should urgent surgery be required.
- Due to competitive binding, platelet transfusion is less effective at reversing tirofiban compared to abciximab (free drug displaces donor platelets from receptors).
- The role of GP IIb/IIIa inhibitors has narrowed in contemporary ACS management with universal adoption of potent P2Y₁₂ inhibitors; they remain relevant in catheterization lab "bailout" scenarios (e.g., thrombus burden, no-reflow) and when oral pretreatment has not been given. — Goldman-Cecil Medicine; Tintinalli's Emergency Medicine
This is a shared conversation. Sign in to Orris to start your own chat.